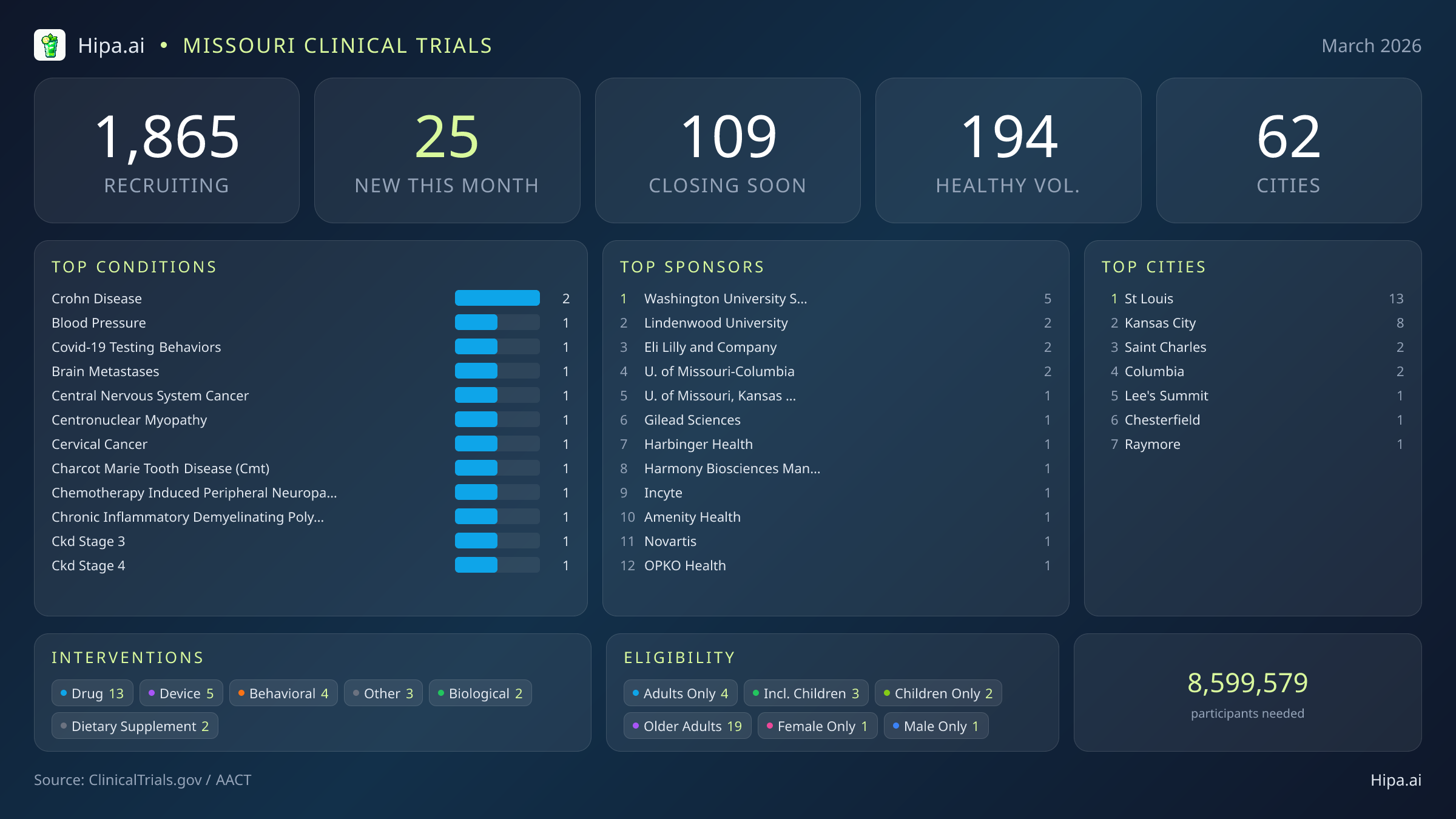
Missouri Clinical Trials Report — March 2026
25 New Studies, 109 Closing Soon
Hipa.ai Research - March 1, 2026 - Source: Research Data
Monthly Clinical Trials Infographic
Approaching Deadlines for Missouri Research Participants
Time is strictly limited for Missouri residents seeking access to several critical medical research programs. Over the next ninety days, 109 clinical trials across the state will permanently close their enrollment windows. For individuals currently managing chronic or terminal diagnoses, this represents a rapidly shrinking opportunity to access experimental therapies, behavioral interventions, and advanced medical devices before they potentially reach the broader commercial market. These impending closures also impact people without underlying medical conditions, as 12 of these soon-to-close studies are actively seeking healthy volunteers to provide essential baseline data. Final enrollment phases are approaching quickly for studies focused on the following conditions:
- Prostate cancer and prostate adenocarcinoma
- Alzheimer disease
- Sickle cell disease
- Sarcopenia
- Stage IV lung cancer
- Breast and gastric cancers
- Obesity and aging
Late-Stage Drug Trials and Industry Innovation
Despite the impending closures, March brings a fresh wave of high-profile research to the state. While the pace of new study launches has cooled slightly—dropping from thirty-nine new trials in January to 25 this month—the caliber of incoming research remains exceptionally high. The research landscape this month is perfectly balanced between commercial and institutional efforts, with thirteen trials sponsored by industry leaders and twelve driven by academic or other organizations. Thirteen of these newly opened studies are focused on drug interventions, while others explore medical devices, behavioral therapies, biologicals, and dietary supplements. Several notable therapeutic trials from major sponsors like Eli Lilly, Novartis, and Harmony Biosciences are currently seeking participants:
- Harmony Biosciences Management, Inc. is recruiting two hundred forty-eight participants for a Phase 3 trial (NCT07500090) to assess the efficacy of HBS-301 in treating idiopathic hypersomnia symptoms, including excessive daytime sleepiness, sleep inertia, and fatigue.
