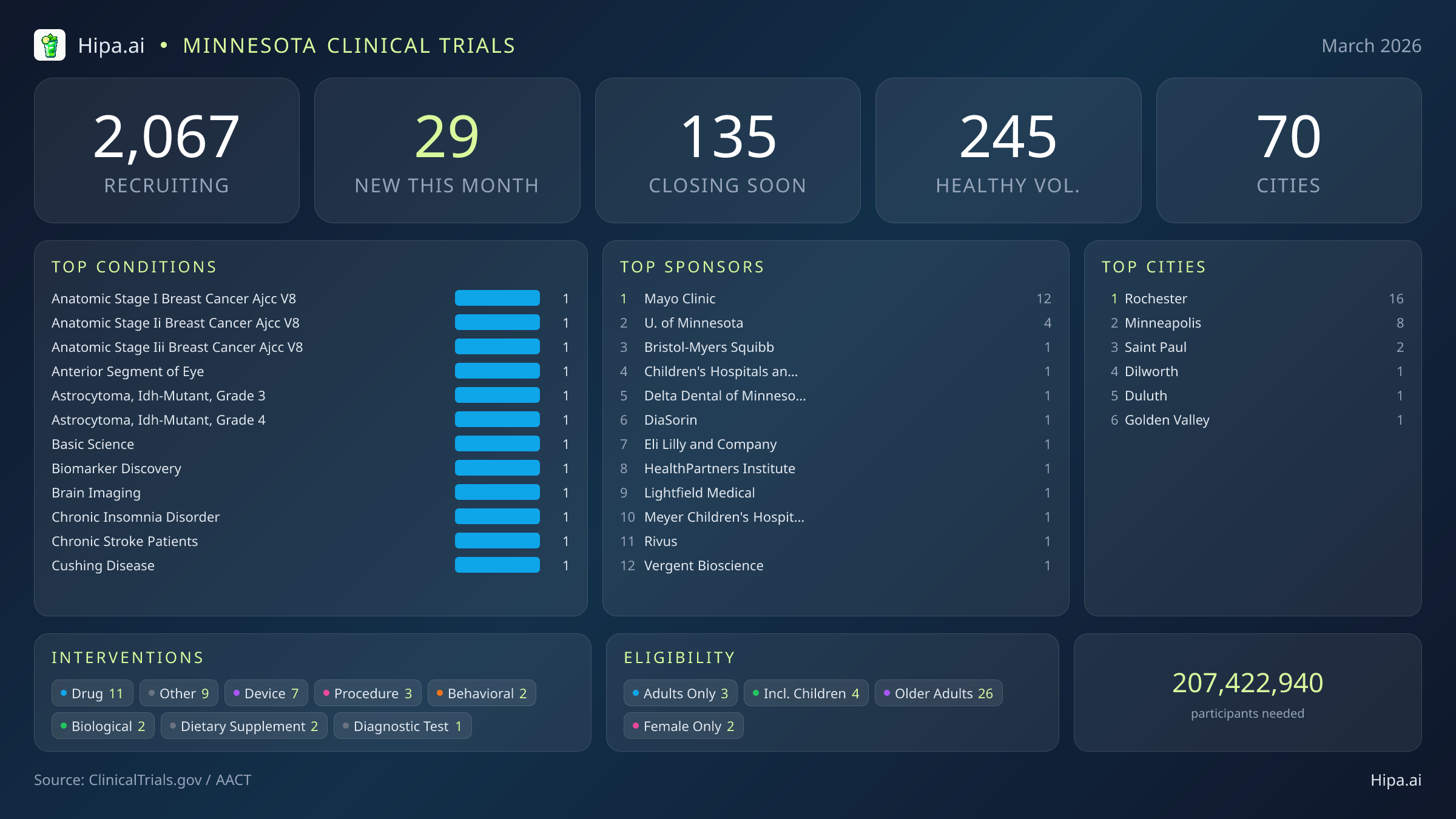
Minnesota Clinical Trials Report — March 2026
29 New Studies, 135 Closing Soon
Hipa.ai Research - March 1, 2026 - Source: Research Data
Monthly Clinical Trials Infographic
Approaching Deadlines for Minnesota Research Programs
A critical window is closing for Minnesotans seeking access to experimental medical therapies and observational studies. Within the next ninety days, 135 clinical trials across the state will permanently close their enrollment phases. This impending deadline presents a unique opportunity for people without underlying medical conditions to contribute to scientific advancement, as twenty-six of these soon-to-close studies are actively seeking healthy volunteers. Patients currently managing specific diagnoses also face a rapidly shrinking timeframe to access experimental interventions before these programs stop accepting new participants. Final enrollment phases are approaching quickly for studies focused on the following conditions:
- Coronary artery disease and cardiovascular diseases
- Multiple myeloma
- Prostate cancer
- Lymphoma
- Gastroesophageal cancer and esophageal adenocarcinoma
- Barrett's esophagus with dysplasia
- Atrial fibrillation
Major Medical Institutions Drive March Trial Launches
Despite the impending closures, March brings a fresh wave of high-profile research to the state, heavily driven by leading academic and medical institutions. The pace of new study launches has rebounded slightly, with 29 new trials opening this month compared to twenty-six in January. The Mayo Clinic leads the charge, sponsoring twelve of these new initiatives, while the University of Minnesota and industry partners like Eli Lilly and Bristol-Myers Squibb are also expanding their local research footprints. Several notable trials are currently seeking participants across the state:
- The Mayo Clinic is spearheading a massive descriptive study (NCT07455370) aiming to enroll over twenty-one thousand nursing staff members to examine levels of moral distress, moral injury, and post-traumatic stress.
