Massachusetts Clinical Trials Report — March 2026
56 New Studies, 218 Closing Soon
Hipa.ai Research - March 1, 2026 - Source: Research Data
Monthly Clinical Trials Infographic
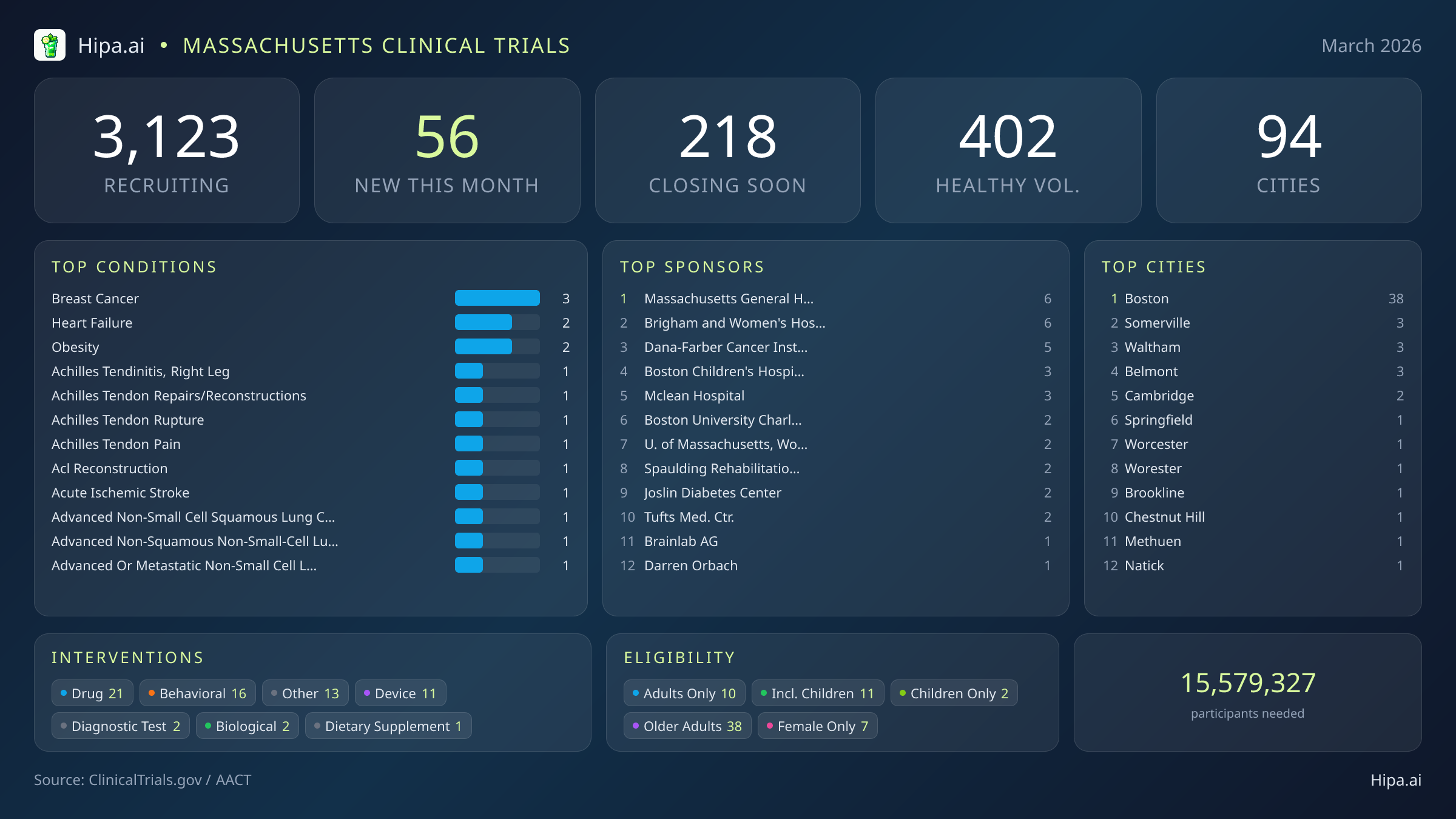
Massachusetts residents have a narrow window to participate in several major medical research initiatives before they permanently close their enrollment phases. Within the next ninety days, 218 clinical trials across the state will stop accepting new participants. This approaching deadline directly impacts individuals managing specific chronic and acute diagnoses, as well as those without underlying conditions. Currently, 49 of these soon-to-close studies are actively seeking healthy volunteers to help establish baseline data for new therapies. Patients seeking experimental treatments or observational studies face an immediate timeline to explore options, particularly those managing the following conditions:
- Major depressive disorder
- Alzheimer's disease
- Prostate cancer
- Coronary artery disease
- Ovarian cancer
- Mild cognitive impairment
- Atrial fibrillation
- Lymphoma
Major Phase 3 Launches and High-Capacity Research
Despite these impending closures, March brings a fresh wave of scientific opportunity to the Commonwealth. Researchers launched 56 new clinical trials this month, representing a slight cooling from the sixty-eight studies opened in January, yet bringing highly anticipated Phase 2 and Phase 3 drug evaluations from major pharmaceutical sponsors. Pfizer, Novo Nordisk, and Novartis are among the industry leaders initiating large-scale protocols in the state, alongside massive data-emulation projects from leading academic institutions.
