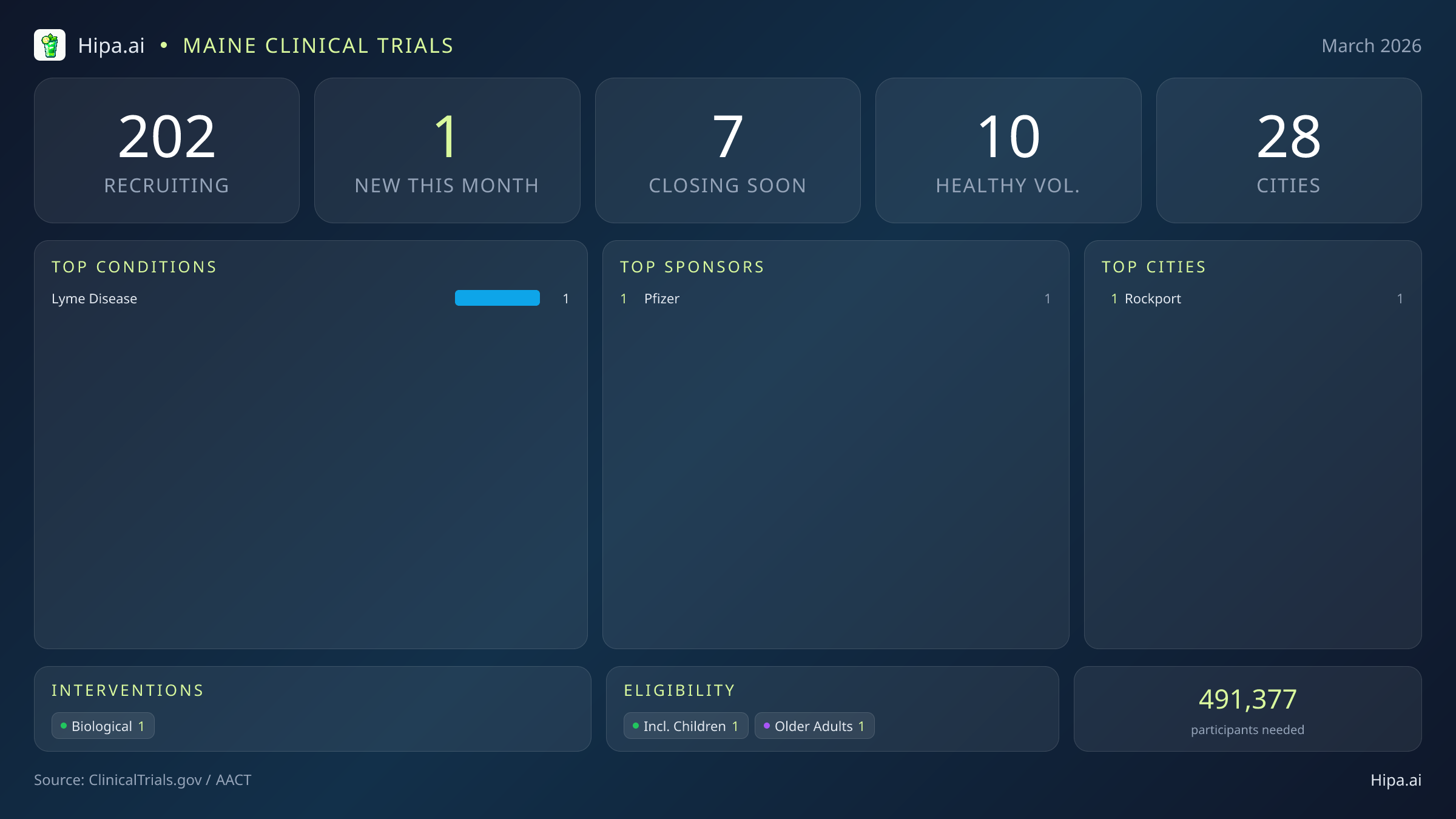
Maine Clinical Trials Report — March 2026
1 New Studies, 7 Closing Soon
Hipa.ai Research - March 1, 2026 - Source: Research Data
Monthly Clinical Trials Infographic
Urgent Deadlines for Advanced Oncology and Gastroenterology Studies
Time is strictly limited for patients in Maine seeking access to several critical medical research initiatives. Over the next ninety days, 7 clinical trials will permanently close their enrollment windows statewide. This impending deadline creates a narrow window of opportunity for individuals actively managing specific, severe diagnoses to access experimental therapies before the research moves into the closed data analysis phase. None of these closing studies are accepting healthy volunteers, meaning these final enrollment spots are exclusively reserved for patients requiring targeted medical interventions.
The urgency is particularly acute for patients navigating advanced oncological diagnoses and chronic gastrointestinal conditions. Final enrollment phases are approaching quickly for studies focused on the following specific conditions:
- Stage IV lung cancer (AJCC v8)
- Breast cancer
- Ulcerative colitis
- Endometrial endometrioid and mucinous adenocarcinoma
- Endometrial clear cell and mixed cell adenocarcinoma
- Endometrial dedifferentiated and undifferentiated carcinoma
For patients diagnosed with these highly specific cellular subtypes of endometrial cancer or advanced lung cancer, these closing trials may represent the final local opportunity to participate in these exact therapeutic protocols. Patients currently managing these conditions should act immediately to determine their eligibility before the ninety-day window expires.