- Meyer Children's Hospital IRCCS is recruiting one thousand participants for a study (NCT07454343) focused on Erdheim-Chester disease, a rare form of non-Langerhans cell histiocytosis that primarily affects adults.
- Children's Hospitals and Clinics of Minnesota is launching a critical public health initiative (NCT07482982) to evaluate firearm screening protocols in the pediatric emergency department, seeking nearly eight hundred participants.
- The Mayo Clinic is conducting a specialized cardiovascular study (NCT07483203) to characterize pulmonary hemodynamic profiles in six hundred patients with left-sided valvular heart disease undergoing valve intervention.
- Delta Dental of Minnesota Foundation is sponsoring a large-scale population study (NCT07451782) surveying nearly six hundred individuals to understand oral hygiene habits and the implications of non-pharmaceutical dental hygiene products.
- Yale University is rolling out a multisite study (NCT07475728) to test the feasibility and safety of EMS-initiated buprenorphine for patients requiring emergency medical services.
- Eli Lilly and Company is seeking four hundred fifty healthy volunteers for a Phase 1 trial (NCT07446101) to assess the safety and tolerability of mirikizumab solution administered via injection.
- DiaSorin Inc. is evaluating a novel stool test biomarker combination (NCT07471490) to distinguish inflammatory bowel disease from irritable bowel syndrome in three hundred participants.
Emerging Focus Areas and Therapeutic Interventions
March's new clinical trials are targeting a highly specific array of chronic, genetic, and oncological conditions. Researchers are actively seeking patients recently diagnosed with or currently managing anatomic stage I, II, and III breast cancer, as well as grade 3 and 4 IDH-mutant astrocytoma. Other newly opened studies focus on chronic insomnia disorder, conditions affecting the anterior segment of the eye, and broader biomarker discovery and brain imaging initiatives.
Pharmaceutical and medical device testing make up a significant portion of this month's research portfolio. Of the newly launched studies, eleven are focused on experimental drug interventions, while seven are evaluating new medical devices. The remaining trials encompass a diverse mix of procedural, behavioral, biological, and dietary supplement interventions, reflecting a comprehensive approach to advancing patient care across multiple medical disciplines. Academic and non-industry sponsors are driving the majority of this month's new research, accounting for twenty-one of the newly launched trials.
Opportunities for Healthy Volunteers and Broad Demographics
Eligibility criteria for this month's new studies are notably inclusive, ensuring that diverse demographic groups can participate in medical advancement. Older adults have extensive options, with twenty-six of the new trials specifically designing their protocols to include senior populations. Pediatric research is also represented, as four new studies are open to children and adolescents. Two of the new trials launched this month restrict participation exclusively to females, while none are restricted solely to males.
For those without a specific medical diagnosis, 9 of this month's new trials are actively welcoming healthy volunteers. This brings the total number of open, recruiting trials for healthy participants across Minnesota to 245. Whether driven by industry partners like HealthPartners Institute or academic heavyweights like the University of Minnesota, these trials rely heavily on healthy individuals to establish crucial baseline data for future medical breakthroughs.
Geographic Distribution Across the North Star State
Minnesota's clinical research infrastructure remains robust and highly accessible, with 2,067 active trials currently recruiting across seventy cities and over seven hundred individual research sites. The geographic distribution ensures that residents across the state have access to cutting-edge medical research, though this month's new launches are heavily concentrated in major medical hubs.
Rochester leads the state with sixteen newly opened trials, largely driven by the Mayo Clinic's extensive research campus. The Twin Cities metropolitan area also provides significant access points, with Minneapolis hosting eight new trials and Saint Paul adding two. Residents living outside these primary urban centers still have opportunities to participate, as new trials have opened their doors this month in the following municipalities:
- Rochester
- Minneapolis
- Saint Paul
- Dilworth
- Duluth
- Golden Valley
As the spring research season progresses, Minnesota's clinical trial landscape is positioned to expand its focus on advanced oncological therapies and behavioral health interventions, while the massive datasets collected from this month's observational studies will likely shape the next generation of healthcare workforce support systems.
Data Highlights
Conditions Closing Soon
- coronary artery disease (4)
- multiple myeloma (3)
- prostate cancer (3)
- cardiovascular diseases (3)
- healthy (3)
- lymphoma (2)
- gastroesophageal cancer (2)
- esophageal adenocarcinoma (2)
Most Common New Trial Conditions
- anatomic stage i breast cancer ajcc v8 (1)
- anatomic stage ii breast cancer ajcc v8 (1)
- anatomic stage iii breast cancer ajcc v8 (1)
- anterior segment of eye (1)
- astrocytoma, idh-mutant, grade 3 (1)
- astrocytoma, idh-mutant, grade 4 (1)
- basic science (1)
- biomarker discovery (1)
Cities With the Most New Trials
- Rochester (16)
- Minneapolis (8)
- Saint Paul (2)
- Dilworth (1)
- Duluth (1)
- Golden Valley (1)
Leading Sponsors
- Mayo Clinic (12)
- University of Minnesota (4)
- Bristol-Myers Squibb (1)
- Children's Hospitals and Clinics of Minnesota (1)
- Delta Dental of Minnesota Foundation (1)
- DiaSorin Inc. (1)
- Eli Lilly and Company (1)
- HealthPartners Institute (1)
| Month | New Trials | Closing Soon |
|---|---|---|
| November 2025 | 29 | 7 |
| December 2025 | 44 | 27 |
| January 2026 | 38 | 11 |
| February 2026 | 24 | 7 |
| March 2026 | 29 | 35 |
| April 2026 | 5 | 0 |