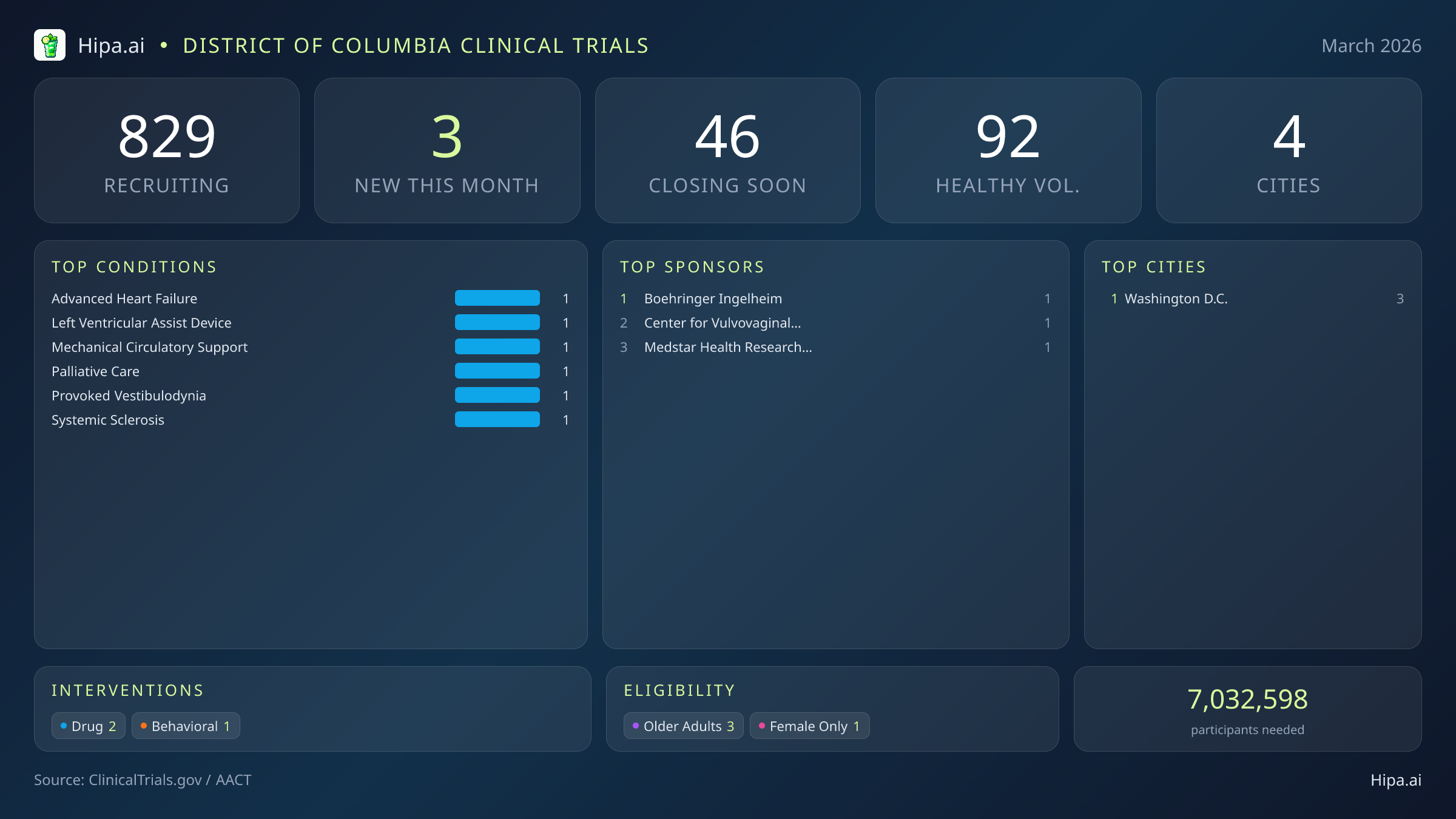
District of Columbia Clinical Trials Report — March 2026
3 New Studies, 46 Closing Soon
Hipa.ai Research - March 1, 2026 - Source: Research Data
Monthly Clinical Trials Infographic
Approaching Deadlines for District Residents
Time is strictly limited for residents of the nation's capital hoping to join several critical medical research initiatives. Within the next ninety days, 46 clinical trials will permanently close their enrollment windows across the District of Columbia. This impending deadline creates a narrow window for patients seeking access to experimental therapies before they advance to the next phase of regulatory review. Individuals without underlying health conditions also have a brief opportunity to contribute to scientific discovery, as five of these closing studies are actively seeking healthy volunteers. Final enrollment phases are rapidly approaching for research programs focused on the following conditions:
- Prostate cancer
- Stage IV lung cancer and advanced non-small cell carcinoma
- Alzheimer's disease
- Ambiguous lineage leukemia or lymphoma
- Anatomic stage II and III breast cancer
- Anxiety disorders
- Arrhythmia and cardiomyopathies
Patients currently managing these specific diagnoses must act quickly to secure a spot in these late-stage enrollment windows. The closure of these trials marks the transition from active patient recruitment to data analysis, meaning local access to these specific experimental interventions will soon be unavailable.
High-Profile Pharmaceutical and Clinical Launches
March brings a highly targeted wave of medical research to the district, with three new trials opening this month. This represents a sharp decrease from the sixteen studies launched in January and the fifteen studies opened in February, reflecting a seasonal shift toward highly specialized, late-stage clinical development from major industry and academic sponsors. Two of these newly launched studies are focused on drug interventions, offering local patients access to experimental treatments before they reach the broader market, while the third explores behavioral approaches to patient care. Several notable therapeutic trials are currently seeking participants:
- Boehringer Ingelheim is recruiting for a massive Phase 3 trial (NCT07497087) investigating whether nerandomilast helps people with systemic sclerosis. This study aims to enroll nearly four hundred and fifty participants to determine if the drug can improve symptoms and slow disease progression for adults with limited or diffuse cutaneous systemic sclerosis.
