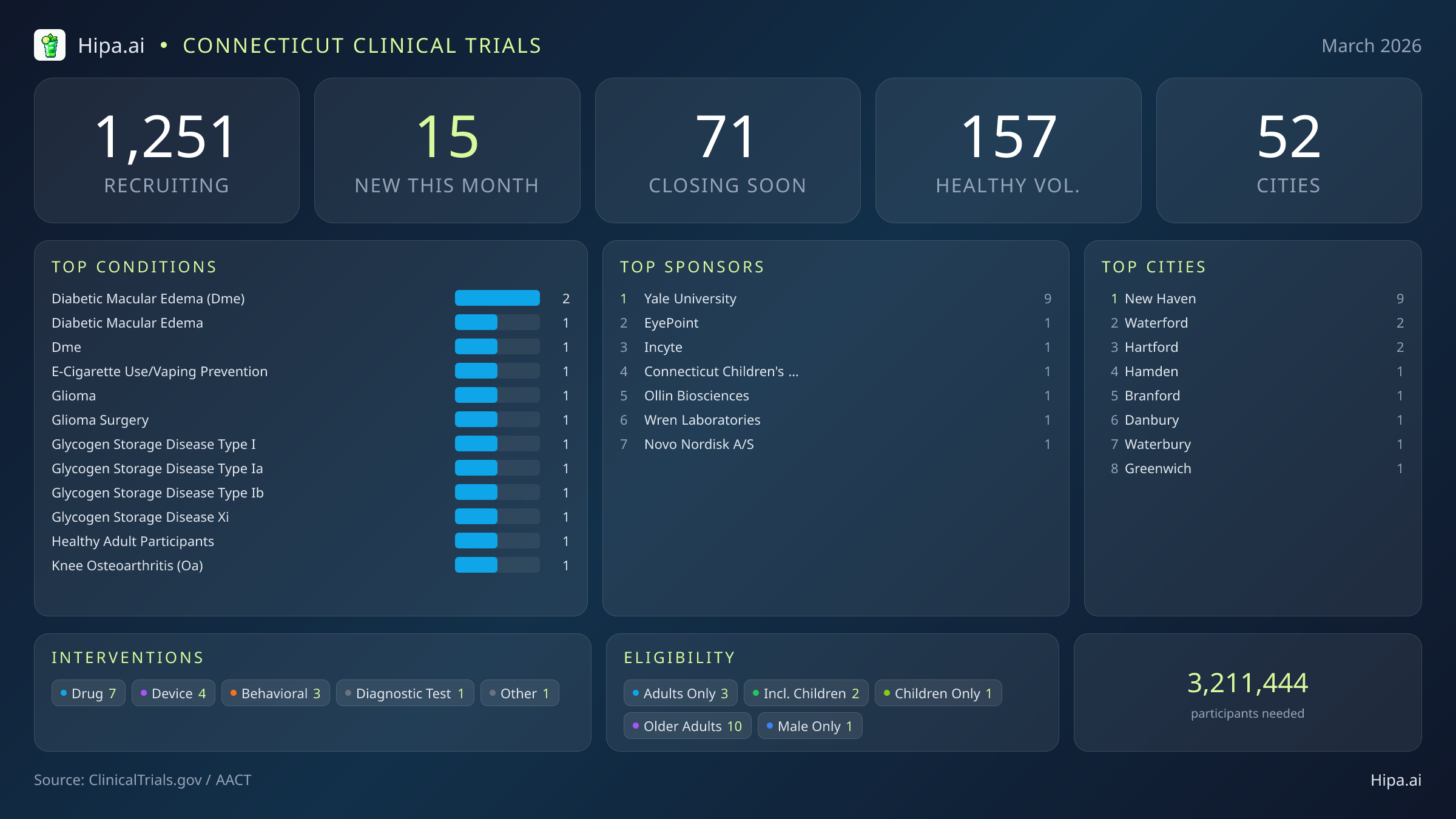
Connecticut Clinical Trials Report — March 2026
15 New Studies, 71 Closing Soon
Hipa.ai Research - March 1, 2026 - Source: Research Data
Monthly Clinical Trials Infographic
Immediate Enrollment Deadlines Across Connecticut
Time is strictly limited for Connecticut residents seeking to participate in several critical medical research initiatives. Over the next ninety days, 71 clinical trials will permanently close their enrollment windows across the state. This rapidly approaching deadline affects both patients seeking novel therapies and individuals without underlying medical conditions. Specifically, 13 of these soon-to-close studies are actively looking for healthy volunteers to provide essential baseline data before their participant rosters finalize.
Patients currently managing specific diagnoses have a brief window remaining to access experimental treatments and behavioral interventions. Final enrollment phases are closing quickly for studies focused on respiratory illnesses, behavioral health, and aggressive oncology targets. Researchers are urging eligible individuals to act immediately if they wish to participate in studies addressing the following conditions:
- COVID-19
- Mental health issues
- Multiple myeloma
- Obesity
- Stage IV lung cancer
- Advanced or metastatic solid tumor malignancies
- Alcohol use disorder
Major Pharmaceutical and Academic Research Launches
Despite a slight cooling in new study launches compared to the start of the year—dropping from thirty new trials in January to 15 this month—March brings a highly targeted wave of research to the state. Academic institutions and industry leaders are driving this month's medical innovation, with Yale University alone sponsoring nine new studies. The clinical landscape is diverse, with seven of the newly opened trials focusing on drug interventions, four evaluating medical devices, three exploring behavioral therapies, and one testing a new diagnostic tool. Several standout trials from major sponsors are currently seeking participants:
- Wren Laboratories LLC is recruiting fifteen hundred men for a massive observational study (NCT07454603) evaluating PROSTest, a blood-based molecular assay designed to assist with risk stratification and biopsy decisions. The study will enroll men referred for a prostate biopsy due to elevated PSA levels or abnormal examinations, collecting peripheral blood prior to the procedure.
