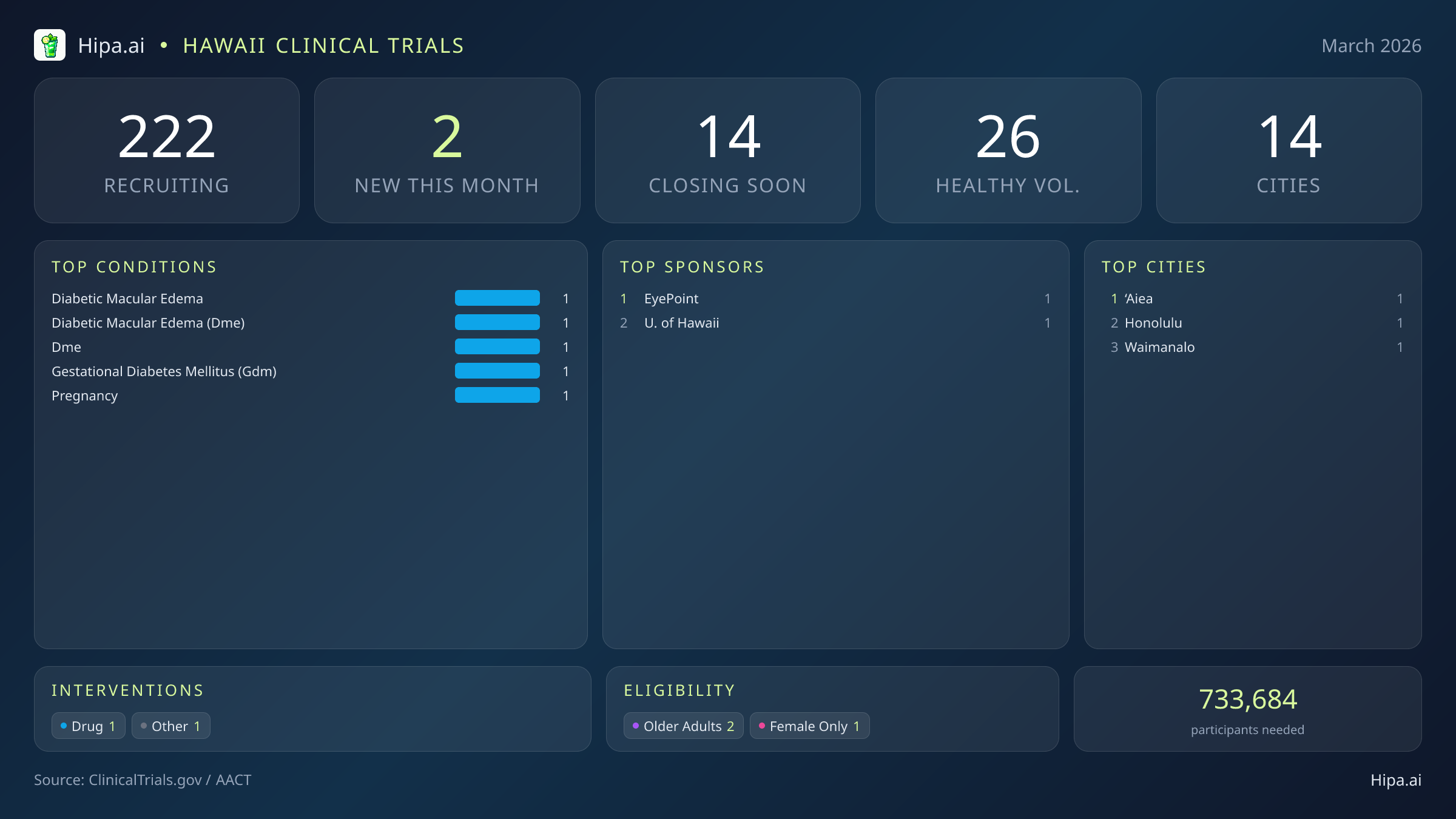
Hawaii Clinical Trials Report — March 2026
2 New Studies, 14 Closing Soon
Hipa.ai Research - March 1, 2026 - Source: Research Data
Monthly Clinical Trials Infographic
Immediate Deadlines for Hawaiian Research Initiatives
Time is strictly limited for residents across the islands to participate in several critical medical studies. Within the next ninety days, 14 clinical trials will permanently close their enrollment windows statewide. This impending cutoff represents a rapidly shrinking opportunity for patients seeking access to experimental therapies, advanced behavioral interventions, and novel medical devices. Individuals without underlying medical conditions also face a strict deadline, as 6 of these closing studies are actively seeking healthy volunteers to help establish vital baseline data for future scientific breakthroughs.
Final enrollment phases are approaching quickly for research programs focused on a diverse array of acute and chronic conditions. Patients currently managing advanced oncological diagnoses or complex psychological conditions have a narrow window remaining to explore these alternative care options. Researchers are finalizing their participant rosters for studies addressing the following conditions:
- Post-traumatic stress disorder
- Stage IV lung cancer
- Chemotherapy-induced peripheral neuropathy
- Gastric and gastroesophageal junction cancers
- HER2-positive advanced solid tumors
- Cannabis use
- Chlamydia and gonorrhea
Late-Stage Vision Research and Pharmaceutical Development
March brings a highly targeted wave of new clinical research to the state. Over the past six months, Hawaii has maintained a steady, specialized stream of new research initiatives. Following four new trials in December and three in both January and February, March introduces 2 newly activated studies. While the overall volume of new studies is highly focused this month, the caliber of the research remains exceptional, split evenly between industry-sponsored pharmaceutical development and localized academic research.
