Louisiana Clinical Trials Report — March 2026
7 New Studies, 43 Closing Soon
Hipa.ai Research - March 1, 2026 - Source: Research Data
Monthly Clinical Trials Infographic
Immediate Deadlines and Expiring Opportunities
Time is rapidly closing for Louisiana residents seeking to participate in several critical medical research programs. Within the next ninety days, 43 clinical trials across the state will permanently close their enrollment windows. This impending deadline creates a narrow window for patients seeking experimental therapies, as well as a unique opportunity for individuals without underlying medical conditions. Currently, 10 of these soon-to-close studies are actively seeking healthy volunteers to establish vital baseline data. Patients managing specific diagnoses must act quickly to access final enrollment phases for studies focused on complex and chronic conditions. Final enrollment phases are approaching quickly for studies focused on the following conditions:
- Major depressive disorder
- Colon cancer
- Interstitial lung disease
- Multiple myeloma
- COVID-19
- Acute limb ischemia
- Hepatic ascites
High-Profile Pharmaceutical Interventions
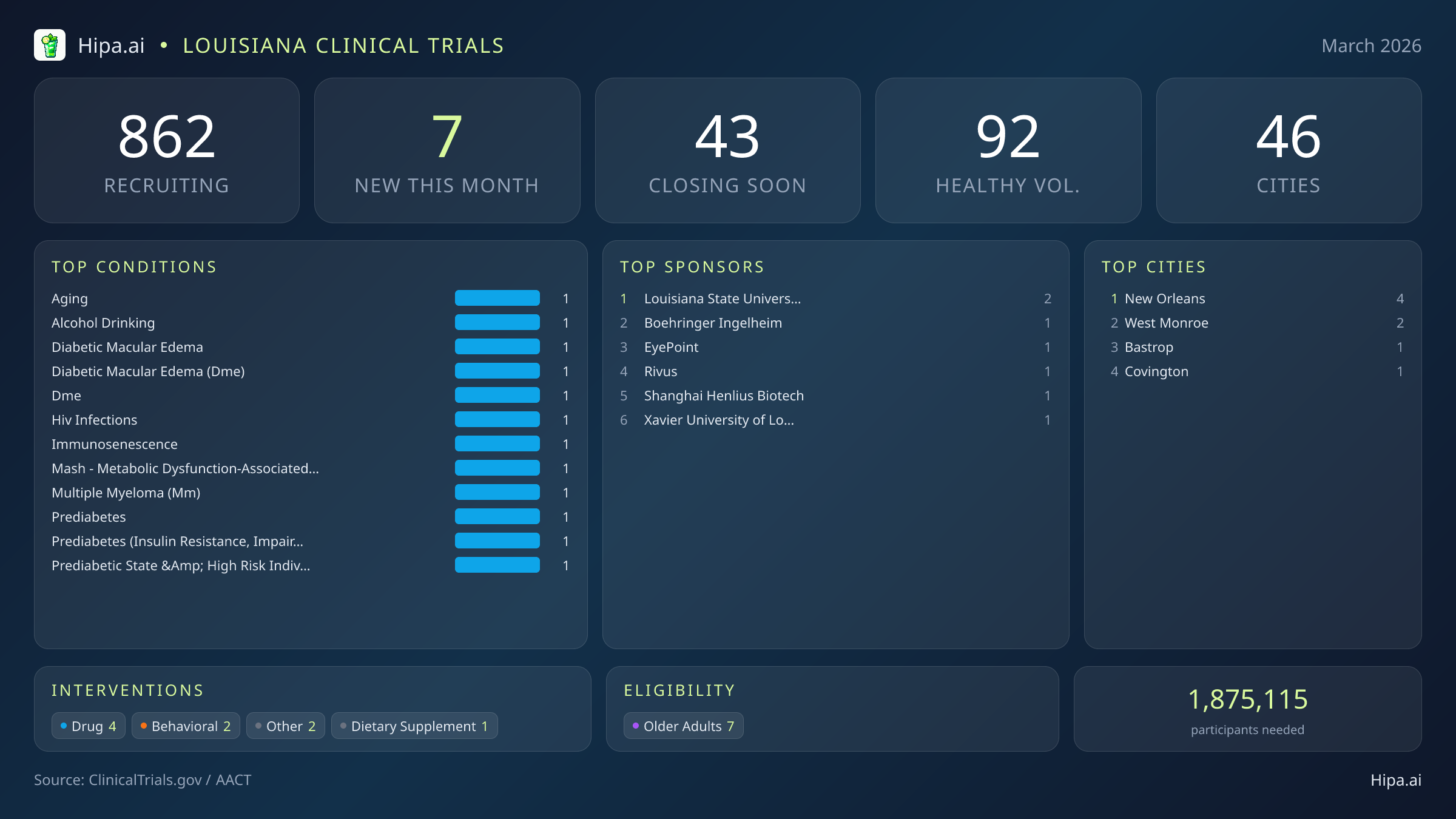
Despite these impending closures, March brings a fresh wave of high-profile research to the state, heavily driven by pharmaceutical innovation. Louisiana researchers opened 7 new trials this month, maintaining a steady pace compared to the eight studies launched in January. The majority of this month's new research focuses on drug interventions, with major industry sponsors initiating large-scale Phase 2 and Phase 3 studies. These late-stage trials offer local patients access to experimental treatments before they reach the broader market. Several notable therapeutic trials are currently seeking participants:
- Boehringer Ingelheim has launched a Phase 3 study (NCT07497087) testing nerandomilast in adults with systemic sclerosis. This trial aims to determine if the experimental drug can improve symptoms and slow disease progression for patients with limited or diffuse cutaneous variants of the disease. The sponsor is seeking over four hundred participants globally.
