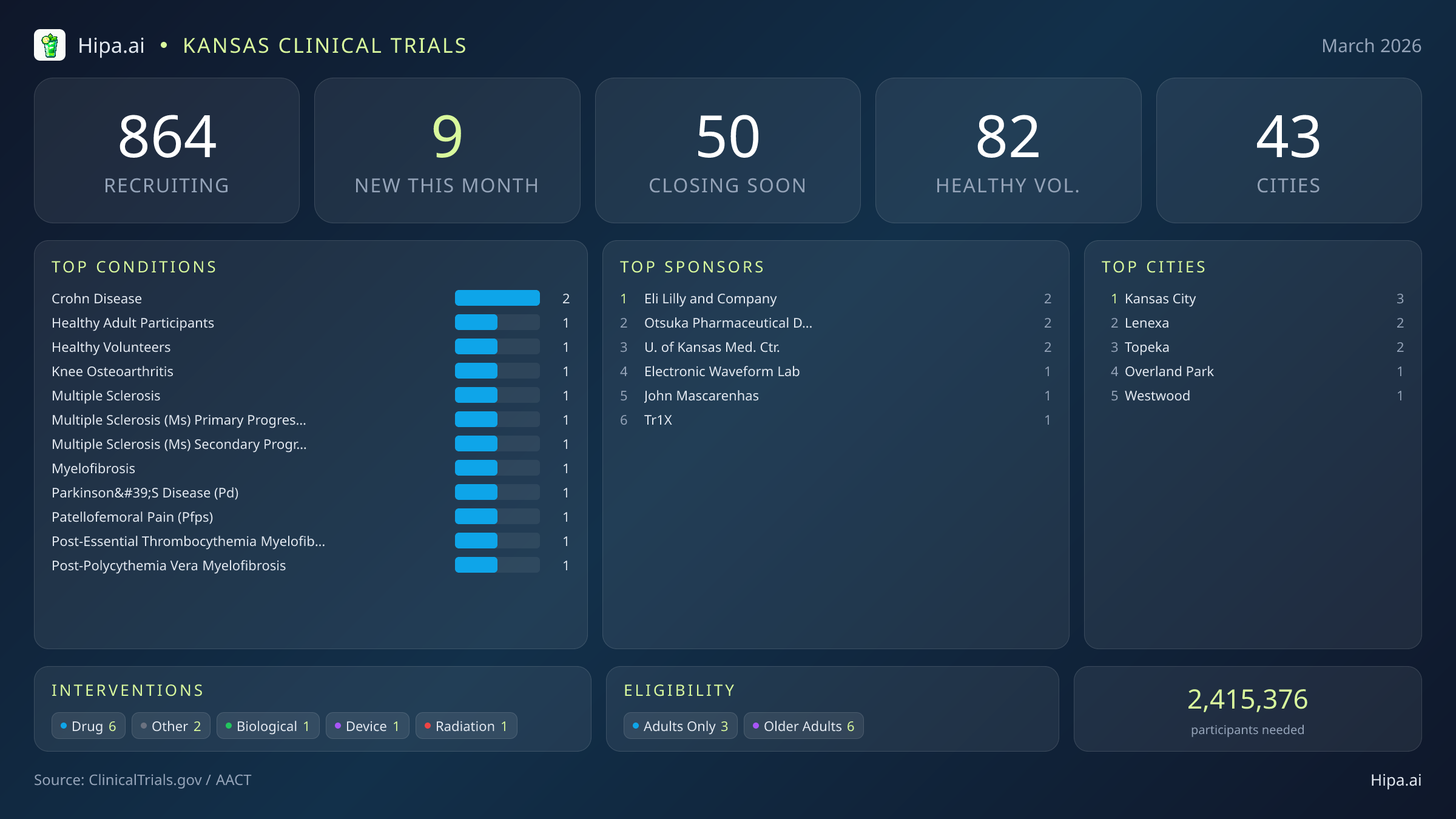
Kansas Clinical Trials Report — March 2026
9 New Studies, 50 Closing Soon
Hipa.ai Research - March 1, 2026 - Source: Research Data
Monthly Clinical Trials Infographic
Approaching Deadlines for Kansas Research Studies
Time is strictly limited for Kansas residents hoping to join several critical medical research initiatives. Over the next ninety days, 50 clinical trials will permanently close their enrollment windows across the state. When a study closes its enrollment period, the research team transitions into the data analysis phase, meaning local patients lose direct access to these specific experimental therapies. This impending deadline creates a distinct opportunity for individuals without underlying health conditions to participate in scientific discovery, as 9 of these closing studies are actively seeking healthy volunteers. For patients currently managing specific diagnoses, the window to access experimental therapies, medical devices, and behavioral interventions is rapidly narrowing. Final enrollment phases are approaching quickly for studies focused on the following conditions:
- Cystic fibrosis
- Coronary artery disease
- Stage IV lung cancer
- Amyotrophic lateral sclerosis
- Anatomic stage II breast cancer
Major Pharmaceutical and Late-Stage Clinical Launches
March brings a highly targeted wave of research to the state, with 9 new trials opening this month. While this represents a cooling period compared to the seventeen studies launched in January, industry leaders are driving significant mid-stage clinical development. Six of these newly launched studies are focused on drug interventions, while others explore biological therapies, medical devices, and radiation treatments. Six of the new trials are industry-sponsored, featuring organizations like Otsuka Pharmaceutical Development & Commercialization, Inc., Tr1X, Inc., and Electronic Waveform Lab. Several notable therapeutic trials from major sponsors are currently seeking participants:
- Eli Lilly and Company is recruiting sixty participants for a Phase 2 master protocol trial (NCT07483073) investigating multiple drugs in adults with ulcerative colitis or Crohn's disease. A master protocol allows researchers to evaluate several treatments under a single overarching trial structure, assigning participants to appropriate sub-studies prior to randomization.
