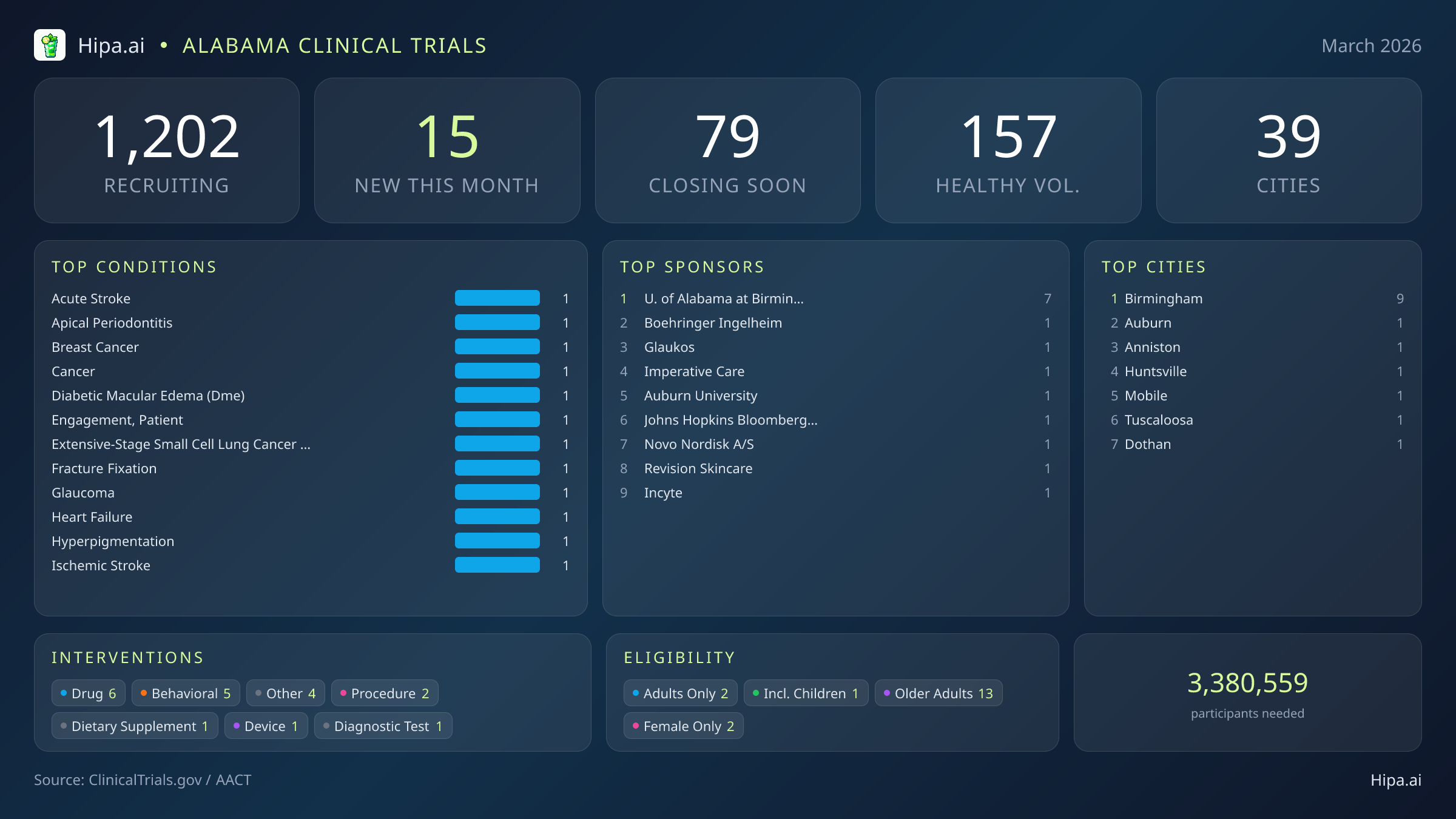
Alabama Clinical Trials Report — March 2026
15 New Studies, 79 Closing Soon
Hipa.ai Research - March 1, 2026 - Source: Research Data
Monthly Clinical Trials Infographic
Immediate Enrollment Deadlines Across Alabama
Time is strictly limited for Alabama residents hoping to join several critical medical research initiatives. Over the next ninety days, 79 clinical trials will permanently close their enrollment windows statewide. This impending deadline creates a distinct opportunity for individuals without underlying health conditions to participate in scientific discovery, as 16 of these closing studies are actively seeking healthy volunteers. For patients currently managing specific diagnoses, the window to access experimental therapies and behavioral interventions is rapidly narrowing. Final enrollment phases are approaching quickly for studies focused on the following conditions:
- Acute kidney injury
- Alzheimer's blood biomarkers
- Anatomic stage III breast cancer
- Alcohol use disorder
- Acute respiratory infections
Major Pharmaceutical and Late-Stage Clinical Launches
March brings a revitalized wave of high-profile research to the state, with 15 new trials opening this month—a steady increase from the twelve studies launched in January. Industry leaders are driving significant late-stage clinical development, offering local patients access to experimental treatments before they reach the broader market. Six of these newly launched studies are focused on drug interventions, while others explore cutting-edge medical devices and surgical procedures. Several notable therapeutic trials from major sponsors are currently seeking participants:
- Novo Nordisk is recruiting six hundred participants for a Phase 3 trial (NCT07503210) investigating whether weekly subcutaneous injections of NNC0487-0111 can help individuals with excess body weight safely maintain their weight loss.
