Georgia Clinical Trials Report — March 2026
26 New Studies, 127 Closing Soon
Hipa.ai Research - March 1, 2026 - Source: Research Data
Monthly Clinical Trials Infographic
Georgians have a rapidly shrinking window to participate in several critical medical studies. Over the next ninety days, 127 clinical trials will permanently close their enrollment phases across the state. This deadline affects patients seeking novel interventions for specific diagnoses, as well as residents without underlying health issues. Currently, 23 of these soon-to-close studies are actively seeking healthy volunteers to help establish baseline medical data. Patients managing chronic and acute conditions face immediate timelines to join research programs before they stop accepting new participants. Final enrollment phases are rapidly approaching for studies focused on the following areas:
- COVID-19
- Post-traumatic stress disorder
- Major depressive disorder
- Coronary artery disease
- HER2-negative breast carcinoma
- Acute myeloid leukemia
- Colorectal cancer
- Lymphoma
High-Capacity Registries and Phase 3 Launches
Despite the wave of closures, March brings a substantial influx of large-scale research projects to the state. Major pharmaceutical sponsors and academic institutions are launching high-capacity studies that require thousands of participants. ModernaTX, Inc. is currently recruiting healthy adults for a massive Phase 3 clinical trial evaluating a pandemic influenza vaccine, offering residents a direct role in advancing global infectious disease preparedness. Gilead Sciences and Novo Nordisk A/S are also initiating major trials targeting HIV prevention and obesity management.
- HeartFlow, Inc. is launching the NAVIGATE-PCI Registry (NCT07479433), an observational study aiming to collect data from 5,000 participants regarding the management of patients before and after the deployment of their PCI Navigator.
- ModernaTX, Inc. is seeking four thousand participants for its Phase 3 pandemic influenza vaccine trial (NCT07496450) to evaluate humoral immunogenicity and safety after two doses of the mRNA-1018-H5 vaccine in adults.
- Gilead Sciences is enrolling three thousand individuals for an observational study (NCT07473778) to generate real-world information on the use of lenacapavir for HIV pre-exposure prophylaxis across diverse clinical settings.
- Boehringer Ingelheim has opened a Phase 3 trial (NCT07472517) for adults with advanced small cell lung cancer, comparing obrixtamig plus standard treatment against standard chemotherapy alone.
- Novo Nordisk A/S is recruiting four hundred participants for a Phase 3 study (NCT07481630) investigating whether weekly injections of NNC0487-0111 can help people with excess body weight and knee osteoarthritis lose weight and reduce pain.
- The University of California, San Francisco is conducting an observational study (NCT07461350) involving a cardiac MRI to understand how stimulant and polysubstance use impacts heart scarring and inflammation in people with HIV.
- Remote Cardiac Enablement is validating the clinical performance of the Infrasensor device (NCT07464704) for the early detection of high-grade obstructive acute coronary syndrome in three hundred fifty patients.
- LB Pharmaceuticals Inc. is running a Phase 2 study (NCT07494305) to assess the efficacy and safety of LB-102 compared to a placebo for patients with Bipolar 1 Disorder experiencing a major depressive episode.
Geographic Distribution Across Georgia
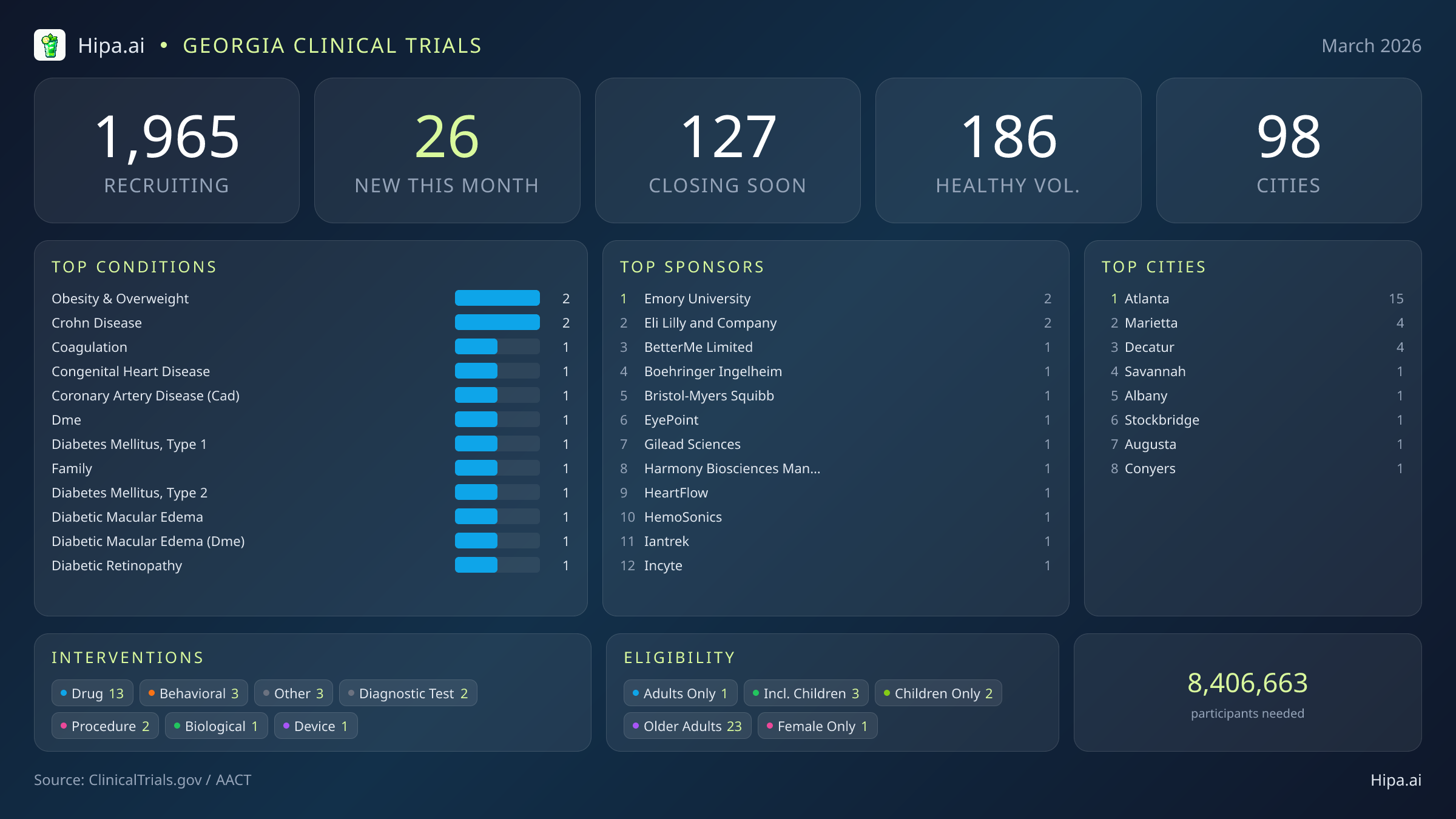