- Pfizer is recruiting 1,712 participants for a Phase 3 trial (NCT07500506) to evaluate the safety and immunogenicity of a fifth dose of its VLA15 Lyme disease vaccine in healthy individuals aged seven and older.
- Brigham and Women's Hospital is launching two massive data emulation studies (NCT07485166 and NCT07485179) utilizing specialty oncology electronic health records to mirror previous randomized controlled trials, requiring hundreds of participant records to build real-world evidence bases.
- Dartmouth College is sponsoring an observational study (NCT07454733) involving eight hundred participants to determine if video recordings of multidisciplinary clinic visits improve the quality of life for people with amyotrophic lateral sclerosis and their caregivers.
- Boehringer Ingelheim is seeking adults with systemic sclerosis for a Phase 3 trial (NCT07497087) testing whether the drug Nerandomilast can improve symptoms and slow disease progression.
- Novo Nordisk A/S has initiated a Phase 3 investigation (NCT07481630) to determine if weekly subcutaneous injections of NNC0487-0111 can effectively reduce pain and promote weight loss in patients with excess body weight and knee osteoarthritis.
- The MGH Institute of Health Professions is evaluating interventions to optimize functional recovery and daily living activities for individuals who have recently completed treatment for early-stage breast cancer (NCT07489053).
- Novartis Pharmaceuticals is enrolling participants with geographic atrophy secondary to age-related macular degeneration for a Phase 2 trial (NCT07441642) to characterize the dose-response relationship of the experimental drug FWY003.
Targeted Conditions and Broad Eligibility
The landscape of new research targets a highly diverse set of medical conditions. Beyond the large-scale oncology and vaccine trials, investigators are actively seeking patients recently diagnosed with or managing heart failure, obesity, acute ischemic stroke, and advanced non-small cell squamous lung cancer. Orthopedic and sports medicine research is also seeing a localized surge, with multiple new protocols focusing on Achilles tendinitis, Achilles tendon ruptures, and ACL reconstruction.
Participation criteria for these newly launched studies ensure broad community access. Older adults have extensive opportunities, with thirty-eight of the new trials specifically designing their protocols to include senior populations. Pediatric research continues to expand, as eleven new studies are open to children and adolescents. Highly targeted demographic research includes seven trials exclusively enrolling female participants. For residents without a specific medical diagnosis, ten of this month's new trials are actively welcoming healthy volunteers. This brings the total number of open, recruiting trials for healthy participants across Massachusetts to 402.
Pharmaceutical interventions dominate the new research landscape, with twenty-one drug trials launching this month. Behavioral therapies follow closely with sixteen new studies, alongside eleven medical device evaluations, two biological testing protocols, and two diagnostic test assessments. The sponsorship of these trials leans heavily toward academic and medical institutions, with forty studies driven by non-industry organizations, fourteen funded by industry partners, and the remainder supported by federal entities including the National Institutes of Health.
Geographic Spread Across the Commonwealth
The clinical research infrastructure across Massachusetts remains highly robust and accessible. Statewide, there are currently 3,123 active trials recruiting across ninety-four cities and over one thousand individual research sites. As the primary medical hub of New England, Boston naturally dominates the geographic distribution, hosting 38 of the newly opened trials this month. The city's world-renowned academic medical centers serve as the primary engines for this scientific output. Massachusetts General Hospital and Brigham and Women's Hospital lead the state, each sponsoring six new protocols, followed closely by the Dana-Farber Cancer Institute, Boston Children's Hospital, and McLean Hospital.
While the immediate Boston area commands the highest concentration of research activity, cutting-edge medical investigations are not confined to the city limits. Residents living in surrounding suburbs and western regions of the state have access to new trials that opened their doors this month in several other municipalities:
- Somerville
- Waltham
- Belmont
- Cambridge
- Springfield
- Worcester
- Brookline
- Chestnut Hill
As the spring research season accelerates, the Massachusetts clinical trial ecosystem is positioned to pivot toward analyzing the extensive real-world data generated by this quarter's massive oncology emulation studies, while preparing clinical sites for an anticipated influx of advanced orthopedic device evaluations and late-stage metabolic drug investigations.
Data Highlights
Conditions Closing Soon
- major depressive disorder (6)
- healthy (6)
- alzheimer disease (5)
- prostate cancer (5)
- coronary artery disease (4)
- sleep (3)
- ovarian cancer (3)
- mild cognitive impairment (3)
Most Common New Trial Conditions
- breast cancer (3)
- heart failure (2)
- obesity (2)
- achilles tendinitis, right leg (1)
- achilles tendon repairs/reconstructions (1)
- achilles tendon rupture (1)
- achilles tendon pain (1)
- acl reconstruction (1)
Cities With the Most New Trials
- Boston (38)
- Somerville (3)
- Waltham (3)
- Belmont (3)
- Cambridge (2)
- Springfield (1)
- Worcester (1)
- Worester (1)
Leading Sponsors
- Massachusetts General Hospital (6)
- Brigham and Women's Hospital (6)
- Dana-Farber Cancer Institute (5)
- Boston Children's Hospital (3)
- Mclean Hospital (3)
- Boston University Charles River Campus (2)
- University of Massachusetts, Worcester (2)
- Spaulding Rehabilitation Hospital (2)
| Month | New Trials | Closing Soon |
|---|---|---|
| November 2025 | 48 | 11 |
| December 2025 | 73 | 77 |
| January 2026 | 56 | 19 |
| February 2026 | 64 | 28 |
| March 2026 | 56 | 56 |
| April 2026 | 7 | 0 |