- Boehringer Ingelheim has launched a Phase 3 study (NCT07472517) testing obrixtamig in combination with standard treatments like atezolizumab, carboplatin, and etoposide for adults with advanced small cell lung cancer to determine if the regimen improves survival rates.
- Glaukos Corporation is conducting a Phase 3 trial (NCT07495852) to evaluate the safety and efficacy of a second-generation travoprost intracameral implant designed for patients managing glaucoma.
- Imperative Care, Inc. is seeking seven hundred fifty volunteers for a large-scale device study (NCT07491952) evaluating a continuous dual aspiration neurothrombectomy system for first-line acute stroke treatment.
- Incyte Corporation is enrolling participants in a Phase 1 trial (NCT07441694) to determine the safety, tolerability, and maximum tolerated dose of INCA036978 for individuals diagnosed with myeloproliferative neoplasms.
Academic Innovation and Targeted Interventions
Beyond pharmaceutical testing, academic and medical institutions are launching highly targeted behavioral and procedural studies this month. The University of Alabama at Birmingham is sponsoring seven of the new trials, cementing its role as a central hub for medical advancement in the South. Among their new initiatives is an adaptive behavioral study (NCT07446907) testing intensive dietary patterns and resistance exercise to promote weight loss while preserving lean muscle mass in older adults with obesity. UAB researchers are also initiating a unique Phase 2 trial (NCT07457632) that combines tauroursodeoxycholate with traditional Chinese medicine to treat diabetic macular edema, aiming to provide a more comfortable alternative to current vascular therapies.
National academic partnerships are also expanding into the state. Johns Hopkins Bloomberg School of Public Health has opened a Phase 3 trial (NCT07458230) focusing on the severe problem of surgical site infections following high-energy bone fractures. Because these complications can affect up to sixty percent of patients with severe injuries, this study utilizes next-generation sequencing to rapidly identify infections and improve wound-healing outcomes.
March's new clinical trials are also targeting a highly specific array of chronic and acute conditions. Researchers are actively seeking patients recently diagnosed with or currently managing apical periodontitis, heart failure, and extensive-stage small cell lung cancer.
Statewide Access and Research Hubs
Alabama's clinical research infrastructure remains highly accessible, with 1,202 active trials currently recruiting across thirty-nine cities and nearly six hundred individual research sites. Geographically, new research opportunities are heavily clustered in the state's largest medical district. Birmingham leads the state with nine newly opened trials, providing a dense concentration of advanced care options. However, residents living outside the immediate metropolitan area still have opportunities to participate. New trials have opened their doors this month in several key municipalities across the state:
- Auburn
- Anniston
- Huntsville
- Mobile
- Tuscaloosa
- Dothan
Broad Eligibility and Healthy Volunteer Opportunities
Eligibility criteria for this month's new studies are heavily skewed toward adult and senior populations, ensuring that aging demographics can participate in medical advancement. Older adults have extensive options, with thirteen of the new trials specifically designing their protocols to include senior participants. Pediatric research is represented by a single new study open to children, while two trials restrict participation exclusively to female patients. None of the new trials launched this month restrict participation exclusively to men.
For those without a specific medical diagnosis, one of this month's newly launched trials is actively welcoming healthy volunteers. This brings the total number of open, recruiting trials for healthy participants across Alabama to 157. These trials rely heavily on healthy individuals to establish crucial baseline data for future medical breakthroughs, making community participation essential for scientific progress.
As the spring research season advances, Alabama's clinical trial landscape is positioned to expand its focus on metabolic therapies and advanced oncological treatments, while the integration of next-generation sequencing in procedural medicine will likely shape the future of post-surgical infection management.
Data Highlights
Conditions Closing Soon
- stress (2)
- acute kidney injury (2)
- obesity (2)
- anxiety (2)
- cognitive impairment (2)
- alzheimer blood biomarkers (1)
- alcohol use disorder (1)
- acute respiratory infections (aris) (1)
Most Common New Trial Conditions
- acute stroke (1)
- apical periodontitis (1)
- breast cancer (1)
- cancer (1)
- diabetic macular edema (dme) (1)
- engagement, patient (1)
- extensive-stage small cell lung cancer (es-sclc) (1)
- fracture fixation (1)
Cities With the Most New Trials
- Birmingham (9)
- Auburn (1)
- Anniston (1)
- Huntsville (1)
- Mobile (1)
- Tuscaloosa (1)
- Dothan (1)
Leading Sponsors
- University of Alabama at Birmingham (7)
- Boehringer Ingelheim (1)
- Glaukos Corporation (1)
- Imperative Care, Inc. (1)
- Auburn University (1)
- Johns Hopkins Bloomberg School of Public Health (1)
- Novo Nordisk A/S (1)
- Revision Skincare (1)
| Month | New Trials | Closing Soon |
|---|---|---|
| November 2025 | 20 | 3 |
| December 2025 | 40 | 19 |
| January 2026 | 19 | 5 |
| February 2026 | 12 | 6 |
| March 2026 | 15 | 18 |
| April 2026 | 5 | 0 |