Late-Stage Biological Development Arrives on the Coast
March brings a revitalized wave of high-profile biological research to the state. Following a quiet start to the year with zero new studies launched in January, this month introduces 1 major new trial driven by a leading industry sponsor. Pharmaceutical companies continue to drive significant late-stage clinical development in the region, offering local residents access to experimental biological interventions before they reach the broader commercial market.
This month's standout research initiative is sponsored by Pfizer, focusing on a condition with deep regional relevance to the Northeast: Lyme disease. Pfizer is currently recruiting for a massive Phase 3 trial (NCT07500506) titled "A Study to Learn About the Safety, Tolerability, and Immunogenicity of a Fifth Dose of 6-Valent OspA-Based Lyme Disease Vaccine." This late-stage study aims to enroll 1,712 participants globally. The primary purpose of this research is to rigorously evaluate the safety profile of the VLA15 Lyme disease vaccine and assess its ability to generate a protective immune response after participants receive a fifth dose of the vaccine.
Phase 3 trials represent the final stage of clinical testing before a sponsor can seek regulatory approval from the FDA. By participating in this study, local residents have the opportunity to contribute to the final safety evaluations of a vaccine designed to prevent a highly prevalent tick-borne illness.
Broad Eligibility and Rare Healthy Volunteer Opportunities
Because this new Pfizer trial focuses on a prophylactic vaccine rather than a disease treatment, it presents a distinct opportunity for individuals without underlying health conditions to participate in scientific discovery. This biological study is actively seeking healthy volunteers, bringing the total number of open, recruiting trials for healthy participants across Maine to 10. These trials rely heavily on healthy individuals to establish crucial baseline safety and immunogenicity data, making community participation essential for the advancement of preventive medicine.
The eligibility criteria for this newly launched study are exceptionally broad, ensuring that multiple generations can contribute to this medical advancement. The protocol is specifically designed to include pediatric populations, welcoming children as young as seven years old. Simultaneously, the trial includes older adults, ensuring the vaccine's safety profile is evaluated across the aging demographic. The study does not restrict participation based on gender, remaining fully open to both male and female volunteers who are generally healthy and have already met the preliminary vaccination requirements outlined by the study investigators.
Statewide Access and Coastal Research Hubs
Maine's clinical research infrastructure remains highly accessible and geographically diverse, with 202 active trials currently recruiting across the state. These ongoing studies are distributed across 28 different cities and operate out of 109 individual research sites. This extensive network ensures that advanced medical care, experimental therapies, and preventive vaccine trials are not strictly confined to the state's largest metropolitan centers, allowing rural and coastal residents to participate in clinical research without facing prohibitive travel barriers.
Geographically, this month's newest research opportunity has opened its doors in Rockport. By placing this late-stage vaccine trial in a coastal community, researchers are directly reaching populations that spend significant time outdoors and may benefit most from advancements in tick-borne illness prevention.
Looking at the broader timeline, Maine's clinical trial landscape has maintained a steady, targeted approach to new study launches. Over the past six months, the state has seen a fluctuating stream of new research opportunities. Following three new studies in October and a peak of six new trials in November of last year, the state has maintained a highly focused rollout, adding one new trial in December, one in January, and now one in March.
As the spring research season accelerates, Maine's clinical trial ecosystem is positioned to continue its vital work in infectious disease prevention, while the impending closure of several advanced oncology studies underscores the critical need for rapid patient enrollment in targeted cancer therapies.
Data Highlights
Conditions Closing Soon
- stage iv lung cancer ajcc v8 (2)
- breast cancer (1)
- colitis, ulcerative (1)
- endometrial adenocarcinoma (1)
- endometrial clear cell adenocarcinoma (1)
- endometrial dedifferentiated carcinoma (1)
- endometrial endometrioid adenocarcinoma (1)
- endometrial mixed cell adenocarcinoma (1)
Most Common New Trial Conditions
- lyme disease (1)
Cities With the Most New Trials
- Rockport (1)
Leading Sponsors
- Pfizer (1)
| Month | New Trials | Closing Soon |
|---|---|---|
| October 2025 | 3 | 0 |
| November 2025 | 6 | 0 |
| December 2025 | 1 | 3 |
| January 2026 | 1 | 3 |
| March 2026 | 1 | 1 |
New Studies This Month (1)
| NCT ID | Title | Phase | Enrollment | Sponsor | Condition | City |
|---|---|---|---|---|---|---|
| NCT07500506 | A Study to Learn About the Safety, Tolerability, and Immunogenicity of a Fifth Dose of 6-Valent OspA-Based Lyme Disease Vaccine | Phase 3 | 1,712 | Pfizer | Lyme Disease | Rockport |