Illinois Clinical Trials Report — March 2026
40 New Studies, 200 Closing Soon
Hipa.ai Research - March 1, 2026 - Source: Research Data
Monthly Clinical Trials Infographic
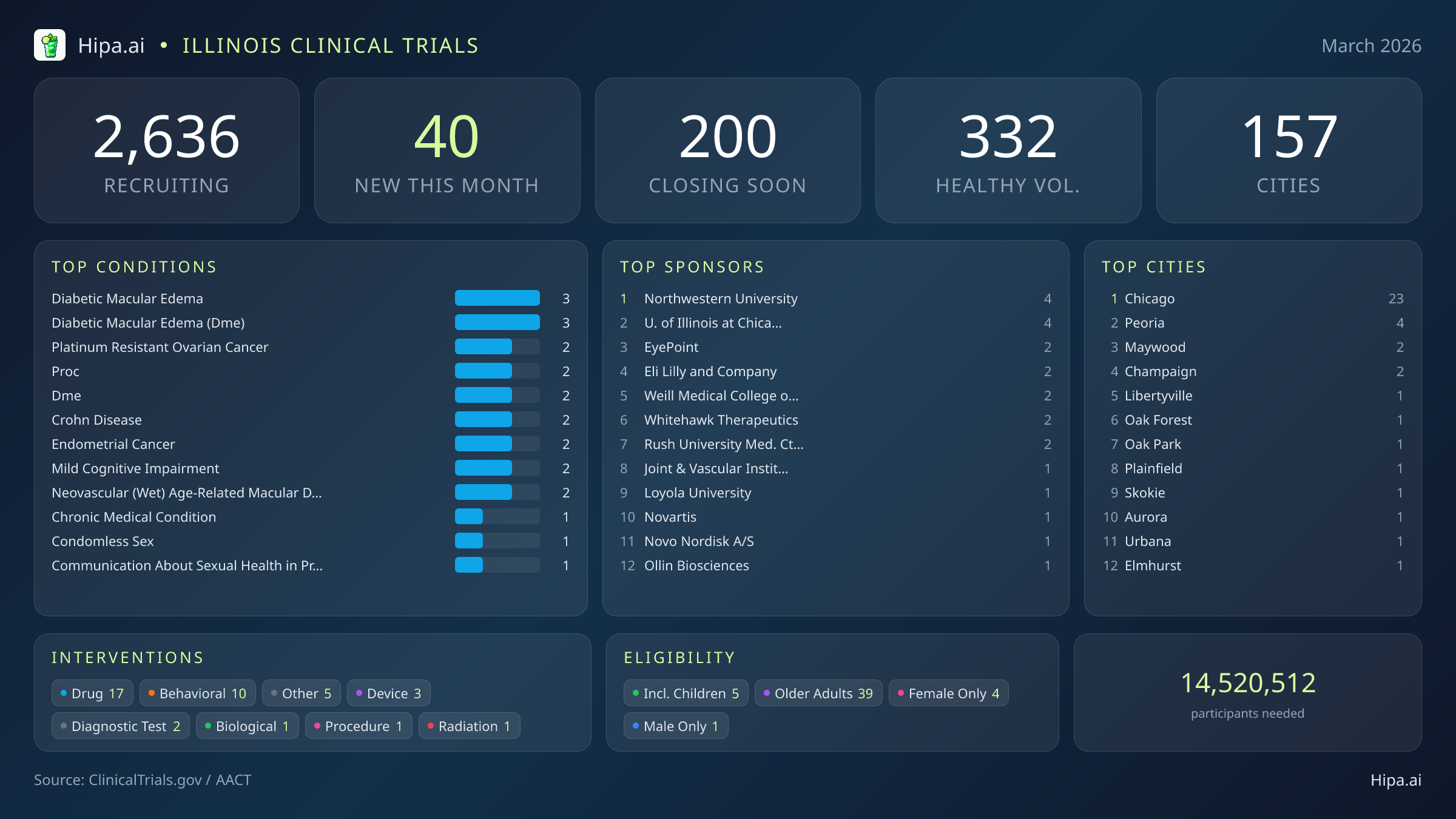
Time is running out for Illinois residents to participate in a significant wave of medical research. Within the next ninety days, 200 clinical trials will permanently close their enrollment windows across the state. For individuals managing chronic conditions or seeking alternative therapeutic options, this represents a critical deadline to access experimental treatments before these studies transition into their data analysis phases. This deadline directly impacts patients seeking novel interventions, as well as those without underlying conditions. Currently, 36 of these soon-to-close studies are actively seeking healthy volunteers. Final enrollment phases are rapidly approaching for research programs focused on several specific diagnoses:
- Mild cognitive impairment
- Major depressive disorder
- Prostate cancer
- Sickle cell disease
- Type 2 diabetes mellitus
- Gastric cancer
- Cholangiocarcinoma
Major Phase 3 and High-Capacity Research Launches
Despite these impending closures, March brings massive new research initiatives to Illinois, driven by major pharmaceutical sponsors and leading academic institutions. The University of Illinois at Chicago is launching an unprecedented health system-wide study to screen and treat opioid use disorder, seeking 271,031 participants. On the pharmaceutical front, industry leaders like Novo Nordisk and Boehringer Ingelheim are rolling out critical Phase 3 drug trials that require hundreds of participants to evaluate new therapeutic compounds.