A major highlight for March is a late-stage drug trial focused on diabetic macular edema, a severe complication of diabetes characterized by fluid accumulation in the retina that can lead to significant vision loss. EyePoint Pharmaceuticals, Inc. is currently seeking participants for a major Phase 3 clinical trial that could shape the future of ocular care.
- NCT07449936 is a Phase 3 randomized, double-masked study comparing the efficacy of the experimental therapy EYP-1901 directly against Aflibercept, an established standard of care. This trial is actively recruiting 240 participants to determine if the new intervention offers improved visual outcomes and a more manageable treatment schedule for patients managing diabetic macular edema.
Because this is a Phase 3 trial, it represents one of the final stages of clinical testing before a new therapy can be submitted for broad regulatory approval. The double-masked design ensures that neither the patients nor the administering physicians know which specific treatment is being provided, guaranteeing the highest level of scientific objectivity in the results.
Maternal Health and Academic Innovation
Beyond pharmaceutical interventions, the University of Hawaii is sponsoring a new study focused entirely on maternal health and prenatal care. This academic initiative is specifically evaluating interventions and monitoring protocols for individuals managing gestational diabetes mellitus during pregnancy. Gestational diabetes requires careful management to protect both maternal health and fetal development, and this localized research aims to refine how the condition is monitored and treated within the state's healthcare system.
By spearheading this initiative, the University of Hawaii continues to cement its role as a central hub for medical advancement in the Pacific, ensuring that research priorities align directly with the specific health needs of the local population.
Research Accessibility Across the Islands
Despite the geographic complexities of an island state, Hawaii maintains a robust and highly accessible clinical research infrastructure. Statewide, 222 clinical trials are currently recruiting participants. These active studies are distributed across 14 cities and operate out of 100 distinct research sites, ensuring that advanced medical care and experimental therapies are not entirely confined to a single metropolitan center.
For the studies launched this month, research activity is clustered on the island of Oahu. Medical facilities and academic centers are actively opening their doors to new participants in the following communities:
- Honolulu
- ‘Aiea
- Waimanalo
This specific geographic distribution allows residents in both the primary urban core of Honolulu and the surrounding communities to access cutting-edge vision care and maternal health research without requiring extensive inter-island travel or lengthy commutes. The inclusion of sites in Waimanalo and ‘Aiea demonstrates a commitment to decentralizing clinical research and bringing experimental care directly into diverse neighborhoods.
Participant Eligibility and Community Involvement
The eligibility criteria for March's newly launched studies reflect a highly targeted approach to specific patient populations. Both of the new trials have designed their protocols to include older adults, ensuring that senior populations have access to advanced therapies, which is particularly vital for the vision-loss study targeting diabetic macular edema. Due to the strict focus on gestational diabetes and pregnancy, the academic trial restricts participation exclusively to female patients. Neither of the new studies activated this month includes pediatric patients or restricts enrollment exclusively to men.
While the two trials launched this month require participants to have specific underlying diagnoses, opportunities for individuals without medical conditions remain plentiful across the state. Currently, 26 active trials are recruiting healthy volunteers statewide. These participants are absolutely essential for advancing scientific discovery. Healthy volunteers provide the vital control data necessary to evaluate the safety, tolerability, and pharmacokinetics of new treatments before they are tested on vulnerable patient populations. Participating as a healthy volunteer offers a unique way for residents to actively contribute to the advancement of global medicine.
As the spring research cycle progresses, Hawaii's clinical trial landscape is positioned to expand its focus on localized academic initiatives and late-stage chronic disease management, potentially bringing more specialized maternal therapies and advanced ocular treatments directly to island residents.
Data Highlights
Conditions Closing Soon
- post traumatic stress disorder (3)
- stage iv lung cancer ajcc v8 (2)
- cannabis use (1)
- chemotherapy induced peripheral neuropathy (cipn) (1)
- chlamydia (1)
- gastric cancer (1)
- gastroesophageal junction cancer (1)
- gonorrhea (1)
Most Common New Trial Conditions
- diabetic macular edema (1)
- diabetic macular edema (dme) (1)
- dme (1)
- gestational diabetes mellitus (gdm) (1)
- pregnancy (1)
Leading Sponsors
- EyePoint Pharmaceuticals, Inc. (1)
- University of Hawaii (1)
| Month | New Trials | Closing Soon |
|---|---|---|
| October 2025 | 4 | 1 |
| November 2025 | 2 | 0 |
| December 2025 | 4 | 1 |
| January 2026 | 3 | 1 |
| February 2026 | 3 | 0 |
| March 2026 | 2 | 5 |
New Studies This Month (2)
| NCT ID | Title | Phase | Enrollment | Sponsor | Condition | City |
|---|---|---|---|---|---|---|
| NCT07446205 | Poi for Mama: a Pilot to Improve Maternal Microbiome and Gestational Diabetes Risk | NA | 50 | University of Hawaii | Gestational Diabetes Mellitus (GDM) | Honolulu |
| NCT07449936 | COMO: A Phase 3 Randomized, Double-Masked Study Comparing the Efficacy of EYP-1901 Against Aflibercept in DME | Phase 3 | 240 | EyePoint Pharmaceuticals, Inc. | Diabetic Macular Edema | ‘Aiea |