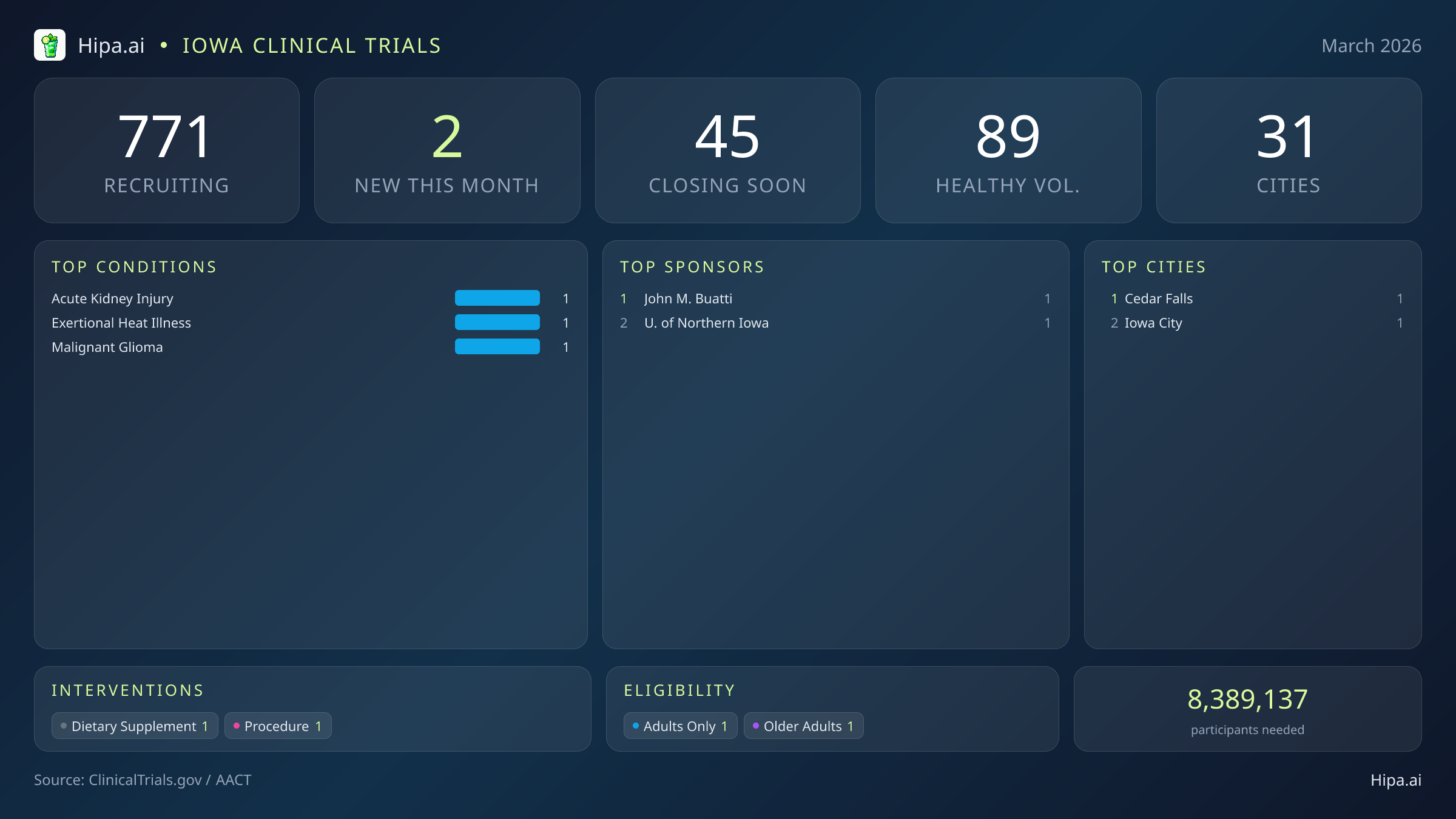
Iowa Clinical Trials Report — March 2026
2 New Studies, 45 Closing Soon
Hipa.ai Research - March 1, 2026 - Source: Research Data
Monthly Clinical Trials Infographic
Immediate Deadlines for Critical Research Initiatives
Residents across the state face a rapidly closing window to participate in dozens of medical research studies. Over the next three months, 45 clinical trials will permanently close their enrollment periods, cutting off local access to experimental therapies, behavioral interventions, and observational studies. This impending deadline is particularly critical for patients managing severe, chronic, or terminal diagnoses who may be seeking alternative interventions after exhausting standard medical treatments. Final enrollment phases are currently underway, and research coordinators are urgently seeking participants for studies focused on the following conditions:
- Dementia and related cognitive decline
- Recurrent endometrial carcinoma
- Stage IV lung cancer and advanced non-small cell lung carcinoma
- Acute asthma exacerbations
- Transitional cell carcinoma
- Anatomic Stage III breast cancer
- Chronic pain management
- Caregiver resilience and stress
- Carrier state genetics
The urgency to fill these final spots extends beyond those with active diagnoses. Among these closing studies, there is a pressing need for participants without underlying health conditions. Currently, 6 of the trials approaching their final enrollment deadlines are actively accepting healthy volunteers. These individuals are essential for providing baseline physiological metrics that allow researchers to accurately measure the efficacy and safety of new treatments against standard biological responses. Furthermore, the inclusion of behavioral studies—such as those focusing on caregiver resilience—highlights that research opportunities exist for those impacted by disease indirectly, rather than just the patients themselves.
