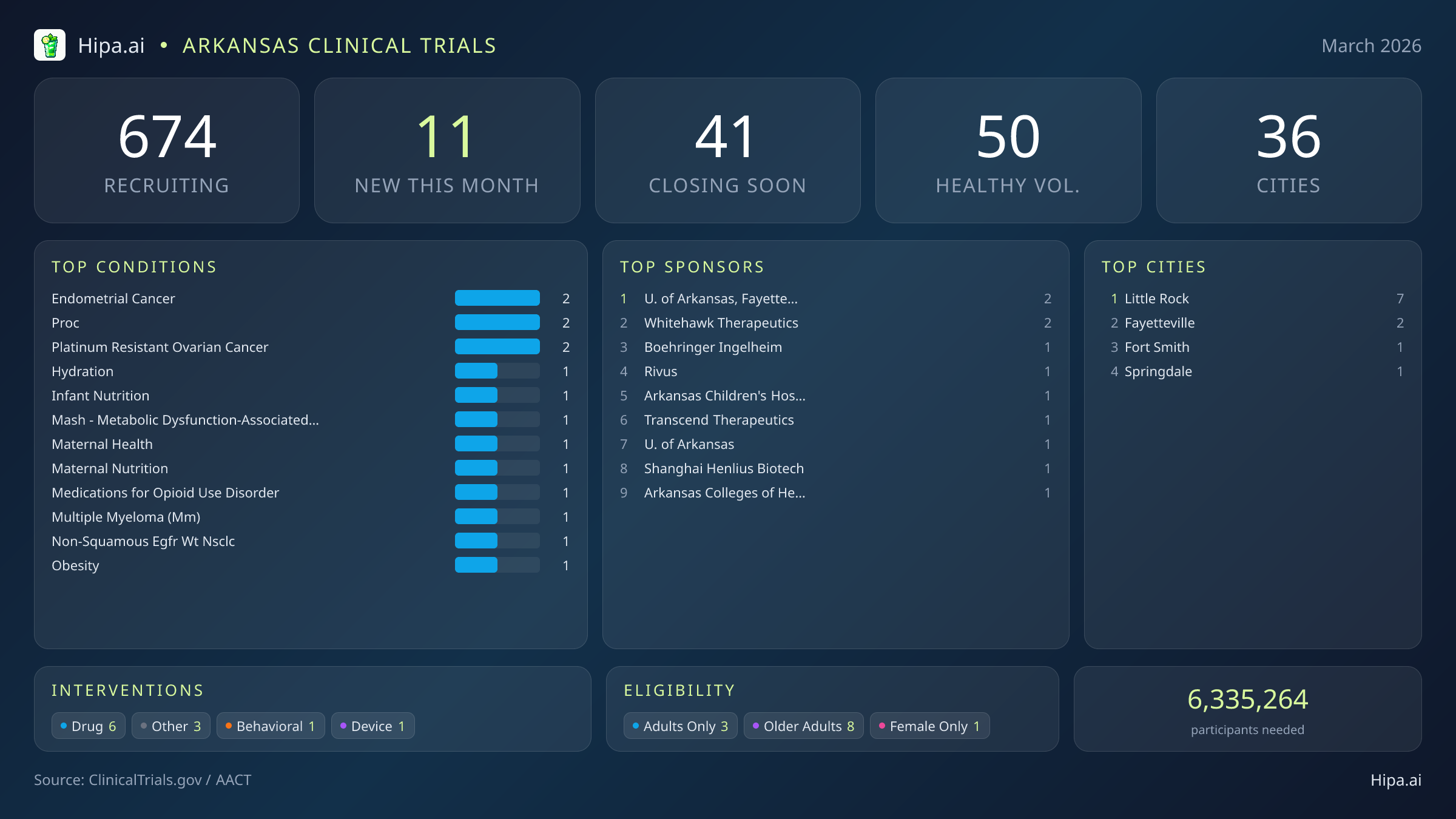
Arkansas Clinical Trials Report — March 2026
11 New Studies, 41 Closing Soon
Hipa.ai Research - March 1, 2026 - Source: Research Data
Monthly Clinical Trials Infographic
Imminent Deadlines for Arkansas Research Studies
Time is strictly limited for Arkansas residents seeking to participate in several critical medical studies. Over the next 90 days, researchers will permanently close enrollment for 41 clinical trials across the state. This closing window presents a distinct opportunity for individuals without underlying health conditions to step forward, as four of these concluding studies are actively seeking healthy volunteers. For patients managing specific diagnoses, the chance to access these experimental therapies is rapidly disappearing. Final enrollment phases are approaching quickly for studies focused on the following conditions:
- Stage IV lung cancer
- Bipolar disorder (manic phase)
- Advanced or metastatic solid tumor malignancies
- Anatomic stage II and III breast cancer
- Atopic dermatitis
- Asthma
Major Pharmaceutical Launches and Late-Stage Clinical Development
March brings a revitalized wave of high-profile research to the state, with 11 new trials opening this month—a noticeable increase from the eight studies launched in February. Industry leaders are driving significant late-stage clinical development, offering local patients access to experimental treatments before they reach the broader market. Six of these newly launched studies are focused on drug interventions, while others explore behavioral therapies and medical devices. Several notable therapeutic trials from major sponsors are currently seeking participants:
- Boehringer Ingelheim has launched a Phase 3 study (NCT07472517) testing obrixtamig in combination with standard treatments like atezolizumab, carboplatin, and etoposide. This large-scale trial is open to adults with advanced small cell lung cancer to determine if the regimen improves survival rates compared to standard chemotherapy alone.
