Kentucky Clinical Trials Report — March 2026
10 New Studies, 66 Closing Soon
Hipa.ai Research - March 1, 2026 - Source: Research Data
Monthly Clinical Trials Infographic
Approaching Deadlines for Kentucky Research Studies
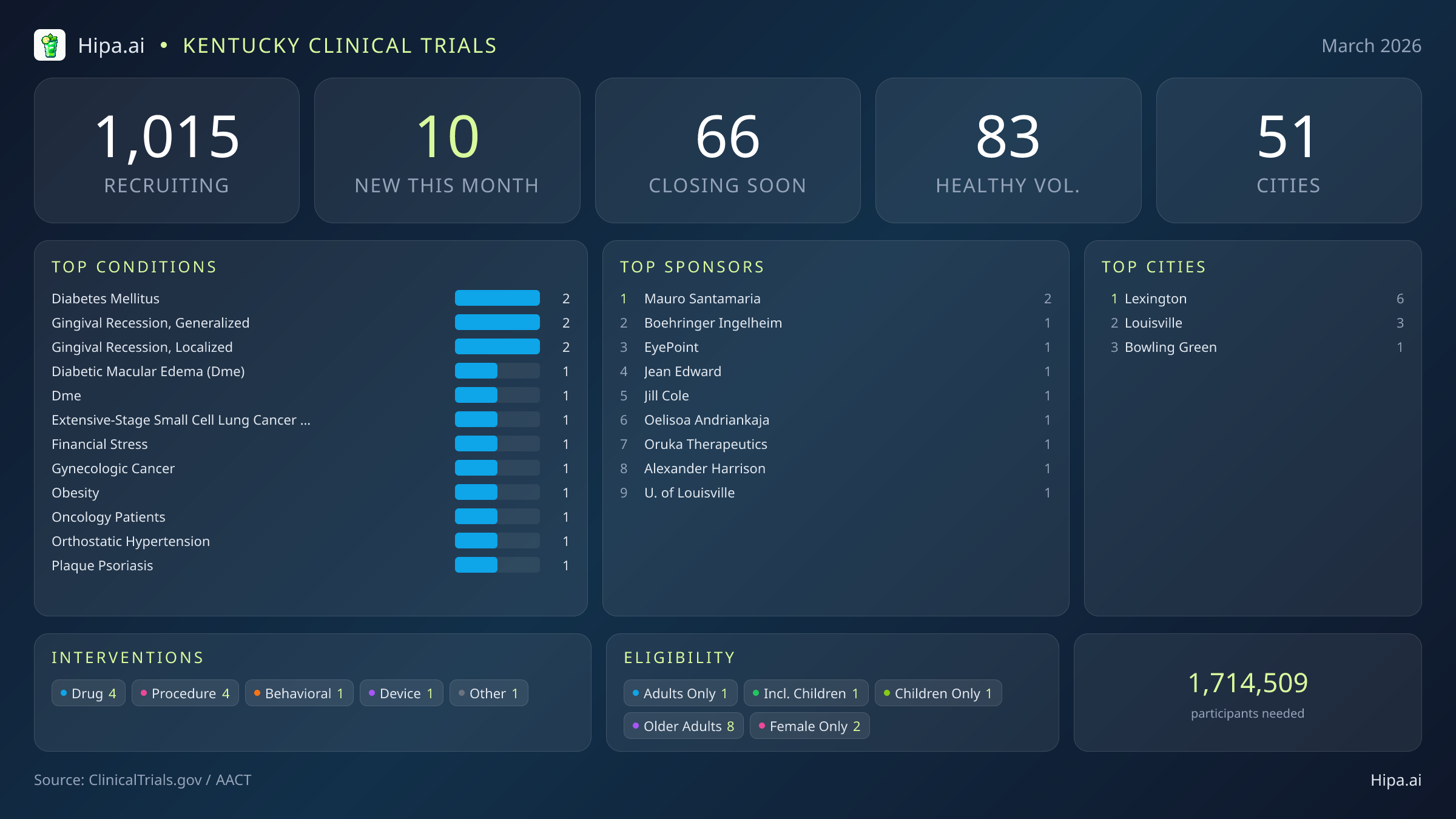
Time is running out for Kentucky residents seeking access to experimental therapies and behavioral interventions. Within the next ninety days, 66 clinical trials across the state will permanently close their enrollment windows. This shrinking timeline creates a critical window for patients managing specific diagnoses to access novel treatments before they are no longer available to the public. People without underlying medical conditions also have an immediate opportunity to contribute to scientific advancement, as 10 of these soon-to-close studies are actively seeking healthy volunteers to establish baseline clinical data. Final enrollment phases are approaching rapidly for studies focused on several chronic and severe conditions, meaning prospective participants must act quickly to complete screening protocols:
- Type 2 diabetes mellitus
- Post-traumatic stress disorder
- Ovarian and endometrial cancer
- Multiple myeloma
- Stage IV lung cancer and anatomic stage II breast cancer
- Chronic spontaneous urticaria and atopic dermatitis
Late-Stage Pharmaceutical Innovations
March brings a highly targeted wave of new medical research to the state. While the overall pace of new study launches has slowed compared to the beginning of the year—dropping from twenty-one new trials in January to 10 this month—the caliber of the incoming research is substantial. Industry leaders are initiating major Phase 2 and Phase 3 studies, giving local patients access to late-stage experimental treatments before broader market approval. Four of the newly launched trials focus on drug interventions, one involves a medical device, and another four test novel medical procedures. The remaining trial focuses on behavioral modifications. Several notable therapeutic trials are currently seeking participants:
- Boehringer Ingelheim is recruiting for a massive Phase 3 trial (NCT07472517) enrolling six hundred seventy participants. The study evaluates whether combining the experimental medicine obrixtamig with standard treatments like atezolizumab, carboplatin, and etoposide improves survival rates for adults with extensive-stage small cell lung cancer.