- The Center for Vulvovaginal Disorders has launched a Phase 2 study (NCT07486830) evaluating the safety and efficacy of IncobotulinumtoxinA for the treatment of provoked vestibulodynia associated with overactive pelvic floor muscle dysfunction. This double-blind, placebo-controlled trial is seeking premenopausal females to test this targeted intervention.
- Medstar Health Research Institute is sponsoring a new behavioral intervention study focused on palliative care for patients managing complex cardiovascular conditions, expanding the district's footprint in holistic patient support.
Targeted Interventions and Expanding Care Options
Beyond pharmaceutical testing, local medical institutions are launching highly specific procedural and behavioral studies this month. The inclusion of Medstar Health Research Institute as a primary sponsor highlights the district's ongoing commitment to complex cardiovascular care and patient quality of life. March's new clinical trials are targeting a highly specific array of chronic and acute conditions, ensuring that patients with severe diagnoses have new avenues for care. Researchers are actively seeking individuals recently diagnosed with or currently managing:
- Advanced heart failure
- Mechanical circulatory support needs
- Left ventricular assist device management
- Palliative care requirements
- Provoked vestibulodynia
- Systemic sclerosis
The focus on mechanical circulatory support and left ventricular assist devices provides crucial research opportunities for patients with advanced heart failure who have exhausted standard therapeutic options. Simultaneously, the introduction of a Phase 3 trial for systemic sclerosis offers hope for individuals managing this rare autoimmune disease, which currently has limited disease-modifying treatments available.
District-Wide Access and Research Hubs
The District of Columbia maintains a robust and highly accessible clinical research infrastructure, with 829 active trials currently recruiting across four cities and nearly three hundred individual research sites. Geographically, new research opportunities are heavily clustered in the urban core. Washington D.C. leads the region with all three of the newly opened trials located directly within the city limits, providing a dense concentration of advanced care options for residents and commuters alike. This centralized approach allows leading research hospitals to pool resources and offer specialized care to a diverse urban population, drawing patients from across the broader metropolitan area into the district for cutting-edge treatment.
Broad Eligibility and Healthy Volunteer Opportunities
Eligibility criteria for this month's new studies are heavily skewed toward adult and senior populations, ensuring that aging demographics can participate in medical advancement. Older adults have extensive options, with all three of the new trials specifically designing their protocols to include senior participants. Pediatric research is not represented in this month's new launches, while one trial restricts participation exclusively to female patients. None of the new trials launched this month restrict participation exclusively to men, reflecting a broader trend toward inclusive trial design in adult medicine.
While none of the three new trials opened this month are accepting healthy volunteers, the broader research landscape in the district remains highly welcoming to those without specific medical diagnoses. There are currently 92 open, recruiting trials for healthy participants across the state. These trials rely heavily on healthy individuals to establish crucial baseline data for future medical breakthroughs, making community participation essential for scientific progress. Healthy volunteers in the district frequently participate in vaccine development, early-phase safety studies, and observational research that shapes the future of preventative medicine.
As the spring research season advances, the District of Columbia's clinical trial landscape is positioned to expand its focus on complex cardiovascular interventions and advanced autoimmune therapies, while the integration of targeted neurotoxin treatments in gynecological care will likely shape the future of pelvic floor dysfunction management.
Data Highlights
Conditions Closing Soon
- prostate cancer (2)
- stage iv lung cancer ajcc v8 (2)
- advanced lung non-small cell carcinoma (1)
- alzheimer disease (1)
- ambiguous lineage leukemia or lymphoma (1)
- anatomic stage ii breast cancer ajcc v8 (1)
- anxiety disorders (1)
- anatomic stage iii breast cancer ajcc v8 (1)
Most Common New Trial Conditions
- advanced heart failure (1)
- left ventricular assist device (1)
- mechanical circulatory support (1)
- palliative care (1)
- provoked vestibulodynia (1)
- systemic sclerosis (1)
Cities With the Most New Trials
- Washington D.C. (3)
Leading Sponsors
- Boehringer Ingelheim (1)
- Center for Vulvovaginal Disorders (1)
- Medstar Health Research Institute (1)
| Month | New Trials | Closing Soon |
|---|---|---|
| November 2025 | 9 | 4 |
| December 2025 | 12 | 20 |
| January 2026 | 9 | 4 |
| February 2026 | 15 | 5 |
| March 2026 | 3 | 15 |
| April 2026 | 1 | 0 |
New Studies This Month (3)
| NCT ID | Title | Phase | Enrollment | Sponsor | Condition | City |
|---|---|---|---|---|---|---|
| NCT07445932 | Palliative Care Intervention to Improve Health Related Quality of Life for Patients on Long-Term LVAD Support | NA | 90 | Medstar Health Research Institute | Advanced Heart Failure | Washington D.C. |
| NCT07486830 | IncobotulinumtoxinA for Provoked Vestibulodynia With Overactive Pelvic Floor Muscle Dysfunction | Phase 2 | 54 | Center for Vulvovaginal Disorders | Provoked Vestibulodynia | Washington D.C. |
| NCT07497087 | A Study to Test Whether Nerandomilast Helps People With Systemic Sclerosis | Phase 3 | 448 | Boehringer Ingelheim | Systemic Sclerosis | Washington D.C. |