California Clinical Trials Report — March 2026
116 New Studies, 407 Closing Soon
Hipa.ai Research - March 1, 2026 - Source: Research Data
Monthly Clinical Trials Infographic
Californians have a rapidly closing window to join hundreds of medical research initiatives across the state. Within the next ninety days, 407 clinical trials will permanently close their enrollment phases. For residents without underlying health conditions, this presents an immediate deadline to participate in scientific advancement, as 89 of these soon-to-close studies are actively seeking healthy volunteers. Patients currently managing specific diagnoses also face urgent timelines. Oncology research and behavioral health studies are particularly affected by this wave of closures. Final enrollment phases are rapidly approaching for studies focused on the following conditions:
- Colorectal cancer
- Acute myeloid leukemia
- Multiple myeloma
- Prostate cancer
- Anatomic stage III breast cancer
- Cholangiocarcinoma
- Major depressive disorder
Major Phase 3 and High-Capacity Research Launches
Despite the impending closures, March brings a robust wave of large-scale research projects to the state. ModernaTX, Inc. is currently recruiting healthy adults for a massive Phase 3 clinical trial evaluating a pandemic influenza vaccine. This undertaking aims to enroll four thousand participants to measure the immune response and safety profile of the mRNA-1018-H5 vaccine, offering residents a direct role in advancing global infectious disease preparedness. Beyond vaccine development, several major healthcare organizations and academic institutions are launching high-capacity studies that require thousands of participants across California.
- ModernaTX, Inc. is seeking participants for its Phase 3 pandemic influenza vaccine trial (NCT07496450) to evaluate humoral immunogenicity after two doses, as well as the safety and reactogenicity of the vaccine in adults.
- The University of California, San Francisco is enrolling four thousand four hundred fifty individuals for a pragmatic randomized controlled trial (NCT07469995) of Covered California's Grocery Support Program. The study will compare the efficacy of providing different benefit amounts to low-income participants with diabetes or hypertension.
- Kaiser Permanente has launched the INITIATE-HF study (NCT07467668), seeking three thousand participants to evaluate whether reminding doctors about treatment guidelines improves intravenous iron usage for hospitalized heart failure patients.
- Hoffmann-La Roche is developing a novel blood-based biomarker test (NCT07455136) to aid in the diagnosis and prognosis of traumatic brain injuries in adults, requiring two thousand volunteers.
- The San Francisco VA Health Care System is running dual studies (NCT07464834 and NCT07477964) testing whether mailed informational postcards can successfully prompt veterans to reconnect their cardiovascular implantable electronic home monitors.
- The University of California, Davis is evaluating the impact of documentary-style educational videos on cancer prevention behaviors among one thousand firefighters (NCT07494617).
- The University of Pittsburgh is recruiting seven hundred fifty participants to test a web-based decision support tool designed to provide evidence-based information about vasectomy and other birth control options (NCT07451067).
Statewide Access and Intervention Types
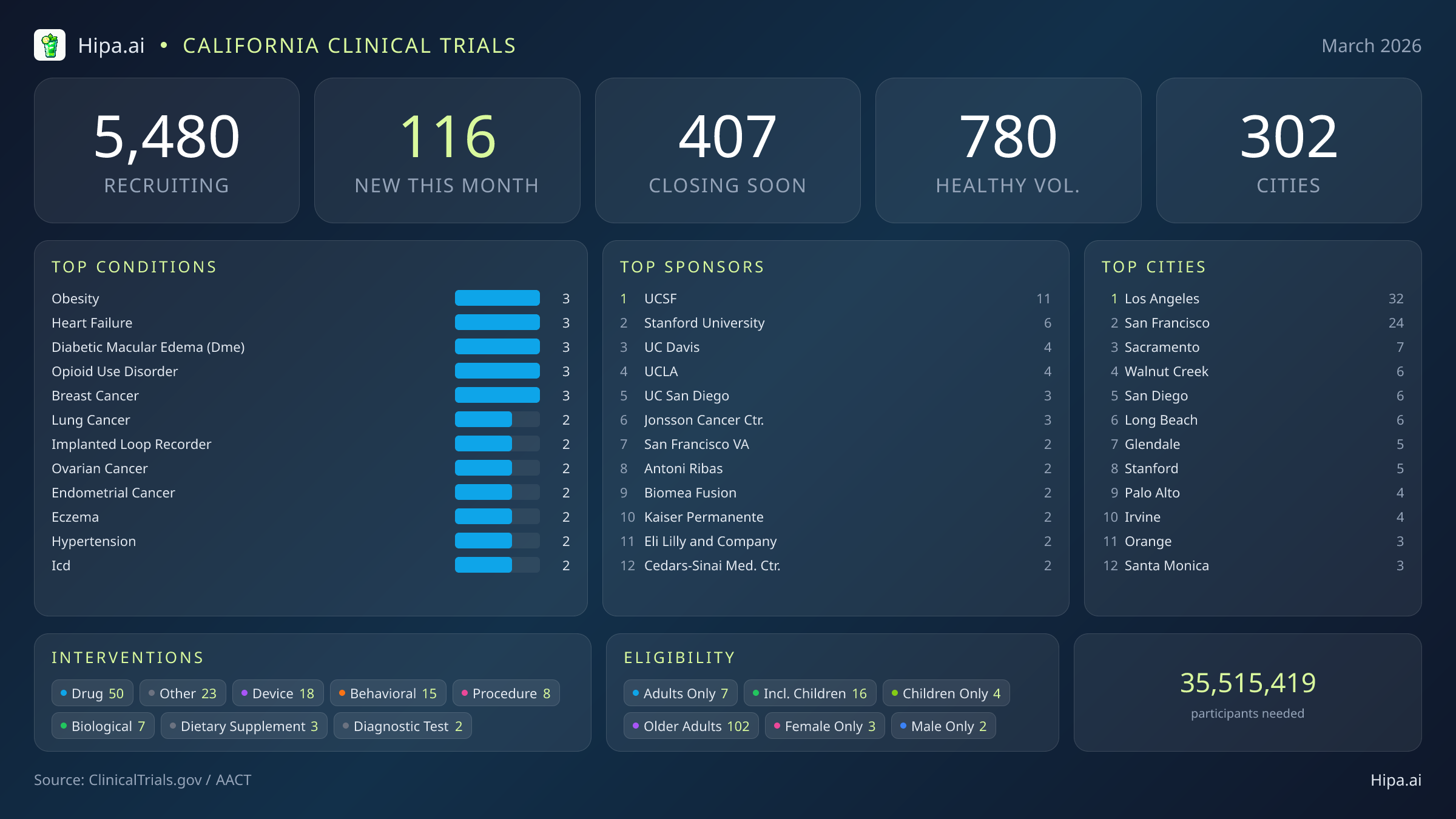
The clinical research infrastructure across the state remains highly accessible, with 5,480 active trials currently recruiting across three hundred two California cities and over five thousand individual research sites. While the pace of new study launches has cooled slightly since the beginning of the year—dropping from one hundred thirty-two new trials in January to 116 this month—the variety of interventions being tested remains diverse. Pharmaceutical interventions dominate the new landscape with 50 drug trials, followed by medical device evaluations, behavioral studies, procedural research, and biological testing.