- EyePoint Pharmaceuticals, Inc. has launched a Phase 3 study (NCT07449923) seeking two hundred forty participants to compare the efficacy of EYP-1901 against aflibercept for patients suffering from diabetic macular edema.
- Oruka Therapeutics, Inc. is conducting an open-label extension Phase 2 trial (NCT07449702) to evaluate the long-term safety and efficacy of ORKA-001 in adult participants managing moderate-to-severe plaque psoriasis who have previously participated in related studies.
Targeted Conditions and Academic Initiatives
Beyond the major pharmaceutical drug trials, academic researchers and independent sponsors are driving the majority of this month's new clinical initiatives. Seven of the newly opened trials are sponsored by non-industry entities and principal investigators such as Mauro Santamaria, Jean Edward, Jill Cole, Oelisoa Andriankaja, and Alexander Harrison. These studies explore a diverse array of physical and behavioral health challenges.
Researchers are actively seeking patients recently diagnosed with or currently managing specific conditions. Dental and oral health represent a surprising focus this month, with four separate trials opening to treat generalized and localized gingival recession. Other targeted conditions include obesity, oncology patients, and gynecologic cancer. One unique behavioral study launched this month is specifically designed to examine the impacts of financial stress on health outcomes, broadening the scope of clinical research beyond traditional medical interventions.
Research Hubs and Statewide Accessibility
The pace of new trial initiation in Kentucky has seen significant fluctuation over the past six months. Following a peak of twenty-six new trials in December and twenty in February, March's addition of ten new studies represents a stabilization in the research pipeline. Despite this month-to-month variance, Kentucky maintains a robust and highly accessible clinical research infrastructure, with 1,015 active trials currently recruiting across fifty-one cities and five hundred seventy-six individual research sites. This extensive network ensures that residents across the commonwealth have access to cutting-edge medical research without necessarily having to cross state lines.
Geographically, this month's new research opportunities are heavily clustered in the state's largest metropolitan areas. Lexington leads the state with six newly opened trials, serving as the primary hub for March's clinical research expansion. Louisville follows with three new studies, while Bowling Green provides an additional access point in the southern region of the state with one newly launched trial. This distribution pattern highlights a concentration of resources in major medical centers, though the broader statewide network continues to support ongoing trials in dozens of other communities.
Inclusive Eligibility and Healthy Volunteer Demand
Eligibility criteria for this month's new studies reflect a strong demand for older adult participants. Eight of the newly launched trials have designed their protocols specifically to include senior populations, ensuring that experimental treatments are tested on the demographics most likely to utilize them. Pediatric research is also represented, with one new study open to children. The gender requirements for this month's trials lean slightly toward female participants, with two studies restricting enrollment exclusively to women, such as those targeting gynecologic cancer, while no new trials are restricted solely to men.
For individuals without a specific medical diagnosis, 3 of this month's new trials are actively welcoming healthy volunteers. This brings the total number of open, recruiting trials for healthy participants across Kentucky to 83. These healthy volunteer cohorts are essential for establishing baseline data, allowing researchers to accurately measure the safety and efficacy of new interventions against standard human biological responses.
As the spring research season accelerates, Kentucky's clinical trial landscape is positioned to expand its focus on advanced oncology treatments and metabolic therapies, potentially offering residents even greater access to next-generation medical breakthroughs in the coming months.
Data Highlights
Conditions Closing Soon
- diabetes mellitus, type 2 (3)
- post traumatic stress disorder (3)
- ovarian cancer (2)
- multiple myeloma (2)
- endometrial cancer (2)
- stage iv lung cancer ajcc v8 (2)
- chronic spontaneous urticaria (2)
- atopic dermatitis (1)
Most Common New Trial Conditions
- diabetes mellitus (2)
- gingival recession, generalized (2)
- gingival recession, localized (2)
- diabetic macular edema (dme) (1)
- dme (1)
- extensive-stage small cell lung cancer (es-sclc) (1)
- financial stress (1)
- gynecologic cancer (1)
Cities With the Most New Trials
- Lexington (6)
- Louisville (3)
- Bowling Green (1)
Leading Sponsors
- Mauro Santamaria (2)
- Boehringer Ingelheim (1)
- EyePoint Pharmaceuticals, Inc. (1)
- Jean Edward (1)
- Jill Cole (1)
- Oelisoa Andriankaja (1)
- Oruka Therapeutics, Inc. (1)
- Alexander Harrison (1)
| Month | New Trials | Closing Soon |
|---|---|---|
| November 2025 | 8 | 1 |
| December 2025 | 26 | 9 |
| January 2026 | 13 | 6 |
| February 2026 | 20 | 1 |
| March 2026 | 10 | 24 |
| April 2026 | 3 | 0 |