- Eli Lilly and Company has also launched a related Phase 2 study (NCT07483099) testing a farnesoid X receptor agonist given together with mirikizumab compared to mirikizumab alone for adults with moderately to severely active Crohn's disease.
- Researchers are conducting a Phase 2 trial (NCT07447817) evaluating the safety and efficacy of pacritinib and selinexor in JAK inhibitor-naive patients managing myelofibrosis who are also experiencing anemia and thrombocytopenia.
Statewide Access and Research Hubs
Kansas maintains a robust and highly accessible clinical research infrastructure, with 864 active trials currently recruiting across forty-three cities and over four hundred individual research sites. Geographically, new research opportunities are heavily clustered in the eastern portion of the state, creating a dense corridor of medical innovation. The University of Kansas Medical Center is sponsoring two of the new trials, cementing its role as a central academic hub for medical advancement in the region. Residents looking to participate in this month's newest research initiatives can find open clinics in several key municipalities:
- Kansas City
- Lenexa
- Topeka
- Overland Park
- Westwood
Broad Eligibility and Healthy Volunteer Opportunities
Eligibility criteria for this month's new studies are heavily skewed toward adult and senior populations, ensuring that aging demographics can participate in medical advancement. Older adults have extensive options, with 6 of the new trials specifically designing their protocols to include senior participants. None of the new trials launched this month restrict participation exclusively to men or women, and pediatric research is not represented in the March cohort.
March's new clinical trials are also targeting a highly specific array of chronic and acute conditions, including different variations of complex neurological disorders. Researchers are actively seeking patients recently diagnosed with or currently managing:
- Primary progressive multiple sclerosis
- Secondary progressive multiple sclerosis
- Parkinson's disease
- Knee osteoarthritis
- Patellofemoral pain
For those without a specific medical diagnosis, two of this month's newly launched trials are actively welcoming healthy volunteers. This brings the total number of open, recruiting trials for healthy participants across Kansas to 82. These trials rely heavily on healthy individuals to establish crucial baseline data for future medical breakthroughs, making community participation essential for scientific progress.
As the spring research season advances, the Kansas clinical trial landscape is positioned to expand its focus on targeted autoimmune therapies and advanced neurological treatments, while the integration of master protocols will likely streamline how patients access experimental gastroenterology medicines.
Data Highlights
Conditions Closing Soon
- cystic fibrosis (2)
- coronary artery disease (2)
- physical activity (2)
- stage iv lung cancer ajcc v8 (2)
- advanced lung non-small cell carcinoma (1)
- advanced nsclc (1)
- adult surgical procedures (1)
- amyotrophic lateral sclerosis (1)
Most Common New Trial Conditions
- crohn disease (2)
- healthy adult participants (1)
- healthy volunteers (1)
- knee osteoarthritis (1)
- multiple sclerosis (1)
- multiple sclerosis (ms) primary progressive (1)
- multiple sclerosis (ms) secondary progressive (1)
- myelofibrosis (1)
Cities With the Most New Trials
- Kansas City (3)
- Lenexa (2)
- Topeka (2)
- Overland Park (1)
- Westwood (1)
Leading Sponsors
- Eli Lilly and Company (2)
- Otsuka Pharmaceutical Development & Commercialization, Inc. (2)
- University of Kansas Medical Center (2)
- Electronic Waveform Lab (1)
- John Mascarenhas (1)
- Tr1X, Inc. (1)
| Month | New Trials | Closing Soon |
|---|---|---|
| November 2025 | 14 | 5 |
| December 2025 | 23 | 17 |
| January 2026 | 9 | 6 |
| February 2026 | 17 | 5 |
| March 2026 | 9 | 16 |
| April 2026 | 1 | 0 |