Geographically, new research opportunities are heavily clustered in Southern California and the Bay Area. Los Angeles leads the state with thirty-two newly opened trials, while San Francisco follows closely with twenty-four. However, residents living outside these primary metropolitan hubs still have access to cutting-edge medical research. New trials have opened their doors this month in several other key municipalities:
- Sacramento
- Walnut Creek
- San Diego
- Long Beach
- Glendale
- Stanford
- Palo Alto
- Irvine
Targeted Diagnoses and Healthy Volunteer Opportunities
March's new clinical trials are targeting a wide array of chronic and acute conditions. Researchers are actively seeking patients recently diagnosed with or currently managing obesity, heart failure, diabetic macular edema, opioid use disorder, and breast cancer. Additional studies are focusing on lung cancer, ovarian cancer, endometrial cancer, and severe eczema. The sponsorship of these trials is evenly split, with fifty-four studies funded by industry partners like Moderna and Hoffmann-La Roche, and fifty-nine driven by academic or other institutional organizations.
Eligibility criteria for these new studies are notably broad, ensuring that diverse demographic groups can participate in medical advancement. Older adults have extensive options, with 102 of the new trials specifically designing their protocols to include senior populations. Pediatric research is also expanding, as sixteen new studies are open to children and adolescents. Highly targeted demographic research includes three trials exclusively for females and two exclusively for males. For those without a specific medical diagnosis, twenty-five of this month's new trials are actively welcoming healthy volunteers. This brings the total number of open, recruiting trials for healthy participants across California to 780.
Academic medical centers continue to serve as the primary engines for this clinical research. The University of California system is driving a significant portion of the state's scientific output, with UCSF, UC Davis, UCLA, and UC San Diego collectively sponsoring dozens of new protocols. Stanford University and the Jonsson Comprehensive Cancer Center also remain highly active in launching new therapeutic investigations across multiple medical disciplines.
Moving into the second quarter of the year, the clinical research ecosystem is poised to shift focus toward analyzing the massive datasets generated by this season's high-capacity behavioral and vaccine trials, while preparing for a new wave of targeted oncological interventions.
Data Highlights
Conditions Closing Soon
- healthy (12)
- colorectal cancer (7)
- acute myeloid leukemia (6)
- multiple myeloma (6)
- prostate cancer (6)
- healthy participants (5)
- major depressive disorder (5)
- cancer (5)
Most Common New Trial Conditions
- obesity (3)
- heart failure (3)
- diabetic macular edema (dme) (3)
- opioid use disorder (3)
- breast cancer (3)
- lung cancer (2)
- implanted loop recorder (2)
- ovarian cancer (2)
Cities With the Most New Trials
- Los Angeles (32)
- San Francisco (24)
- Sacramento (7)
- Walnut Creek (6)
- San Diego (6)
- Long Beach (6)
- Glendale (5)
- Stanford (5)
Leading Sponsors
- University of California, San Francisco (11)
- Stanford University (6)
- University of California, Davis (4)
- University of California, Los Angeles (4)
- University of California, San Diego (3)
- Jonsson Comprehensive Cancer Center (3)
- San Francisco VA Health Care System (2)
- Antoni Ribas (2)
| Month | New Trials | Closing Soon |
|---|---|---|
| November 2025 | 118 | 21 |
| December 2025 | 191 | 142 |
| January 2026 | 122 | 55 |
| February 2026 | 130 | 33 |
| March 2026 | 116 | 104 |
| April 2026 | 23 | 0 |