- Transcend Therapeutics is evaluating the safety and efficacy of a new compound for mental health in a Phase 3 trial (NCT07456696). This study will randomize participants to receive weekly doses of TSND-201 or a placebo during a four-week treatment period to treat post-traumatic stress disorder.
- Rivus Pharmaceuticals, Inc. is conducting a Phase 2 trial (NCT07491458) to assess the safety and tolerability of HU6. This study targets patients diagnosed with metabolic dysfunction-associated steatohepatitis, measuring changes in liver fat alongside overall pharmacokinetics.
- Whitehawk Therapeutics, Inc. is enrolling participants in two separate Phase 1 trials (NCT07470853 and NCT07444814) to evaluate targeted antibody-drug conjugates for adult participants with advanced or metastatic solid tumors. These studies employ sequential dose escalation and expansion designs to test MUC16-directed and PTK7-directed therapies.
- Shanghai Henlius Biotech has opened a Phase 1 study (NCT07477587) comparing the pharmacokinetic characteristics and safety of HLX15-SC versus DARZALEX FASPRO in newly diagnosed multiple myeloma patients who are ineligible for transplant.
Academic Innovation and Targeted Interventions
Beyond pharmaceutical testing, academic and medical institutions are launching highly targeted behavioral and procedural studies this month. The University of Arkansas and its affiliated research centers are sponsoring several new trials, cementing their role as central hubs for medical advancement in the region. Among their new initiatives is a comprehensive behavioral study (NCT07490717) developing innovative care pathways for screening and treating opioid use disorder and post-traumatic stress disorder in jail populations. This research addresses the critical period following release from incarceration, aiming to mitigate the risk of opioid overdose and concurrent stimulant use among justice-involved individuals.
The Arkansas Colleges of Health Education is also initiating a unique Phase 2 trial (NCT07487090) exploring the cardiovascular benefits of fasted versus fed state exercise. This study provides a lifestyle-based alternative for individuals seeking natural solutions to heart disease and obesity management, bypassing the costs and side effects associated with traditional medications.
March's new clinical trials are also targeting a highly specific array of chronic and acute conditions. Researchers are actively seeking patients recently diagnosed with or currently managing:
- Endometrial cancer
- Platinum-resistant ovarian cancer
- Metabolic dysfunction-associated steatohepatitis
- Opioid use disorder
- Multiple myeloma
- Maternal health and infant nutrition
Statewide Access and Research Hubs
Arkansas maintains a robust and highly accessible clinical research infrastructure, with 674 active trials currently recruiting across 36 cities and 334 individual research sites. Geographically, new research opportunities are heavily clustered in the state's largest medical districts. Little Rock leads the state with seven newly opened trials, providing a dense concentration of advanced care options for residents in the central region. However, individuals living outside the immediate metropolitan area still have opportunities to participate in cutting-edge research. New trials have opened their doors this month in several key municipalities across the state:
- Little Rock
- Fayetteville
- Fort Smith
- Springdale
Broad Eligibility and Healthy Volunteer Opportunities
Eligibility criteria for this month's new studies are heavily skewed toward adult and senior populations, ensuring that aging demographics can participate in medical advancement. Older adults have extensive options, with eight of the new trials specifically designing their protocols to include senior participants. One trial restricts participation exclusively to female patients, focusing on maternal health and nutrition, while none of the new trials launched this month restrict participation exclusively to men or focus on pediatric populations.
For those without a specific medical diagnosis, four of this month's newly launched trials are actively welcoming healthy volunteers. This brings the total number of open, recruiting trials for healthy participants across Arkansas to 50. These trials rely heavily on healthy individuals to establish crucial baseline data for future medical breakthroughs, making community participation essential for scientific progress.
As the spring research season advances, the Arkansas clinical trial landscape is positioned to expand its focus on targeted oncology therapies and metabolic interventions, while the integration of behavioral health pathways in justice-involved populations will likely shape the future of addiction recovery protocols.
Data Highlights
Conditions Closing Soon
- stage iv lung cancer ajcc v8 (2)
- bipolar disorder, manic (2)
- adiposity (1)
- advanced lung non-small cell carcinoma (1)
- advanced or metastatic solid tumor malignancies (1)
- anatomic stage ii breast cancer ajcc v8 (1)
- asthma (1)
- anatomic stage iii breast cancer ajcc v8 (1)
Most Common New Trial Conditions
- endometrial cancer (2)
- proc (2)
- platinum resistant ovarian cancer (2)
- hydration (1)
- infant nutrition (1)
- mash - metabolic dysfunction-associated steatohepatitis (1)
- maternal health (1)
- maternal nutrition (1)
Cities With the Most New Trials
- Little Rock (7)
- Fayetteville (2)
- Fort Smith (1)
- Springdale (1)
Leading Sponsors
- University of Arkansas, Fayetteville (2)
- Whitehawk Therapeutics, Inc. (2)
- Boehringer Ingelheim (1)
- Rivus Pharmaceuticals, Inc. (1)
- Arkansas Children's Hospital Research Institute (1)
- Transcend Therapeutics (1)
- University of Arkansas (1)
- Shanghai Henlius Biotech (1)
| Month | New Trials | Closing Soon |
|---|---|---|
| November 2025 | 9 | 1 |
| December 2025 | 22 | 11 |
| January 2026 | 15 | 3 |
| February 2026 | 8 | 2 |
| March 2026 | 11 | 19 |
| April 2026 | 1 | 0 |