Arizona Clinical Trials Report — March 2026
19 New Studies, 93 Closing Soon
Hipa.ai Research - March 1, 2026 - Source: Research Data
Monthly Clinical Trials Infographic
Immediate Deadlines for Arizona Residents
Time is running out for Arizonans looking to participate in several major medical research initiatives. Within the next ninety days, 93 clinical trials across the state will permanently close their enrollment windows. This deadline presents a unique opportunity for people without underlying medical conditions to contribute to scientific advancement, as 21 of these soon-to-close studies are actively seeking healthy volunteers. Patients currently managing specific diagnoses also face a rapidly shrinking window to access experimental therapies and behavioral interventions. Final enrollment phases are approaching quickly for studies focused on the following conditions:
- Post-traumatic stress disorder
- Coronary artery disease
- High-grade glioma
- Mild cognitive impairment
- Barrett's esophagus and esophageal adenocarcinoma
Major Pharmaceutical and Late-Stage Clinical Launches
Despite the impending closures, March brings a fresh wave of high-profile research to the state, heavily driven by pharmaceutical innovation. Of the new trials launched this month, 11 are focused on drug interventions. Industry leaders like Novo Nordisk and Boehringer Ingelheim are initiating large-scale Phase 3 studies, offering local patients access to late-stage experimental treatments before they reach the broader market. Several notable therapeutic trials are currently seeking participants:
- Novo Nordisk is recruiting four hundred participants for a Phase 3 trial (NCT07481630) investigating whether weekly injections of NNC0487-0111 can help individuals with excess body weight and knee osteoarthritis lose weight and reduce joint pain.
- Boehringer Ingelheim has launched a Phase 3 study (NCT07497087) testing nerandomilast in adults with systemic sclerosis, aiming to improve symptoms and slow disease progression.
- EyePoint Pharmaceuticals is conducting a Phase 3 trial (NCT07449923) comparing the efficacy of EYP-1901 against aflibercept for patients with diabetic macular edema.
- Veru Inc. is seeking two hundred volunteers for a Phase 2 study (NCT07446998) evaluating enobosarm's effect on total body weight and physical function in patients currently taking GLP-1 receptor agonists.
- Rivus Pharmaceuticals is enrolling one hundred eighty participants in a Phase 2 trial (NCT07491458) to assess HU6 as a potential treatment for metabolic dysfunction-associated steatohepatitis.
- Tectonic Operating Company is running a targeted Phase 2 study (NCT07473700) testing TX000045 in patients suffering from pulmonary hypertension secondary to interstitial lung disease.
Massive Observational Studies and Targeted Conditions
Beyond pharmaceutical testing, academic and medical institutions are launching massive observational and behavioral studies this month. The Mayo Clinic is spearheading an enormous descriptive study (NCT07455370) aiming to enroll over twenty-one thousand nursing staff members to examine levels of moral distress, moral injury, and post-traumatic stress. Meanwhile, Dartmouth College is sponsoring a nationwide effort (NCT07454733) that includes Arizona sites, recruiting eight hundred participants to determine if video recordings of multidisciplinary clinic visits can improve the quality of life for patients with amyotrophic lateral sclerosis and their caregivers.
