Indiana Clinical Trials Report — March 2026
20 New Studies, 75 Closing Soon
Hipa.ai Research - March 1, 2026 - Source: Research Data
Monthly Clinical Trials Infographic
Immediate Enrollment Deadlines Across Indiana
Time is running out for Indiana residents who want to participate in several critical medical studies. Over the next ninety days, 75 clinical trials will permanently close their enrollment windows across the state. This impending deadline offers a distinct opportunity for individuals without underlying health conditions to step forward, as 13 of these soon-to-close studies are actively seeking healthy volunteers to provide essential baseline data. For patients managing specific chronic or advanced diagnoses, the window to access experimental therapies is rapidly shrinking. Final participant slots are filling up quickly for studies focused on the following conditions:
- Prostate cancer
- Alzheimer's disease and mild cognitive impairment
- Stage IV and advanced non-small cell lung cancer
- Advanced or metastatic solid tumor malignancies
- Aortic stenosis
- Anatomic stage III breast cancer
Major Pharmaceutical and Diagnostic Launches
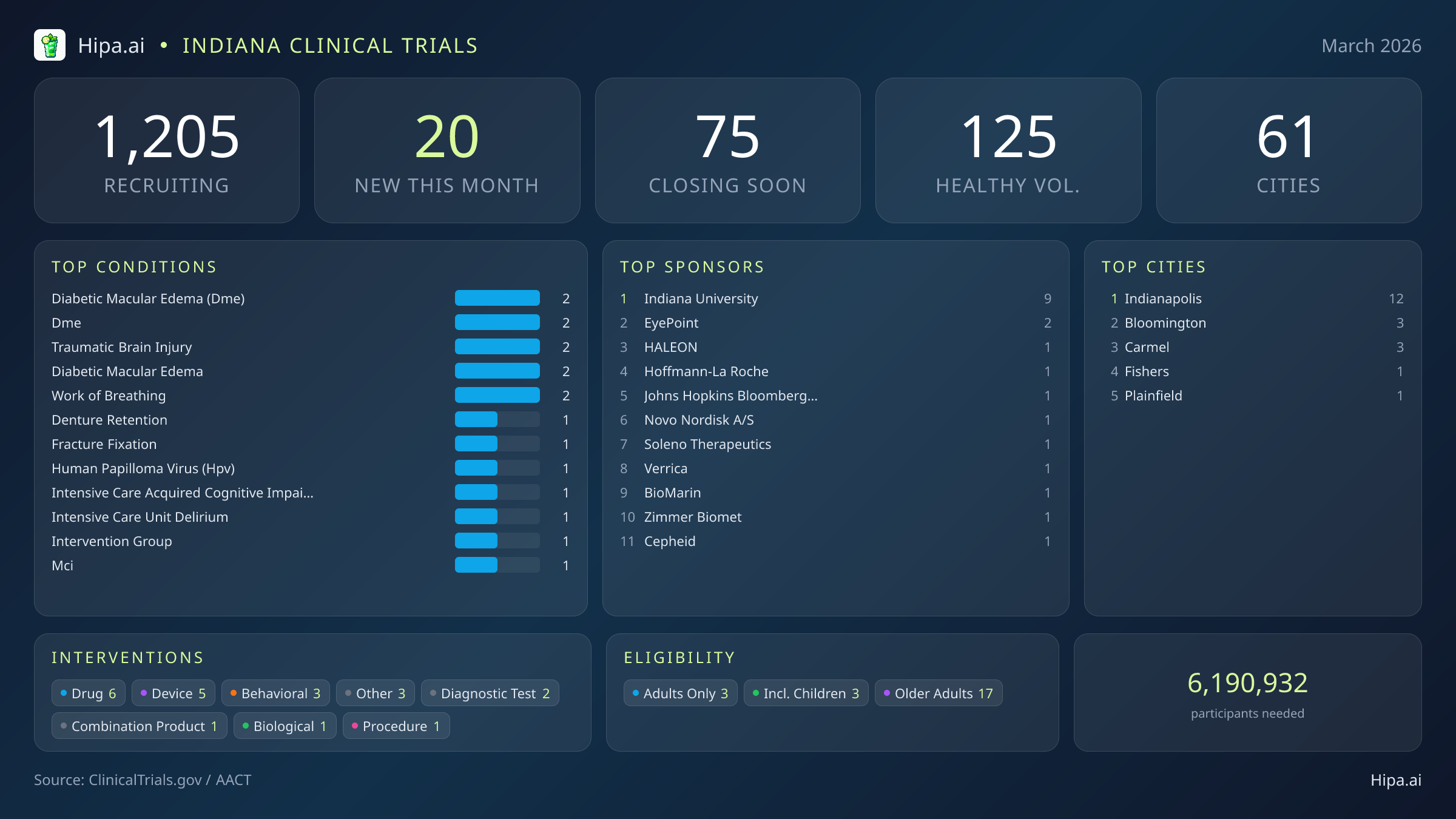
March brings a fresh wave of high-profile research to the state, evenly split between industry leaders and academic institutions. While the pace of new study launches has cooled slightly—dropping from twenty-five new trials in January to 20 this month—the caliber of the research is exceptional. Six of these new initiatives are drug interventions, while five focus on medical devices and diagnostics. Major pharmaceutical developers and research institutions are initiating large-scale Phase 3 and observational studies, offering local patients access to late-stage experimental treatments and cutting-edge testing before they reach the broader market. Several notable trials are currently seeking participants:
- Cepheid is launching a massive observational study (NCT07489846) seeking over three thousand participants to evaluate the Xpert ERP on the GeneXpert System across geographically diverse sites, aiming to gather comprehensive data from a wide variety of patients.
