Michigan Clinical Trials Report — March 2026
31 New Studies, 122 Closing Soon
Hipa.ai Research - March 1, 2026 - Source: Research Data
Monthly Clinical Trials Infographic
Michigan residents have a brief window to participate in several medical research programs before they permanently stop accepting new patients. Over the next ninety days, 122 clinical trials across the state will close their enrollment phases. This deadline impacts both individuals managing chronic conditions and those without underlying diagnoses, as 12 of these soon-to-close studies are actively seeking healthy volunteers. Patients currently navigating specific health challenges face immediate timelines to join these final enrollment cohorts. Cardiovascular, neurological, and oncology research are particularly affected by this wave of closures. Researchers are urgently seeking participants for studies focused on the following conditions:
- Mild cognitive impairment
- Prostate cancer
- Coronary artery disease
- Inflammatory bowel diseases
- Recurrent endometrial carcinoma
- Stage IV lung cancer
- Atrial fibrillation
- Focal segmental glomerulosclerosis
- HIV-1 infection
Massive Registries and Phase 3 Drug Developments
While many studies are wrapping up, March brings a fresh wave of high-capacity research to the state. The National Minority Quality Forum is launching an unprecedented observational initiative aiming to enroll one hundred thousand participants to investigate health disparities and barriers to cancer screening. Gilead Sciences is also rolling out a massive real-world study to track the use of lenacapavir for HIV pre-exposure prophylaxis across diverse clinical settings. Beyond these large-scale observational efforts, major pharmaceutical sponsors like Boehringer Ingelheim and Eli Lilly and Company are initiating critical Phase 3 drug trials that require hundreds of participants.
- The National Minority Quality Forum is establishing the Cancer Stage Shifting Initiative (NCT07461493), a registry and biorepository seeking one hundred thousand participants to address cancer care disparities and improve access to early detection technologies.
- Gilead Sciences is recruiting three thousand individuals for an implementation study (NCT07473778) evaluating how lenacapavir is initiated, used, and discontinued for HIV prevention in routine clinical care.
- Boehringer Ingelheim has launched the Phase 3 DAREON-Lung-1 trial (NCT07472517), seeking six hundred seventy patients with advanced small cell lung cancer to compare obrixtamig plus standard treatment against standard chemotherapy alone to measure survival improvements.
- The University of Michigan is evaluating the MiWeigh Weight Navigation Program (NCT07465341) with five hundred participants to determine its effectiveness compared to standard weight loss information and to identify which program components drive success.
- Boehringer Ingelheim is also sponsoring a Phase 3 study (NCT07497087) testing whether nerandomilast can improve symptoms and slow disease progression in four hundred forty-eight adults with limited or diffuse cutaneous systemic sclerosis.
- ViroGates A/S is conducting a retrospective cohort study (NCT07466524) utilizing stored plasma samples from three hundred sixty-seven adults hospitalized with confirmed COVID-19 to measure baseline suPAR levels.
- Western Michigan University School of Medicine is testing an augmented reality software system (NCT07504211) designed to improve prehospital pediatric medication dosing accuracy among three hundred twelve EMS providers.
- Whitehawk Therapeutics is initiating a Phase 1 dose-escalation study (NCT07470853) of a targeted antibody-drug conjugate for two hundred sixty-five adults with advanced solid tumors.
Research Hubs and Statewide Accessibility
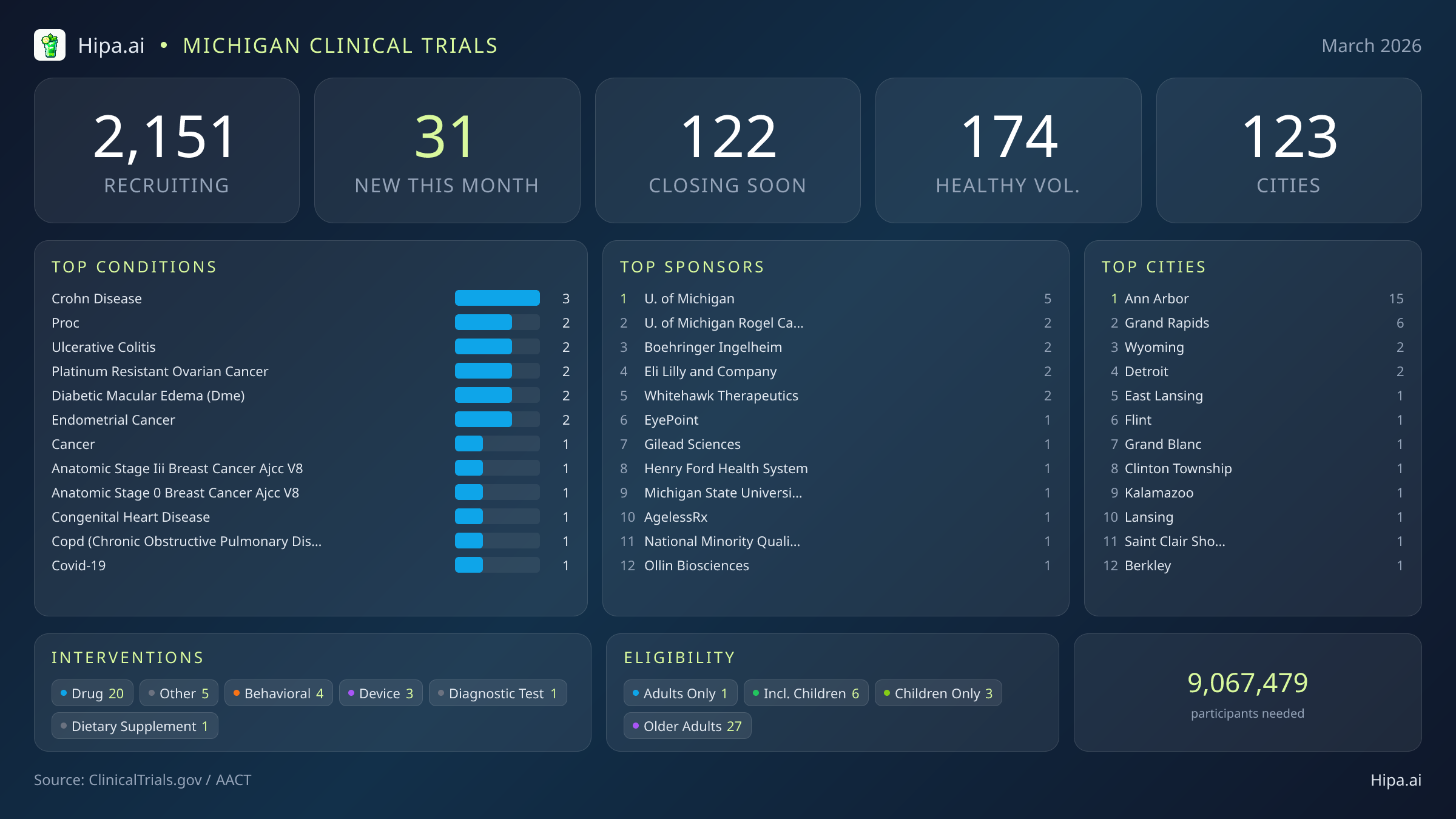
