Colorado Clinical Trials Report — March 2026
18 New Studies, 102 Closing Soon
Hipa.ai Research - March 1, 2026 - Source: Research Data
Monthly Clinical Trials Infographic
Coloradans have a brief window to participate in several critical medical studies before they permanently close to new enrollment. Within the next ninety days, 102 clinical trials will conclude their recruitment phases across the state. For residents without underlying medical conditions, time is running out to secure one of the remaining spots, as 19 of these soon-to-close studies are actively seeking healthy volunteers to establish baseline health metrics. Patients managing specific diagnoses also face an immediate deadline to explore experimental therapies before these cohorts are finalized. Final enrollment phases are rapidly approaching for studies focused on the following conditions:
- Myasthenia gravis
- Breast cancer
- Multiple myeloma
- Paroxysmal atrial fibrillation
- Migraine
- Advanced solid tumors
- Advanced non-small cell lung cancer
- Acute respiratory distress syndrome
- Alzheimer's disease
- Allogeneic stem cell transplantation
Major Phase 2 and Phase 3 Pharmaceutical Launches
Despite the wave of impending closures, March brings a fresh slate of advanced-stage pharmaceutical research to the region. Major industry sponsors are launching Phase 2 and Phase 3 clinical trials, offering patients access to novel drug candidates for complex autoimmune, oncological, and ocular conditions. These high-capacity studies require hundreds of participants to accurately measure drug safety and efficacy.
- Boehringer Ingelheim is recruiting for a massive Phase 3 study (NCT07497087) evaluating Nerandomilast in adults with systemic sclerosis. The trial aims to enroll four hundred forty-eight participants to determine if the drug improves symptoms and slows disease progression in individuals with limited or diffuse cutaneous manifestations.
- Bristol-Myers Squibb has launched an open-label, multicenter Phase 2 trial (NCT07492680) testing BMS-986504. The study seeks two hundred sixty participants with advanced or metastatic solid tumors featuring an MTAP deletion, evaluating the drug both as a monotherapy and in combination with other anti-cancer agents.
- EyePoint Pharmaceuticals, Inc. is running dual Phase 3 randomized, double-masked studies (NCT07449923 and NCT07449936) comparing the efficacy of EYP-1901 against Aflibercept. Each trial requires two hundred forty patients diagnosed with diabetic macular edema.
- Eli Lilly and Company is sponsoring a Phase 2 master protocol (NCT07483073) to evaluate multiple new treatments for adults with ulcerative colitis or Crohn's disease. Participants will be assigned to specific sub-studies prior to randomization to evaluate the safety and efficacy of emerging therapies.
- A related Phase 2 sub-study by Eli Lilly (NCT07483099) will specifically compare the safety and efficacy of an FXR agonist known as LY4395089 combined with mirikizumab against mirikizumab alone in patients with moderately to severely active Crohn's disease.
Statewide Access and Intervention Types
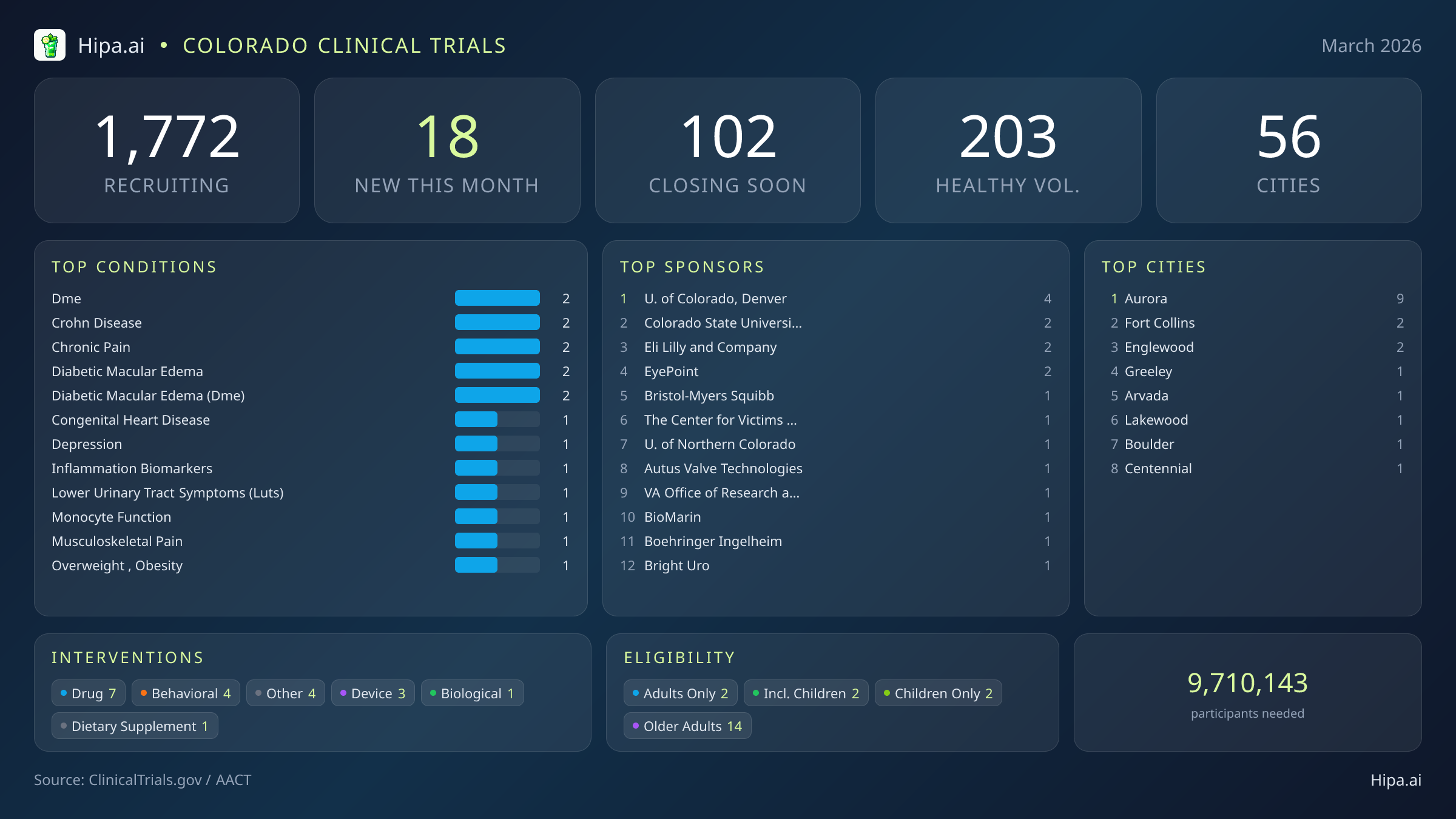
The clinical research infrastructure in Colorado remains robust and highly accessible. Currently, 1,772 active trials are recruiting across fifty-six cities and over one thousand individual research sites statewide. While the pace of new study launches has slowed slightly since the beginning of the year—dropping from thirty-two new trials in January to 18 this month—the variety of interventions being tested remains diverse. Pharmaceutical interventions dominate the new landscape with seven drug trials, followed by four behavioral studies, three medical device evaluations, one biological test, and one dietary supplement research initiative.