- Hoffmann-La Roche is recruiting two thousand adults for a diagnostic study (NCT07455136) to develop a blood-based biomarker test that aids in diagnosing traumatic brain injuries, predicting outcomes, and monitoring the development of secondary events.
- Novo Nordisk is enrolling six hundred participants in a Phase 3 trial (NCT07503210) investigating whether weekly injections of NNC0487-0111 can help individuals with excess body weight maintain their weight loss safely and effectively.
- Verrica Pharmaceuticals is conducting a Phase 3 long-term follow-up study (NCT07457918) assessing the safety of YCANTH for patients managing common warts, specifically targeting individuals who previously participated in earlier trial phases.
- Johns Hopkins Bloomberg School of Public Health has initiated a Phase 3 trial (NCT07458230) testing the efficacy of integrating next-generation sequencing for treating surgical site infections after high-energy fracture fixation, addressing a severe complication that can affect up to sixty percent of patients with major injuries.
- EyePoint Pharmaceuticals is running two parallel Phase 3 studies (NCT07449923 and NCT07449936) comparing the efficacy of EYP-1901 against aflibercept in patients suffering from diabetic macular edema.
Targeted Conditions and Behavioral Research
Beyond pharmaceutical testing, academic institutions are driving compelling behavioral and observational research this month. Indiana University is sponsoring nine of the newly opened trials, including a highly specific behavioral study (NCT07460453) recruiting nearly five hundred participants to examine racial disparities in the expression of paranoia. This research explores how suspicious thinking patterns affect daily life, relationships, and overall well-being, even in individuals without a formal mental health diagnosis.
March's new clinical trials are also targeting a highly specific array of acute and chronic conditions. Researchers are actively seeking patients recently diagnosed with or currently managing traumatic brain injury, human papillomavirus, intensive care unit delirium, and intensive care acquired cognitive impairment. Specialized dental and respiratory research is also expanding, with new studies focusing on denture retention and work of breathing.
Statewide Clinic Access and Research Hubs
Indiana's clinical research infrastructure remains robust and highly accessible, with 1,205 active trials currently recruiting across sixty-one cities and over seven hundred individual research sites. Geographically, new research opportunities are heavily clustered in the central part of the state. Indianapolis leads the state with twelve newly opened trials, serving as the primary hub for this month's medical advancements. However, residents living outside the immediate capital region still have opportunities to participate. New trials have opened their doors this month in several key municipalities:
- Indianapolis
- Bloomington
- Carmel
- Fishers
- Plainfield
Broad Eligibility and Healthy Volunteer Opportunities
Eligibility criteria for this month's new studies are notably inclusive, ensuring that diverse demographic groups can participate in medical advancement. Older adults have extensive options, with seventeen of the new trials specifically designing their protocols to include senior populations. Pediatric research is also represented, as three new studies are open to children and adolescents. None of the new trials launched this month restrict participation exclusively by sex, ensuring equal access for both male and female participants.
For those without a specific medical diagnosis, 6 of this month's new trials are actively welcoming healthy volunteers. This brings the total number of open, recruiting trials for healthy participants across Indiana to 125. These trials rely heavily on healthy individuals to establish crucial baseline data for future medical breakthroughs, particularly in the development of new diagnostic blood tests and behavioral assessments.
As the spring research season progresses, Indiana's clinical trial landscape is positioned to expand its focus on advanced diagnostic tools and metabolic therapies, paving the way for highly personalized approaches to chronic disease management and acute injury recovery.
Data Highlights
Conditions Closing Soon
- prostate cancer (3)
- alzheimer disease (3)
- healthy volunteers (2)
- stage iv lung cancer ajcc v8 (2)
- mild cognitive impairment (2)
- advanced or metastatic solid tumor malignancies (1)
- advanced lung non-small cell carcinoma (1)
- adult surgical procedures (1)
Most Common New Trial Conditions
- diabetic macular edema (dme) (2)
- dme (2)
- traumatic brain injury (2)
- diabetic macular edema (2)
- work of breathing (2)
- denture retention (1)
- fracture fixation (1)
- human papilloma virus (hpv) (1)
Cities With the Most New Trials
- Indianapolis (12)
- Bloomington (3)
- Carmel (3)
- Fishers (1)
- Plainfield (1)
Leading Sponsors
- Indiana University (9)
- EyePoint Pharmaceuticals, Inc. (2)
- HALEON (1)
- Hoffmann-La Roche (1)
- Johns Hopkins Bloomberg School of Public Health (1)
- Novo Nordisk A/S (1)
- Soleno Therapeutics, Inc. (1)
- Verrica Pharmaceuticals Inc. (1)
| Month | New Trials | Closing Soon |
|---|---|---|
| November 2025 | 18 | 2 |
| December 2025 | 32 | 20 |
| January 2026 | 22 | 7 |
| February 2026 | 25 | 6 |
| March 2026 | 20 | 25 |
| April 2026 | 1 | 0 |