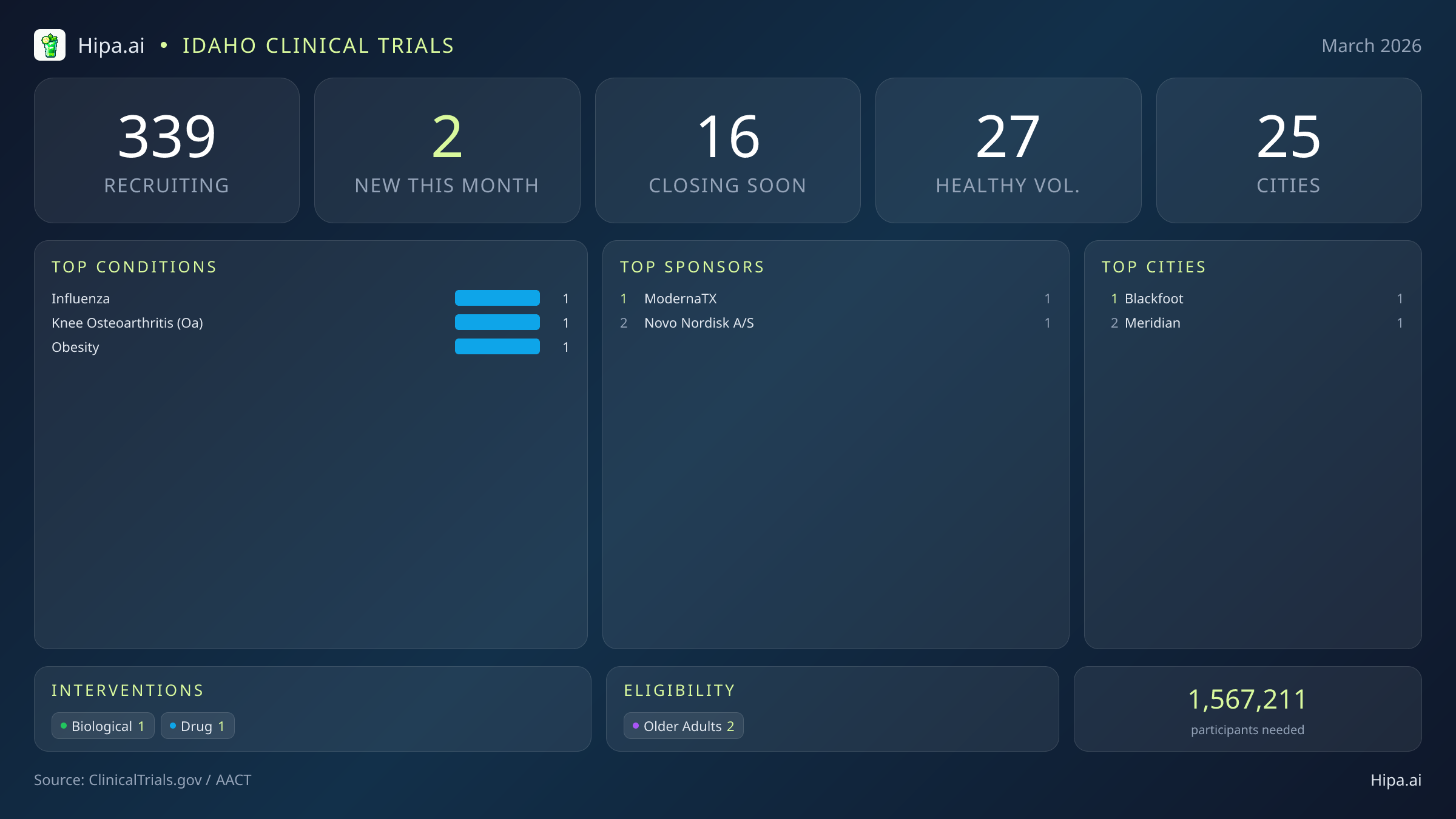
Idaho Clinical Trials Report — March 2026
2 New Studies, 16 Closing Soon
Hipa.ai Research - March 1, 2026 - Source: Research Data
Monthly Clinical Trials Infographic
Approaching Deadlines for Critical Medical Research
Time is running out for Idaho residents seeking access to several experimental therapies and medical interventions. Over the next ninety days, 16 clinical trials will permanently close their enrollment windows across the state. This impending cutoff creates a narrow window for patients managing specific diagnoses to explore alternative treatment pathways before these studies transition into their final analytical phases. Individuals without underlying health conditions also face a strict deadline, as 2 of these closing studies are actively seeking healthy volunteers to establish critical baseline data.
