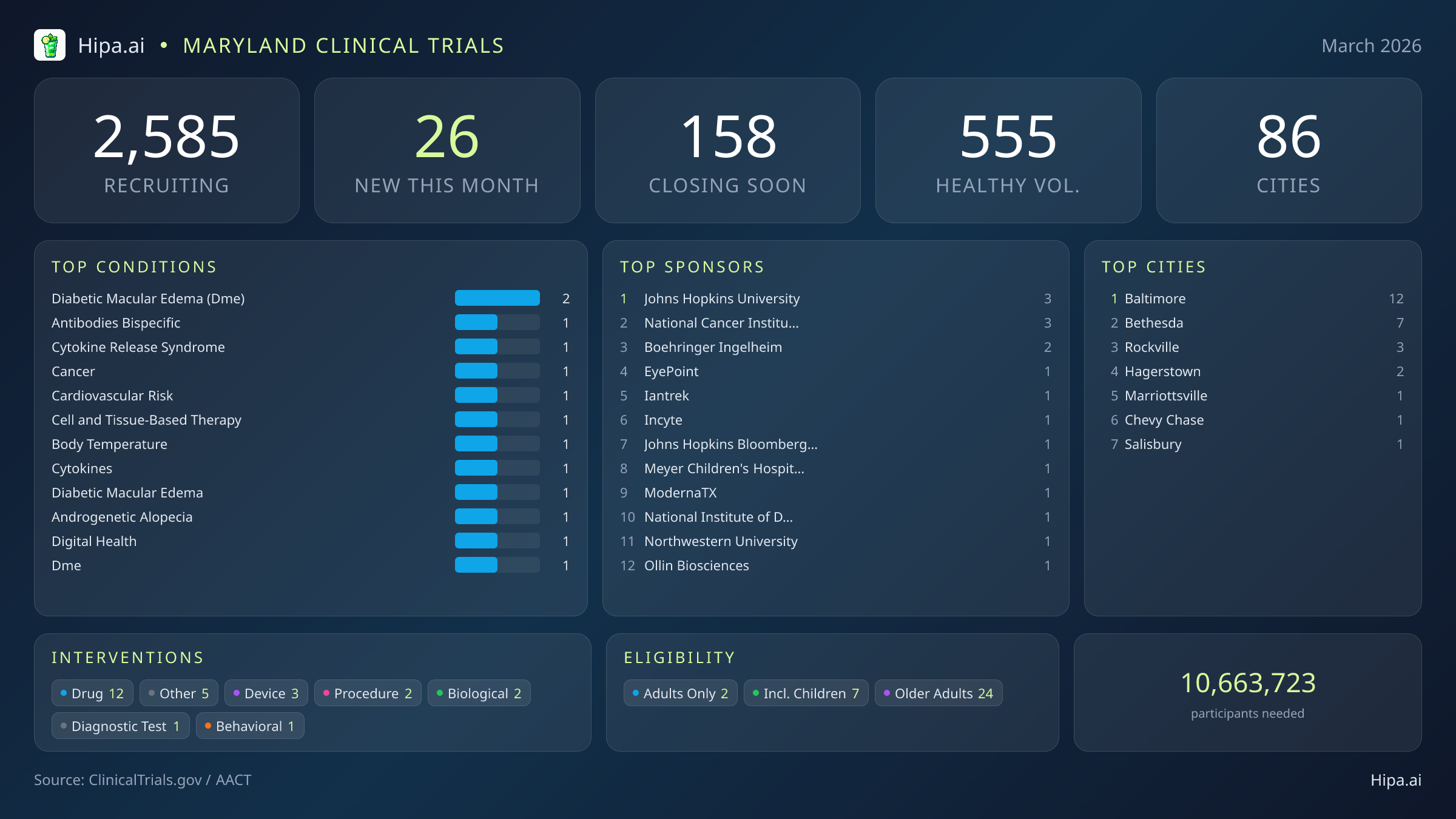
Maryland Clinical Trials Report — March 2026
26 New Studies, 158 Closing Soon
Hipa.ai Research - March 1, 2026 - Source: Research Data
Monthly Clinical Trials Infographic
Approaching Deadlines for Maryland Research Studies
Time is running out for Maryland residents hoping to join several critical medical studies. Within the next ninety days, 158 clinical trials across the state will permanently close their enrollment windows. This shrinking timeline creates an immediate opportunity for people without underlying medical conditions to contribute to scientific advancement, as 46 of these soon-to-close studies are actively seeking healthy volunteers. Patients currently managing specific diagnoses also face a rapidly closing window to access experimental therapies and behavioral interventions. Final enrollment phases are approaching quickly for studies focused on the following conditions:
- Prostate cancer
- Metastatic malignant solid neoplasms
- Post-traumatic stress disorder
- Atopic dermatitis
- Diffuse large B-cell lymphoma
- Metastatic breast cancer
- Chronic pain
- Influenza
High-Profile Phase 3 Launches and Massive Registries
Despite the impending closures, March brings a fresh wave of major research initiatives to the state. While the pace of new study launches has cooled slightly since the beginning of the year—dropping from forty-five new trials in January to 26 this month—the caliber of the incoming research is exceptionally high. Pharmaceutical giants, academic institutions, and federal health agencies are initiating massive late-stage studies. Drug interventions make up the bulk of the new launches with twelve dedicated trials, followed by device testing, procedural research, and biological therapies. Several notable trials are currently seeking participants:
- ModernaTX, Inc. is recruiting four thousand healthy adults for a massive Phase 3 trial (NCT07496450) evaluating the safety and immune response of the mRNA-1018-H5 pandemic influenza vaccine.