Academic Institutions Drive March Research Launches
Following a highly active winter season, the pace of new study launches in the state has temporarily cooled. Researchers initiated just 2 new clinical trials this month, representing a noticeable dip compared to the 9 studies that opened in January. Unlike previous months, which frequently featured large-scale pharmaceutical investments, March’s additions are entirely driven by academic and independent sponsors. The University of Northern Iowa and independent researcher John M. Buatti are spearheading this month's newest scientific endeavors.
This month’s newly launched studies pivot away from traditional pharmaceutical drug testing, focusing instead on alternative therapeutic approaches. The active interventions being evaluated include a dietary supplement regimen and a specialized medical procedure. While neither of these studies falls into the category of massive Phase 2 or Phase 3 trials with hundreds of participants, they target highly specific and severe medical conditions that require localized, intensive research efforts. Patients and healthy individuals in the area can explore these new opportunities, which are dedicated to advancing treatment protocols for acute kidney injury, exertional heat illness, and malignant glioma.
The focus on exertional heat illness aligns closely with academic sports science and kinesiology programs, aiming to better understand physiological responses to extreme physical stress. Meanwhile, the investigations into acute kidney injury and malignant glioma represent highly specialized oncological and renal research. Although massive industry-sponsored trials are absent from this month's specific rollout, the localized academic focus ensures that highly specialized, niche medical challenges continue to receive rigorous scientific attention.
Inclusive Demographics and Healthy Volunteer Opportunities
The eligibility criteria for the latest research initiatives reflect a continued commitment to accessible and inclusive scientific study. Neither of the newly launched trials restricts participation based on biological sex, ensuring that both male and female residents have equal opportunity to contribute to and benefit from these medical advancements. Furthermore, the aging population remains a priority in the state's research ecosystem, with at least one of the new studies specifically designing its protocols to accommodate and monitor older adults. Pediatric research did not see any new additions this month, keeping the immediate focus entirely on adult populations.
For those without a specific medical diagnosis, the landscape remains highly active and welcoming. One of the newly opened trials is actively seeking healthy volunteers to participate alongside patient cohorts. When looking at the broader statewide picture, residents without specific medical diagnoses have extensive options to contribute to scientific progress. There are currently 89 active trials recruiting healthy volunteers across the state. These participants play an indispensable role in the research pipeline. By volunteering for dietary, behavioral, or early-phase safety studies, healthy individuals form the foundational control groups that make future medical breakthroughs possible.
Research Hubs and Statewide Accessibility
Despite the slower pace of new trial launches this month, the overall clinical research infrastructure remains expansive and highly accessible. There are currently 771 active, recruiting trials distributed across the state, ensuring that residents do not necessarily have to travel out of state to access cutting-edge medical care and experimental therapies. This robust network spans 31 different municipalities and operates out of 287 distinct clinical research sites, ranging from large university hospitals to specialized private community clinics.
Geographically, the newest research opportunities are anchored in established academic communities. This month's new trials have opened their doors in Iowa City and Cedar Falls. Iowa City continues to serve as a primary epicenter for medical advancement, leveraging its extensive university-affiliated hospital networks to host complex procedural and oncological studies. The concentration of medical expertise in this area makes it a natural home for rigorous investigations into conditions like malignant glioma. Meanwhile, the addition of Cedar Falls highlights how regional universities contribute to the broader scientific landscape, particularly in specialized fields like dietary supplementation and physiological stress responses.
For residents living outside these immediate academic hubs, the broader network of active trials ensures that opportunities remain within driving distance. The distribution of research sites across dozens of cities significantly reduces the travel burden—a common barrier to clinical trial participation. This means that rural and suburban populations can still engage with clinical studies targeting chronic disease management, behavioral health, and preventative medicine in their own local communities.
As the academic calendar transitions toward the summer research season, the state's clinical trial landscape is poised to accelerate, with anticipated expansions in late-stage pharmaceutical testing and broader observational studies that will further integrate regional clinics into the national healthcare innovation pipeline.
Data Highlights
Conditions Closing Soon
- dementia (2)
- recurrent endometrial carcinoma (2)
- stage iv lung cancer ajcc v8 (2)
- advanced lung non-small cell carcinoma (1)
- asthma acute (1)
- carcinoma, transitional cell (1)
- anatomic stage iii breast cancer ajcc v8 (1)
- caregiver resilience and stress (1)
Most Common New Trial Conditions
- acute kidney injury (1)
- exertional heat illness (1)
- malignant glioma (1)
Cities With the Most New Trials
- Cedar Falls (1)
- Iowa City (1)
Leading Sponsors
- John M. Buatti (1)
- University of Northern Iowa (1)
| Month | New Trials | Closing Soon |
|---|---|---|
| November 2025 | 9 | 1 |
| December 2025 | 23 | 10 |
| January 2026 | 5 | 3 |
| February 2026 | 9 | 2 |
| March 2026 | 2 | 9 |
| April 2026 | 2 | 0 |
New Studies This Month (2)
| NCT ID | Title | Phase | Enrollment | Sponsor | Condition | City |
|---|---|---|---|---|---|---|
| NCT07447531 | T2 Star Magnetic Resonance Imaging and Biomarker Blood Testing to Predict the Change and Progress of Malignant Gliomas | NA | 15 | John M. Buatti | Malignant Glioma | Iowa City |
| NCT07472426 | Effects of Creatine Supplementation on Cognitive Measures and Markers of Acute Kidney Injury After Exercise in the Heat | NA | 20 | University of Northern Iowa | Acute Kidney Injury | Cedar Falls |