- EyePoint Pharmaceuticals is conducting a Phase 3 trial (NCT07449936) comparing the efficacy of EYP-1901 against aflibercept. This double-masked study targets patients managing diabetic macular edema, a leading cause of vision loss, and aims to enroll two hundred forty participants.
- Rivus Pharmaceuticals is enrolling one hundred eighty participants in a Phase 2 trial (NCT07491458) to assess the safety, tolerability, and pharmacokinetics of HU6. This research targets patients diagnosed with metabolic dysfunction-associated steatohepatitis and will closely monitor changes in liver fat content.
- Shanghai Henlius Biotech is recruiting for a Phase 1 study (NCT07477587) comparing the pharmacokinetic similarity, safety, and immunogenicity of HLX15-SC with DARZALEX FASPRO. This trial is specifically designed for transplant-ineligible patients who have been newly diagnosed with multiple myeloma.
Academic Initiatives and Behavioral Research
Beyond pharmaceutical testing, Louisiana's academic institutions are launching targeted behavioral and observational studies this month. Xavier University of Louisiana is spearheading a unique pilot study (NCT07451314) focused entirely on the local workforce. This one-year project will assess prediabetes rates among hospitality workers in New Orleans. Researchers will evaluate the impact of pharmacist-led lifestyle education sessions, the provision of wellness tools, and access to healthcare resources to prevent the onset of full diabetes in this highly active workforce. The Louisiana State University Health Sciences Center in New Orleans is also expanding its research footprint, sponsoring two new trials this month to address regional health disparities.
March's new clinical trials are targeting a highly specific array of chronic conditions and health behaviors. Researchers are actively seeking patients recently diagnosed with or currently managing:
- HIV infections
- Alcohol drinking behaviors
- Immunosenescence and aging
- Prediabetes
- Diabetic macular edema
- Metabolic dysfunction-associated steatohepatitis
Geographic Distribution Across the Pelican State
Louisiana's clinical research infrastructure remains robust and highly accessible, with 862 active trials currently recruiting across the state. These studies are distributed among forty-six cities and operate out of 648 individual research sites, ensuring that residents across various parishes have access to cutting-edge medical research.
Geographically, new research opportunities launched this month are clustered in both major metropolitan hubs and smaller regional centers. New Orleans leads the state with four newly opened trials, serving as the primary anchor for academic and industry research in the southern portion of the state. However, residents living outside the immediate Gulf Coast region still have distinct opportunities to participate. The northern and eastern parishes are seeing fresh research investments, bridging the gap in rural and suburban healthcare access. New trials have opened their doors this month in the following municipalities:
- New Orleans
- West Monroe
- Bastrop
- Covington
Broad Eligibility and Healthy Volunteer Access
Eligibility criteria for this month's new studies are notably inclusive for adult populations, ensuring that diverse demographic groups can participate in medical advancement. Older adults have extensive options, with all seven of the new trials specifically designing their protocols to include senior populations. This is particularly relevant for studies targeting immunosenescence, aging, and metabolic disorders. None of the new trials launched this month restrict participation exclusively by sex, ensuring equal access for both male and female participants, though pediatric populations are excluded from this specific batch of new studies.
For those without a specific medical diagnosis, one of this month's new trials is actively welcoming healthy volunteers. This addition brings the total number of open, recruiting trials for healthy participants across Louisiana to 92. These trials rely heavily on healthy individuals to establish crucial baseline data for future medical breakthroughs, offering a direct way for the general public to contribute to scientific progress without needing a prior diagnosis.
As the spring research season progresses, Louisiana's clinical trial landscape is positioned to expand its focus on metabolic therapies and targeted oncology treatments, while community-based behavioral studies will likely shape new preventive healthcare strategies for the state's essential hospitality workforce.
Data Highlights
Conditions Closing Soon
- major depressive disorder (2)
- colon cancer (2)
- interstitial lung disease (2)
- multiple myeloma (2)
- covid-19 (2)
- behavior and behavior mechanisms (1)
- ascites hepatic (1)
- acute limb ischemia (1)
Most Common New Trial Conditions
- aging (1)
- alcohol drinking (1)
- diabetic macular edema (1)
- diabetic macular edema (dme) (1)
- dme (1)
- hiv infections (1)
- immunosenescence (1)
- mash - metabolic dysfunction-associated steatohepatitis (1)
Cities With the Most New Trials
- New Orleans (4)
- West Monroe (2)
- Bastrop (1)
- Covington (1)
Leading Sponsors
- Louisiana State University Health Sciences Center in New Orleans (2)
- Boehringer Ingelheim (1)
- EyePoint Pharmaceuticals, Inc. (1)
- Rivus Pharmaceuticals, Inc. (1)
- Shanghai Henlius Biotech (1)
- Xavier University of Louisiana. (1)
| Month | New Trials | Closing Soon |
|---|---|---|
| November 2025 | 8 | 4 |
| December 2025 | 24 | 22 |
| January 2026 | 5 | 5 |
| February 2026 | 8 | 2 |
| March 2026 | 7 | 17 |
| April 2026 | 1 | 0 |