- Rivus Pharmaceuticals, Inc. is enrolling one hundred eighty participants in a Phase 2 trial (NCT07491458) evaluating HU6 as a potential treatment for metabolic dysfunction-associated steatohepatitis, while also measuring changes in liver fat.
- Novartis Pharmaceuticals has launched a Phase 2 study (NCT07467993) testing the safety, tolerability, and treatment response of GXV813 in hospitalized adults experiencing an acute episode of schizophrenia.
- Incyte Corporation is seeking two hundred eighteen volunteers for a Phase 1 study (NCT07441694) to determine the safety and maximum tolerated dose of INCA036978, both as a monotherapy and in combination with standard therapies, for participants with myeloproliferative neoplasms.
- Washington University School of Medicine is conducting a Phase 2 imaging trial (NCT07462507) using specialized PET scans to describe patterns of tracer uptake in adult patients with primary or secondary central nervous system malignancies.
Large-Scale Community Health and Diagnostic Research
Beyond traditional pharmaceutical testing, medical institutions are launching massive observational and behavioral studies this month. Gilead Sciences is spearheading an enormous observational study (NCT07473778) aiming to enroll three thousand participants. This project will generate real-world data on how lenacapavir is initiated, used, and discontinued for HIV pre-exposure prophylaxis across diverse clinical settings in the United States. Harbinger Health is also initiating a massive diagnostic effort (NCT07479381), seeking nearly fifteen hundred participants to evaluate a new blood-based screening test designed to detect colorectal cancer.
Community-based interventions are also taking center stage. The University of Missouri, Kansas City is sponsoring a Phase 2 behavioral trial (NCT07450209) that will recruit nine hundred adults. This two-arm clustered, randomized community trial will test a religiously-tailored COVID-19 rapid testing and treatment intervention within African American churches, comparing it against a standard educational approach.
March's new clinical trials are also targeting a highly specific array of chronic, neurological, and genetic conditions. Researchers are actively seeking patients recently diagnosed with or currently managing:
- Crohn disease
- Chemotherapy-induced peripheral neuropathy
- Centronuclear myopathy
- Charcot-Marie-Tooth disease
- Chronic inflammatory demyelinating polyneuropathy
- Cervical cancer and brain metastases
- High blood pressure
Research Hubs and Statewide Accessibility
Missouri's clinical research infrastructure remains robust and highly accessible, with 1,865 active trials currently recruiting across sixty-two cities and over nine hundred individual research sites. Geographically, new research opportunities are heavily clustered in the state's two largest metropolitan areas, ensuring that a significant portion of the population has direct access to cutting-edge medical research.
St. Louis leads the state with thirteen newly opened trials, bolstered by the strong presence of Washington University School of Medicine, which is sponsoring five new studies this month alone. On the western side of the state, Kansas City follows closely with eight new studies. Academic contributions from Lindenwood University and the University of Missouri-Columbia further expand the state's research footprint. Residents living outside the immediate urban cores still have opportunities to participate, as new trials have opened their doors this month in several key municipalities:
- St. Louis
- Kansas City
- Saint Charles
- Columbia
- Lee's Summit
- Chesterfield
- Raymore
Broad Eligibility and Healthy Volunteer Opportunities
Eligibility criteria for this month's new studies are notably inclusive, ensuring that diverse demographic groups can participate in medical advancement. Older adults have extensive options, with nineteen of the new trials specifically designing their protocols to include senior populations. Pediatric research is also represented, as three new studies are open to children and adolescents. Gender-specific research remains highly targeted, with only one new trial restricting participation exclusively to females and one exclusively to males.
For those without a specific medical diagnosis, 7 of this month's new trials are actively welcoming healthy volunteers. This brings the total number of open, recruiting trials for healthy participants across Missouri to 194. These trials rely heavily on healthy individuals to establish crucial baseline data for future medical breakthroughs, offering a direct way for the general public to contribute to scientific progress without needing a prior diagnosis.
As the spring research season accelerates, Missouri's clinical trial landscape is positioned to drive significant advancements in community-based infectious disease management and targeted neurological therapies, shaping the next generation of preventative healthcare protocols.
Data Highlights
Conditions Closing Soon
- prostate cancer (3)
- alzheimer disease (3)
- sickle cell disease (2)
- prostate adenocarcinoma (2)
- sarcopenia (2)
- stage iv lung cancer ajcc v8 (2)
- breast cancer (2)
- aging (2)
Most Common New Trial Conditions
- crohn disease (2)
- blood pressure (1)
- covid-19 testing behaviors (1)
- brain metastases (1)
- central nervous system cancer (1)
- centronuclear myopathy (1)
- cervical cancer (1)
- charcot marie tooth disease (cmt) (1)
Cities With the Most New Trials
- St Louis (13)
- Kansas City (8)
- Saint Charles (2)
- Columbia (2)
- Lee's Summit (1)
- Chesterfield (1)
- Raymore (1)
Leading Sponsors
- Washington University School of Medicine (5)
- Lindenwood University (2)
- Eli Lilly and Company (2)
- University of Missouri-Columbia (2)
- University of Missouri, Kansas City (1)
- Gilead Sciences (1)
- Harbinger Health (1)
- Harmony Biosciences Management, Inc. (1)
| Month | New Trials | Closing Soon |
|---|---|---|
| November 2025 | 20 | 7 |
| December 2025 | 56 | 27 |
| January 2026 | 26 | 10 |
| February 2026 | 37 | 8 |
| March 2026 | 25 | 31 |
| April 2026 | 3 | 0 |