March's new clinical trials are also targeting a highly specific array of chronic and genetic conditions. Researchers are actively seeking patients recently diagnosed with or currently managing asthma exacerbations, autism spectrum disorder, astigmatism, aphakia, and colorectal cancer. Specialized genetic research is also expanding, with new studies focusing on individuals with CTNNB1 and APC gene mutations.
Statewide Access and Research Hubs
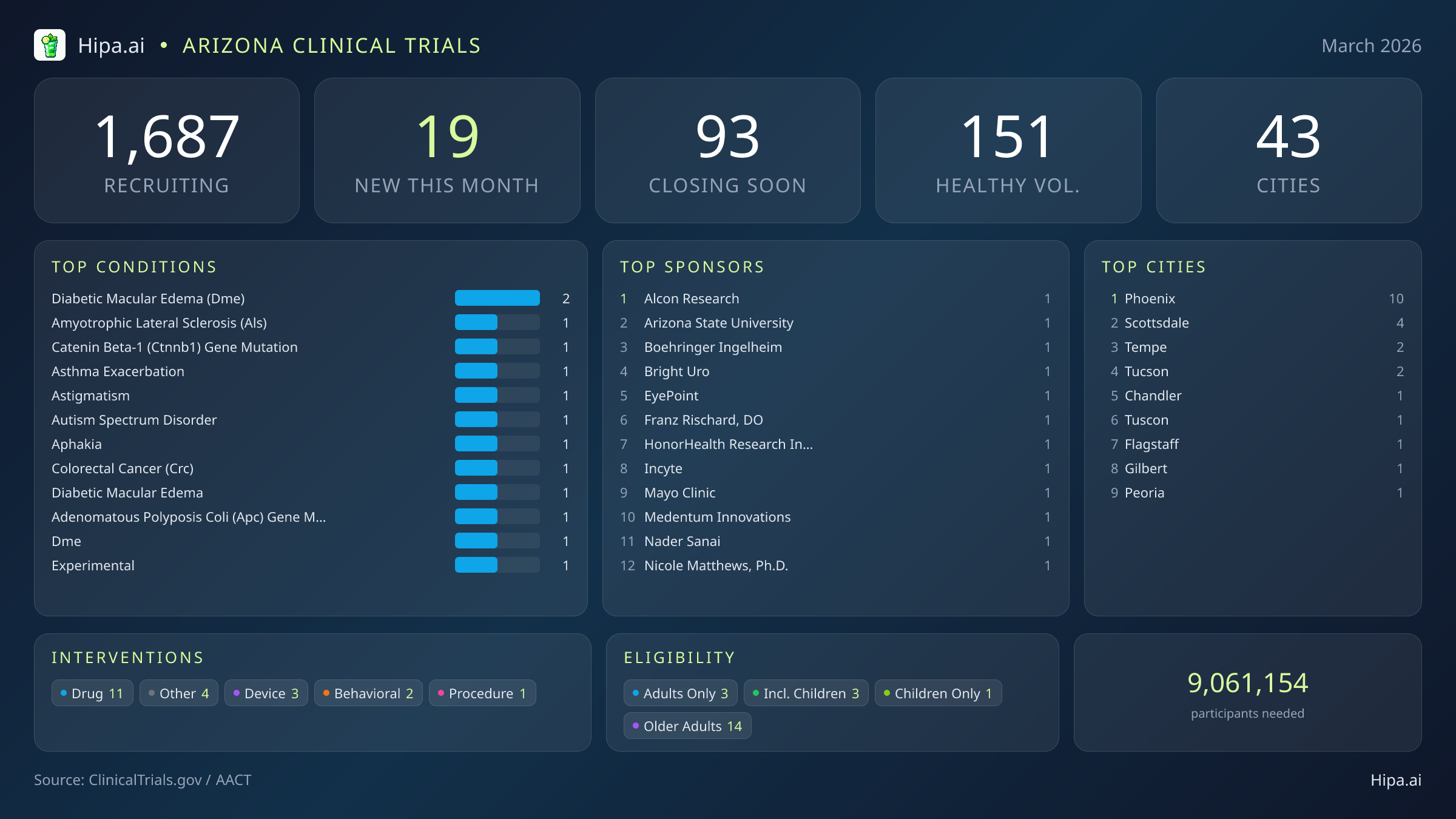
Arizona's clinical research infrastructure remains robust and highly accessible, with 1,687 active trials currently recruiting across forty-three cities and over one thousand individual research sites. While the pace of new study launches has cooled slightly since the beginning of the year—dropping from thirty new trials in January to 19 this month—the geographic distribution ensures that residents across the state have access to cutting-edge medical research.
Geographically, new research opportunities are heavily clustered in the Phoenix metropolitan area. Phoenix leads the state with ten newly opened trials, while neighboring cities provide additional access points. However, residents living outside the immediate capital region still have opportunities to participate. New trials have opened their doors this month in several key municipalities:
- Phoenix
- Scottsdale
- Tempe
- Tucson
- Chandler
- Flagstaff
- Gilbert
- Peoria
Broad Eligibility and Healthy Volunteer Opportunities
Eligibility criteria for this month's new studies are notably inclusive, ensuring that diverse demographic groups can participate in medical advancement. Older adults have extensive options, with fourteen of the new trials specifically designing their protocols to include senior populations. Pediatric research is also represented, as three new studies are open to children and adolescents. None of the new trials launched this month restrict participation exclusively by sex, ensuring equal access for both male and female participants.
For those without a specific medical diagnosis, 4 of this month's new trials are actively welcoming healthy volunteers. This brings the total number of open, recruiting trials for healthy participants across Arizona to 151. Whether driven by industry partners like Alcon Research and Incyte Corporation, or academic institutions like Arizona State University, these trials rely heavily on healthy individuals to establish crucial baseline data for future medical breakthroughs.
As the spring research season progresses, Arizona's clinical trial landscape is positioned to expand its focus on metabolic and respiratory therapies, while the massive datasets collected from this month's behavioral studies will likely shape the next generation of healthcare workforce support systems.
Data Highlights
Conditions Closing Soon
- post traumatic stress disorder (4)
- healthy participants (2)
- coronary artery disease (2)
- high grade glioma (2)
- aging (2)
- mild cognitive impairment (2)
- barrett esophagus (2)
- barretts esophagus with dysplasia (2)
Most Common New Trial Conditions
- diabetic macular edema (dme) (2)
- amyotrophic lateral sclerosis (als) (1)
- catenin beta-1 (ctnnb1) gene mutation (1)
- asthma exacerbation (1)
- astigmatism (1)
- autism spectrum disorder (1)
- aphakia (1)
- colorectal cancer (crc) (1)
Cities With the Most New Trials
Leading Sponsors
- Alcon Research (1)
- Arizona State University (1)
- Boehringer Ingelheim (1)
- Bright Uro (1)
- EyePoint Pharmaceuticals, Inc. (1)
- Franz Rischard, DO (1)
- HonorHealth Research Institute (1)
- Incyte Corporation (1)
| Month | New Trials | Closing Soon |
|---|---|---|
| November 2025 | 25 | 11 |
| December 2025 | 59 | 37 |
| January 2026 | 29 | 7 |
| February 2026 | 29 | 10 |
| March 2026 | 19 | 30 |
| April 2026 | 1 | 0 |