Michigan maintains a robust clinical research infrastructure, with 2,151 active trials currently recruiting across one hundred twenty-three cities and over one thousand three hundred individual research sites. The pace of new study launches remains steady, with 31 new trials opening this month. This represents a slight increase from the twenty-nine studies initiated in February and aligns with the state's consistent output over the past six months, which saw peaks of sixty-two new trials in December and forty-eight in October. Pharmaceutical interventions dominate this month's landscape, accounting for twenty new drug trials. The remaining research portfolio includes five unclassified interventions, four behavioral studies, three medical device evaluations, one diagnostic test, and one dietary supplement trial.
Geographically, the University of Michigan's strong presence heavily anchors the state's research footprint. Ann Arbor leads Michigan with fifteen newly opened trials this month, while Grand Rapids follows with six. The remaining new research opportunities are distributed across a mix of metropolitan and suburban communities, ensuring that residents outside the primary university hubs still have access to experimental therapies. New trials have opened their doors this month in the following municipalities:
- Wyoming
- Detroit
- East Lansing
- Flint
- Grand Blanc
- Clinton Township
- Kalamazoo
- Lansing
Targeted Diagnoses and Broad Eligibility
The clinical trials launched in March address a diverse spectrum of medical conditions. Gastrointestinal research is highly active, with three new trials targeting Crohn's disease and two focusing on ulcerative colitis. Ophthalmology and oncology are also primary focus areas, with new protocols opening for individuals managing diabetic macular edema, platinum-resistant ovarian cancer, endometrial cancer, and early-to-late stage breast cancer. Additional studies are recruiting patients with congenital heart disease. Industry sponsors are driving the majority of this new research, funding eighteen of the recent launches, while academic and regional health systems like the University of Michigan Rogel Cancer Center, EyePoint Pharmaceuticals, and Henry Ford Health System sponsor the remainder.
Eligibility criteria for these new studies emphasize broad demographic inclusion. Older adults have extensive opportunities to participate, with 27 of the new trials specifically designing their protocols to accommodate senior populations. Pediatric research is also represented, as six new studies are open to children and adolescents. None of this month's trials restrict participation exclusively to males or females, ensuring equitable access across gender lines. For residents without a specific medical diagnosis, three of this month's new trials are actively welcoming healthy volunteers. This brings the total number of open, recruiting trials for healthy participants across Michigan to 174.
As the state moves deeper into the spring, the clinical research ecosystem is positioned to transition from active enrollment toward the complex data analysis required for these massive new cancer registries and infectious disease prevention programs.
Data Highlights
Conditions Closing Soon
- mild cognitive impairment (2)
- prostate cancer (2)
- coronary artery disease (2)
- inflammatory bowel diseases (2)
- recurrent endometrial carcinoma (2)
- stage iv lung cancer ajcc v8 (2)
- atrial fibrillation (2)
- cardiovascular diseases (2)
Most Common New Trial Conditions
- crohn disease (3)
- proc (2)
- ulcerative colitis (2)
- platinum resistant ovarian cancer (2)
- diabetic macular edema (dme) (2)
- endometrial cancer (2)
- cancer (1)
- anatomic stage iii breast cancer ajcc v8 (1)
Cities With the Most New Trials
- Ann Arbor (15)
- Grand Rapids (6)
- Wyoming (2)
- Detroit (2)
- East Lansing (1)
- Flint (1)
- Grand Blanc (1)
- Clinton Township (1)
Leading Sponsors
- University of Michigan (5)
- University of Michigan Rogel Cancer Center (2)
- Boehringer Ingelheim (2)
- Eli Lilly and Company (2)
- Whitehawk Therapeutics, Inc. (2)
- EyePoint Pharmaceuticals, Inc. (1)
- Gilead Sciences (1)
- Henry Ford Health System (1)
| Month | New Trials | Closing Soon |
|---|---|---|
| November 2025 | 28 | 8 |
| December 2025 | 62 | 31 |
| January 2026 | 35 | 16 |
| February 2026 | 29 | 12 |
| March 2026 | 31 | 37 |
| April 2026 | 6 | 0 |