The clinical research infrastructure in Georgia remains highly accessible. There are currently 1,965 active trials recruiting across ninety-eight cities, utilizing over one thousand three hundred individual research sites. The pace of new study launches has cooled slightly since the beginning of the year, dropping from thirty-five new trials in January to 26 this month. Pharmaceutical interventions dominate the new landscape, with 13 drug trials opening their doors, followed by behavioral studies, diagnostic tests, and procedural research.
Geographically, new research opportunities are heavily clustered around the Atlanta metropolitan area. Atlanta leads the state with fifteen newly opened trials, while neighboring communities provide additional access points for residents. New trials have opened this month in the following Georgia cities:
- Atlanta
- Marietta
- Decatur
- Savannah
- Albany
- Stockbridge
- Augusta
- Conyers
Targeted Diagnoses and Healthy Volunteer Access
March's new clinical trials target a wide array of chronic and acute conditions. Researchers are actively seeking patients managing obesity, Crohn's disease, congenital heart disease, coronary artery disease, and both Type 1 and Type 2 diabetes mellitus. The sponsorship of these trials skews heavily toward commercial enterprises, with eighteen studies funded by industry partners like Eli Lilly and Company, Bristol-Myers Squibb, and Harmony Biosciences Management, Inc. Academic and institutional organizations, including Emory University, are driving the remaining protocols.
Eligibility criteria for these new studies ensure that diverse demographic groups can participate in medical advancement. Older adults have extensive options, with twenty-three of the new trials specifically designing their protocols to include senior populations. Pediatric research is also available, as three new studies are open to children and adolescents. Highly targeted demographic research includes one trial exclusively for females. For those without a specific medical diagnosis, three of this month's new trials are actively welcoming healthy volunteers. This brings the total number of open, recruiting trials for healthy participants across Georgia to 186.
As the spring research calendar progresses, the state's clinical trial ecosystem is positioned to process the extensive observational data gathered from the latest cardiovascular registries, while preparing clinical sites for the next phase of advanced metabolic and infectious disease interventions.
Data Highlights
Conditions Closing Soon
- covid-19 (3)
- post traumatic stress disorder (3)
- major depressive disorder (2)
- coronary artery disease (2)
- her2-negative breast carcinoma (2)
- lymphoma (2)
- anatomic stage iii breast cancer ajcc v8 (2)
- anatomic stage ii breast cancer ajcc v8 (2)
Most Common New Trial Conditions
- obesity & overweight (2)
- crohn disease (2)
- coagulation (1)
- congenital heart disease (1)
- coronary artery disease (cad) (1)
- dme (1)
- diabetes mellitus, type 1 (1)
- family (1)
Cities With the Most New Trials
Leading Sponsors
- Emory University (2)
- Eli Lilly and Company (2)
- BetterMe Limited (1)
- Boehringer Ingelheim (1)
- Bristol-Myers Squibb (1)
- EyePoint Pharmaceuticals, Inc. (1)
- Gilead Sciences (1)
- Harmony Biosciences Management, Inc. (1)
| Month | New Trials | Closing Soon |
|---|---|---|
| November 2025 | 31 | 5 |
| December 2025 | 65 | 34 |
| January 2026 | 29 | 16 |
| February 2026 | 34 | 7 |
| March 2026 | 26 | 37 |
| April 2026 | 3 | 0 |