- Yale University has launched a large-scale behavioral intervention (NCT07460622) seeking over twelve hundred middle school students. This novel school-based program uses virtual reality to deliver e-cigarette and vaping prevention content, alongside emotion regulation skills-building, directly into real-world classrooms.
- Novo Nordisk A/S is enrolling four hundred participants in a Phase 3 clinical trial (NCT07481630) to determine if weekly subcutaneous injections of NNC0487-0111 can effectively help individuals with excess body weight and knee osteoarthritis reduce joint pain and lose weight.
- EyePoint Pharmaceuticals, Inc. is conducting a Phase 3 study (NCT07449923) to compare the efficacy of EYP-1901 against aflibercept. This randomized, double-masked trial is seeking two hundred forty patients diagnosed with diabetic macular edema.
- Incyte Corporation is seeking over two hundred volunteers for a Phase 1 trial (NCT07441694) testing INCA036978. The study aims to determine the safety, tolerability, and maximum tolerated dose of the drug, administered both as a monotherapy and alongside standard disease-directed therapies for participants with myeloproliferative neoplasms.
- Yale University is running a highly specialized Phase 2 trial (NCT07458113) evaluating sacituzumab tirumotecan. This multi-site, open-label study is recruiting twenty patients with metastatic triple-negative breast cancer and active brain metastases to receive intravenous treatments on a 28-day cycle.
Targeted Conditions and Expanding Healthy Volunteer Access
March's newly launched trials address a highly specific array of medical conditions. Researchers are actively recruiting patients recently diagnosed with or currently managing diabetic macular edema, glioma, and various forms of glycogen storage disease, including types I, Ia, Ib, and XI. The focus on metabolic, oncological, and ophthalmic conditions provides fresh avenues for patients who may have exhausted standard care options.
Eligibility criteria for this month's research opportunities remain broad, ensuring diverse demographic participation. Older adults have extensive access, with ten of the new trials specifically designing their protocols to accommodate senior populations. Pediatric research is also moving forward, as two new studies are open to children and adolescents. Participation is generally open to all sexes, with only one new trial restricting its enrollment exclusively to male participants.
For individuals without a specific medical diagnosis, the opportunity to contribute to scientific advancement is particularly strong this month. Seven of the fifteen newly opened studies accept healthy participants. This brings the total number of actively recruiting trials for healthy volunteers across Connecticut to 157. These individuals are crucial for establishing baseline safety and efficacy metrics in both pharmaceutical testing and behavioral research.
Research Hubs and Statewide Accessibility
Connecticut maintains a dense and highly accessible clinical research infrastructure. Currently, 1,251 active trials are recruiting participants across 52 cities, utilizing 625 individual research sites. This geographic spread ensures that cutting-edge medical research is not entirely confined to major metropolitan hospitals, though certain academic hubs naturally lead the state in volume.
New Haven continues to dominate the state's research landscape, driven heavily by Yale University's expansive clinical network. Nine of this month's newly opened trials are located in New Haven, making it the primary destination for patients seeking access to the latest experimental therapies and massive behavioral studies. Residents outside the immediate New Haven area still have local opportunities to participate, as new trials have opened their doors this month in several other key municipalities across the state:
- Waterford
- Hartford
- Hamden
- Branford
- Danbury
- Waterbury
- Greenwich
As the spring research season accelerates, Connecticut's clinical trial ecosystem is positioned to make significant strides in metabolic therapies and adolescent behavioral health, while the massive datasets generated by this month's diagnostic studies will likely refine how oncologists approach early cancer detection nationwide.
Data Highlights
Conditions Closing Soon
- covid-19 (3)
- mental health issue (3)
- multiple myeloma (2)
- obesity (2)
- healthy volunteer (2)
- stage iv lung cancer ajcc v8 (2)
- cancer (2)
- alcohol use disorder (1)
Most Common New Trial Conditions
- diabetic macular edema (dme) (2)
- diabetic macular edema (1)
- dme (1)
- e-cigarette use/vaping prevention (1)
- glioma (1)
- glioma surgery (1)
- glycogen storage disease type i (1)
- glycogen storage disease type ia (1)
Cities With the Most New Trials
Leading Sponsors
- Yale University (9)
- EyePoint Pharmaceuticals, Inc. (1)
- Incyte Corporation (1)
- Connecticut Children's Medical Center (1)
- Ollin Biosciences, Inc. (1)
- Wren Laboratories LLC (1)
- Novo Nordisk A/S (1)
| Month | New Trials | Closing Soon |
|---|---|---|
| November 2025 | 17 | 3 |
| December 2025 | 31 | 33 |
| January 2026 | 21 | 9 |
| February 2026 | 27 | 9 |
| March 2026 | 15 | 12 |
| April 2026 | 4 | 0 |