Geographically, new research opportunities are heavily concentrated around the Denver metropolitan area and major university hubs. Aurora leads the state with nine newly opened trials, largely driven by the presence of the University of Colorado, Denver. However, residents living outside the immediate capital region still have access to cutting-edge medical research. New trials have opened their doors this month in several other key municipalities:
- Fort Collins
- Englewood
- Greeley
- Arvada
- Lakewood
- Boulder
- Centennial
Targeted Diagnoses and Healthy Volunteer Opportunities
March's new clinical trials target a wide array of chronic and acute conditions. Researchers are actively seeking patients recently diagnosed with or currently managing diabetic macular edema, Crohn's disease, chronic pain, congenital heart disease, depression, and lower urinary tract symptoms. Additional studies are investigating inflammation biomarkers and monocyte function. The sponsorship of these trials is evenly split between commercial and academic entities, with nine studies funded by industry partners like Bristol-Myers Squibb and EyePoint Pharmaceuticals, Inc., and eight driven by academic or other institutional organizations such as Colorado State University, the University of Northern Colorado, and The Center for Victims of Torture.
Eligibility criteria for these new studies ensure that diverse demographic groups can participate in medical advancement. Older adults have extensive options, with 14 of the new trials specifically designing their protocols to include senior populations. Pediatric research is also represented, as two new studies are open to children and adolescents. Neither males nor females are excluded based on gender-specific protocols this month, ensuring broad access across the adult population. For those without a specific medical diagnosis, five of this month's new trials are actively welcoming healthy volunteers. This brings the total number of open, recruiting trials for healthy participants across Colorado to 203.
As the spring research calendar unfolds, the state's clinical trial ecosystem is positioned to expand its focus on advanced autoimmune therapies and targeted oncology treatments, paving the way for next-generation interventions to reach the clinic and potentially transform standard care protocols.
Data Highlights
Conditions Closing Soon
- myasthenia gravis (2)
- breast cancer (2)
- multiple myeloma (2)
- paroxysmal atrial fibrillation (2)
- migraine (2)
- advanced solid tumors (1)
- advanced nsclc (1)
- acute respiratory distress syndrome (1)
Most Common New Trial Conditions
- dme (2)
- crohn disease (2)
- chronic pain (2)
- diabetic macular edema (2)
- diabetic macular edema (dme) (2)
- congenital heart disease (1)
- depression (1)
- inflammation biomarkers (1)
Cities With the Most New Trials
- Aurora (9)
- Fort Collins (2)
- Englewood (2)
- Greeley (1)
- Arvada (1)
- Lakewood (1)
- Boulder (1)
- Centennial (1)
Leading Sponsors
- University of Colorado, Denver (4)
- Colorado State University (2)
- Eli Lilly and Company (2)
- EyePoint Pharmaceuticals, Inc. (2)
- Bristol-Myers Squibb (1)
- The Center for Victims of Torture, United States (1)
- University of Northern Colorado (1)
- Autus Valve Technologies, Inc. (1)
| Month | New Trials | Closing Soon |
|---|---|---|
| November 2025 | 29 | 7 |
| December 2025 | 42 | 43 |
| January 2026 | 26 | 9 |
| February 2026 | 30 | 5 |
| March 2026 | 18 | 29 |
| April 2026 | 4 | 0 |