- The National Cancer Institute is launching a nationwide registry (NCT07489378) aiming to enroll four thousand children, adolescents, and young adults to gather critical data on very rare solid tumors—defined as cancers diagnosed in two or fewer out of one million people each year.
- Boehringer Ingelheim has opened a Phase 3 study (NCT07472517) testing whether obrixtamig combined with standard chemotherapy improves survival rates for patients with advanced small cell lung cancer.
- Boehringer Ingelheim is also conducting a Phase 3 trial (NCT07497087) evaluating nerandomilast as a potential treatment to improve symptoms and slow disease progression in adults with systemic sclerosis.
- Johns Hopkins University is seeking one thousand participants for a digital health initiative (NCT07478887) designed to optimize lipid-lowering treatments using a smartphone application.
- The Eunice Kennedy Shriver National Institute of Child Health and Human Development is enrolling five hundred individuals for an observational study (NCT07502586) exploring the genetic considerations, heart defects, and long-term health impacts of Turner syndrome.
- The University of California, San Francisco is sponsoring an observational study (NCT07461350) utilizing cardiac MRIs to understand how stimulant and polysubstance use impacts heart scarring and inflammation in patients with HIV.
Targeted Conditions and Inclusive Enrollment Criteria
March's new clinical trials are targeting a highly specific array of chronic, genetic, and acute conditions. Researchers are actively seeking patients recently diagnosed with or currently managing diabetic macular edema, cytokine release syndrome, androgenetic alopecia, and cardiovascular risk. Specialized research is also expanding into rare diseases, such as the Meyer Children's Hospital study (NCT07454343) investigating Erdheim-Chester disease, a rare form of non-Langerhans cell histiocytosis characterized by the accumulation of foamy histiocytes.
Eligibility criteria for this month's new studies are notably inclusive, ensuring that diverse demographic groups can participate in medical advancement. Older adults have extensive options, with 24 of the new trials specifically designing their protocols to include senior populations. Pediatric research is also well-represented, as seven new studies are open to children and adolescents. None of the new trials launched this month restrict participation exclusively by sex, ensuring equal access for both male and female participants.
Statewide Access and Research Hubs
Maryland's clinical research infrastructure remains robust and highly accessible, with 2,585 active trials currently recruiting across eighty-six cities and over one thousand individual research sites. The geographic distribution ensures that residents across the state have access to cutting-edge medical research, though new opportunities are heavily clustered around major academic and federal research hubs.
Baltimore leads the state with twelve newly opened trials, driven largely by institutions like Johns Hopkins University and the Johns Hopkins Bloomberg School of Public Health. The Interstate 270 technology corridor also continues to serve as a major epicenter for clinical research, heavily supported by the National Institutes of Health. New trials have opened their doors this month in several key municipalities:
- Baltimore
- Bethesda
- Rockville
- Hagerstown
- Marriottsville
- Chevy Chase
- Salisbury
Opportunities for Healthy Volunteers
For those without a specific medical diagnosis, two of this month's new trials are actively welcoming healthy volunteers, including the massive Moderna pandemic influenza vaccine study. This brings the total number of open, recruiting trials for healthy participants across Maryland to 555. Whether driven by industry partners like Incyte Corporation and EyePoint Pharmaceuticals, or federal agencies, these trials rely heavily on healthy individuals to establish crucial baseline data for future medical breakthroughs.
As the spring research season progresses, Maryland's clinical trial landscape is positioned to expand its focus on advanced biological therapies and digital health interventions, while the massive datasets collected from this month's pediatric cancer registries will likely shape the next generation of rare disease oncology protocols.
Data Highlights
Conditions Closing Soon
- prostate cancer (3)
- metastatic malignant solid neoplasm (3)
- post traumatic stress disorder (3)
- healthy participants (3)
- atopic dermatitis (3)
- lymphoma (2)
- influenza (2)
- metastatic breast cancer (2)
Most Common New Trial Conditions
- diabetic macular edema (dme) (2)
- antibodies bispecific (1)
- cytokine release syndrome (1)
- cancer (1)
- cardiovascular risk (1)
- cell and tissue-based therapy (1)
- body temperature (1)
- cytokines (1)
Cities With the Most New Trials
- Baltimore (12)
- Bethesda (7)
- Rockville (3)
- Hagerstown (2)
- Marriottsville (1)
- Chevy Chase (1)
- Salisbury (1)
Leading Sponsors
- Johns Hopkins University (3)
- National Cancer Institute (NCI) (3)
- Boehringer Ingelheim (2)
- EyePoint Pharmaceuticals, Inc. (1)
- Iantrek, Inc. (1)
- Incyte Corporation (1)
- Johns Hopkins Bloomberg School of Public Health (1)
- Meyer Children's Hospital IRCCS (1)
| Month | New Trials | Closing Soon |
|---|---|---|
| November 2025 | 36 | 11 |
| December 2025 | 59 | 28 |
| January 2026 | 27 | 13 |
| February 2026 | 41 | 10 |
| March 2026 | 26 | 31 |
| April 2026 | 9 | 0 |