Final enrollment phases are rapidly approaching for studies focused on severe and chronic conditions. Patients currently navigating complex diagnoses should act quickly to evaluate their eligibility, as these trials often represent the final opportunity to access specific experimental protocols before potential regulatory review. The closing studies cover a diverse range of medical fields, from oncology to cardiovascular health. Enrollment is currently winding down for trials investigating the following conditions:
- Stage IV lung cancer
- Anatomic stage III breast cancer
- Coronary artery disease and aortic stenosis
- Chronic spontaneous urticaria
- Diabetic peripheral neuropathic pain
- Endometrial adenocarcinoma
- Asthma and COVID-19
Major Phase 3 Launches from Industry Leaders
March brings a highly targeted wave of late-stage clinical development to Idaho. While the overall volume of new research has cooled—with just 2 new trials opening this month compared to five in January—the caliber of these studies is exceptional. Both newly launched initiatives are Phase 3 trials sponsored by major pharmaceutical leaders, offering local residents access to advanced biological and drug interventions just steps away from potential market availability. Phase 3 trials are particularly notable for patients, as they involve treatments that have already passed initial safety and efficacy testing and are now being evaluated on a much larger scale.
These industry-sponsored studies are focused on pressing public health challenges, specifically pandemic preparedness and metabolic health. ModernaTX, Inc. is recruiting four thousand participants for a massive Phase 3 trial (NCT07496450) evaluating a pandemic influenza vaccine. This biological study will measure the humoral immunogenicity, or the immune system's antibody response, after administering two doses of the mRNA-1018-H5 vaccine. Researchers are also closely monitoring the overall safety and reactogenicity of the vaccine in healthy adults.
Novo Nordisk A/S has launched a Phase 3 study (NCT07481630) investigating a new weekly subcutaneous injection for individuals managing both excess body weight and knee osteoarthritis. The trial aims to determine if the experimental medicine NNC0487-0111 can effectively help patients lose weight while simultaneously reducing joint pain. Participants will be assigned to receive either the active study drug or a placebo to measure the treatment's true impact on mobility and weight management.
Research Footprint Expanding Beyond the Capital
Idaho maintains a robust clinical research infrastructure, ensuring that advanced medical care is not restricted to a single metropolitan area. Currently, 339 clinical trials are actively recruiting participants across 25 cities and 198 individual research sites statewide. This decentralized approach allows patients in rural and suburban communities to access experimental treatments without enduring extensive travel, which is often a significant barrier to clinical trial participation.
This month's newly launched trials reflect this ongoing commitment to regional accessibility. Rather than clustering exclusively in Boise, the latest research opportunities have opened their doors in distinct communities across the state. Residents in Meridian now have local access to late-stage metabolic and osteoarthritis research, bringing advanced weight management therapies directly to the community. Meanwhile, a new clinical site in Blackfoot is hosting the large-scale influenza vaccine initiative. This geographic distribution ensures that breakthrough biological and drug therapies reach a diverse cross-section of the state's population, improving the overall quality and representation of the clinical data collected.
Broad Access for Older Adults and Healthy Volunteers
Eligibility criteria for March's new studies are intentionally broad, ensuring that adult populations can seamlessly participate in scientific advancement. Both of the newly launched trials have designed their protocols to include older adults, providing senior demographics with direct access to cutting-edge vaccine technology and weight management therapies. This inclusion is vital, as older adults are often disproportionately affected by conditions like osteoarthritis and severe influenza. Neither of the new studies restricts participation based on gender, ensuring equal opportunity for all adult residents. Pediatric research remains paused this month, with zero new trials currently accepting children.
For individuals without a specific medical diagnosis, the opportunity to contribute to medical science remains incredibly strong. One of this month's new late-stage trials is actively welcoming healthy adults to test the efficacy of the pandemic influenza vaccine. Statewide, the total number of open, recruiting trials for healthy participants stands at 27. These healthy volunteer cohorts are essential for researchers to map standard immune responses and verify the safety profiles of emerging biological treatments before they are administered to vulnerable populations.
As the spring research cycle progresses, Idaho's clinical trial landscape is positioned to prioritize preventative biological interventions and dual-action metabolic therapies, signaling a broader shift toward treatments that address multiple chronic conditions simultaneously.
Data Highlights
Conditions Closing Soon
- stage iv lung cancer ajcc v8 (2)
- anatomic stage iii breast cancer ajcc v8 (1)
- coronary stenosis (1)
- asthma (1)
- chronic spontaneous urticaria (1)
- coronary artery disease (1)
- aortic stenosis (1)
- covid-19 (1)
Most Common New Trial Conditions
- influenza (1)
- knee osteoarthritis (oa) (1)
- obesity (1)
Leading Sponsors
- ModernaTX, Inc. (1)
- Novo Nordisk A/S (1)
| Month | New Trials | Closing Soon |
|---|---|---|
| November 2025 | 3 | 0 |
| December 2025 | 8 | 2 |
| January 2026 | 4 | 0 |
| February 2026 | 4 | 1 |
| March 2026 | 2 | 1 |
| April 2026 | 1 | 0 |
New Studies This Month (2)
| NCT ID | Title | Phase | Enrollment | Sponsor | Condition | City |
|---|---|---|---|---|---|---|
| NCT07481630 | A Research Study Investigating How Well the Medicine NNC0487-0111 Helps People With Excess Body Weight and Knee Osteoarthritis Lose Weight and Reduce Pain | Phase 3 | 400 | Novo Nordisk A/S | Obesity | Blackfoot |
| NCT07496450 | A Study of mRNA-1018-H5 Pandemic Influenza Vaccine in Healthy Adults | Phase 3 | 4,000 | ModernaTX, Inc. | Influenza | Meridian |