- The University of Illinois at Chicago is initiating the C-DOSETaP System study (NCT07498322) to test a new clinical care model for opioid use disorder across a massive patient population, aiming to improve screening, engagement, and treatment planning.
- Northwestern University is building a comprehensive neuromuscular disease biorepository (NCT07477509), collecting clinical data and tissue samples from two thousand five hundred adult patients cared for at the Northwestern Memorial Hospital.
- Rush University Medical Center is evaluating protocolized weaning of high-flow nasal cannula oxygen therapy (NCT07452406) in two thousand adult patients to establish standard methods for reducing and stopping this common breathing treatment.
- Xijing Hospital is investigating a plasma exosome RNA signature (NCT07492927) to serve as a liquid biopsy for diagnosing clinically significant prostate cancer in one thousand seven hundred participants.
- Novo Nordisk A/S is recruiting for the Phase 3 AMAZE 12 study (NCT07503210) to test the safety and efficacy of weekly NNC0487-0111 injections for weight maintenance in individuals with excess body weight.
- Boehringer Ingelheim has opened a Phase 3 trial (NCT07497087) evaluating whether Nerandomilast can improve symptoms and slow disease progression in adults with limited or diffuse cutaneous systemic sclerosis.
- Weill Medical College of Cornell University is developing digital assistant technology tools (NCT07485725) tailored to help older adults manage healthcare finances, access support services, and navigate medical tasks.
- The University of Illinois at Chicago is also evaluating patient-reported outcomes of artificial intelligence-inferred lung cancer risk through the longitudinal SENTINL-1 study (NCT07458425).
Targeted Diagnoses and Healthy Volunteer Opportunities
March's new clinical trials target a wide array of chronic and acute conditions. Diabetic macular edema and neovascular age-related macular degeneration are major focal points for ophthalmological research this month. Oncology studies are also expanding, with new protocols opening for patients diagnosed with platinum-resistant ovarian cancer and endometrial cancer. Gastroenterological research is represented through new Crohn's disease trials, while neurological studies are targeting mild cognitive impairment. The sponsorship of these trials is evenly split, with nineteen studies funded by industry partners like Eli Lilly and Company and EyePoint Pharmaceuticals, Inc., and twenty-one driven by academic or other institutional organizations.
This month's influx of 40 new trials offers broad eligibility criteria, ensuring diverse populations can contribute to scientific advancement. Thirty-nine of these newly opened studies are specifically designed to include older adults, while five protocols are open to children and adolescents. Gender-specific research includes four trials exclusively for females and one exclusively for males. Healthy volunteers play an indispensable role in the early phases of drug development and the establishment of baseline metrics for diagnostic tools. With ten new opportunities opening this month alone, residents without underlying conditions have immediate avenues to contribute to scientific discovery. This expands the total number of open, recruiting trials for healthy participants across Illinois to 332.
Statewide Access and Intervention Types
The statewide clinical research infrastructure remains robust and highly accessible. Illinois currently hosts 2,636 active trials distributed across one hundred fifty-seven cities and over one thousand five hundred individual research sites. While the pace of new study launches has cooled from fifty-eight in January and fifty-six in February, the variety of interventions being tested remains diverse. Pharmaceutical interventions dominate the new landscape with 17 drug trials, followed by ten behavioral studies, three medical device evaluations, two diagnostic tests, and several biological and procedural protocols.
Geographically, new research opportunities are heavily clustered around the greater Chicago metropolitan area, which leads the state with twenty-three newly opened trials. Peoria follows with four new studies, while Maywood and Champaign each added two. However, residents living outside these primary hubs still have access to cutting-edge medical research. New trials have opened their doors this month in several other municipalities across the state:
- Libertyville
- Oak Forest
- Oak Park
- Plainfield
- Skokie
- Aurora
Moving into the spring, the Illinois clinical research ecosystem is positioned to shift focus toward analyzing the massive datasets generated by this season's high-capacity behavioral and systemic disease trials, while preparing for a new wave of targeted oncological and metabolic interventions.
Data Highlights
Conditions Closing Soon
- mild cognitive impairment (4)
- major depressive disorder (3)
- prostate cancer (3)
- depression (3)
- sickle cell disease (3)
- diabetes mellitus, type 2 (3)
- gastric cancer (2)
- depressive disorder, major (2)
Most Common New Trial Conditions
- diabetic macular edema (3)
- diabetic macular edema (dme) (3)
- platinum resistant ovarian cancer (2)
- proc (2)
- dme (2)
- crohn disease (2)
- endometrial cancer (2)
- mild cognitive impairment (2)
Cities With the Most New Trials
- Chicago (23)
- Peoria (4)
- Maywood (2)
- Champaign (2)
- Libertyville (1)
- Oak Forest (1)
- Oak Park (1)
- Plainfield (1)
Leading Sponsors
- Northwestern University (4)
- University of Illinois at Chicago (4)
- EyePoint Pharmaceuticals, Inc. (2)
- Eli Lilly and Company (2)
- Weill Medical College of Cornell University (2)
- Whitehawk Therapeutics, Inc. (2)
- Rush University Medical Center (2)
- Joint & Vascular Institute (1)
| Month | New Trials | Closing Soon |
|---|---|---|
| November 2025 | 36 | 13 |
| December 2025 | 72 | 61 |
| January 2026 | 42 | 27 |
| February 2026 | 56 | 20 |
| March 2026 | 40 | 68 |
| April 2026 | 7 | 0 |