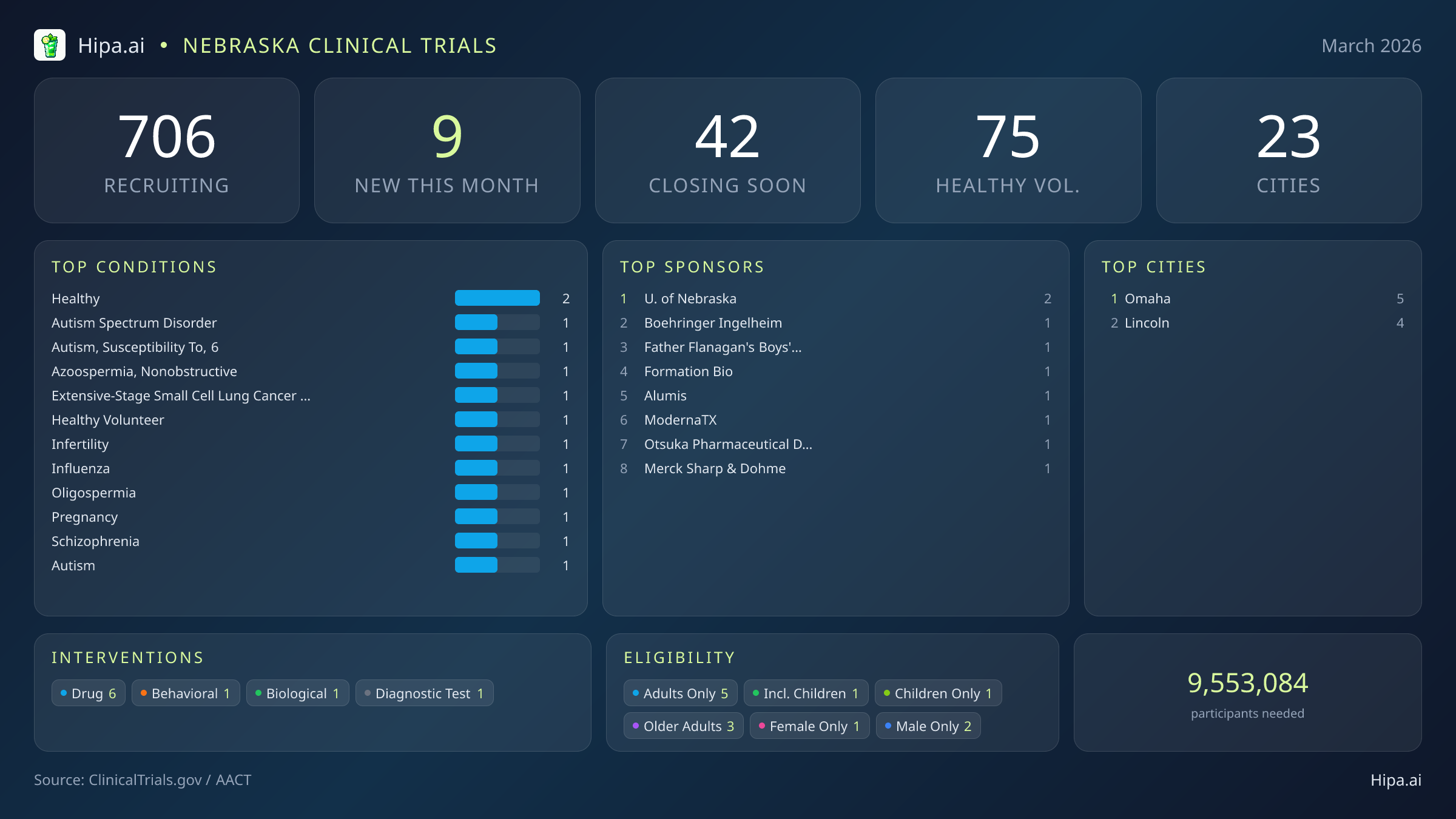
Nebraska Clinical Trials Report — March 2026
9 New Studies, 42 Closing Soon
Hipa.ai Research - March 1, 2026 - Source: Research Data
Monthly Clinical Trials Infographic
Immediate Enrollment Deadlines for Nebraska Residents
Time is strictly limited for Nebraskans hoping to join several critical medical research initiatives. Over the next ninety days, 42 clinical trials will permanently close their enrollment windows across the state. This impending deadline creates a distinct opportunity for individuals without underlying health conditions to participate in scientific discovery, as 9 of these closing studies are actively seeking healthy volunteers. For patients currently managing specific diagnoses, the window to access experimental therapies and behavioral interventions is rapidly narrowing.
Cardiovascular research is particularly affected this month, with multiple studies focusing on heart failure, atrial fibrillation, and coronary artery disease nearing their final enrollment dates. Oncology trials are also facing strict deadlines, specifically for patients managing colorectal cancer and cancers harboring BRAF alterations. Final enrollment phases are approaching quickly for studies focused on the following conditions:
- Heart failure and coronary artery disease
- Colorectal cancer
- Asthma and chronic stress
- Aortic stenosis and atrial fibrillation
- Anaplastic oligodendroglioma
- Alcohol drinking behaviors
Major Pharmaceutical and Academic Trial Launches
March brings a revitalized wave of high-profile research to the state, with 9 new trials opening this month. While this matches the research output seen earlier this year in January, it represents a steady stabilization following the significant peak of twenty-one new trials recorded at the end of last year. The current landscape is heavily driven by commercial investment, with six of the new studies sponsored by industry leaders. The remaining three are spearheaded by academic and institutional organizations, including the University of Nebraska and Father Flanagan's Boys' Home.
Six of these newly launched studies focus on pharmaceutical interventions, offering local patients access to experimental drugs before they reach the broader market. The remaining new studies are diversifying the state's research portfolio by introducing one behavioral intervention, one biological study, and one diagnostic test trial. Several notable therapeutic trials from major sponsors—including Formation Bio, Alumis Inc, Otsuka Pharmaceutical Development, and Merck Sharp & Dohme—are currently seeking participants. Standout trials launching this month include:
- ModernaTX, Inc. is recruiting four thousand participants for a massive Phase 3 trial (NCT07496450) evaluating a pandemic influenza vaccine. This study will assess humoral immunogenicity, safety, and reactogenicity after two doses of mRNA-1018-H5 in healthy adults.
- Boehringer Ingelheim has launched a Phase 3 study (NCT07472517) testing obrixtamig in combination with standard treatments like atezolizumab, carboplatin, and etoposide. This trial is open to adults with advanced small cell lung cancer to determine if the experimental regimen improves survival rates compared to standard chemotherapy alone.
- The University of Nebraska is conducting a highly specialized Phase 2 trial (NCT07481370) comparing two medical treatments, isotretinoin and hCG, for male infertility. This study aims to see how well these treatments improve sperm production in men managing low sperm counts or nonobstructive azoospermia, conditions that historically have limited nonsurgical options.
Expanding Opportunities for Healthy Volunteers and Specific Demographics
Individuals without specific medical diagnoses have an exceptional opportunity to contribute to scientific discovery this month. Six of the newly opened trials are actively welcoming healthy volunteers. This surge in accessibility brings the total number of open, recruiting trials for healthy participants across Nebraska to 75. These studies rely heavily on healthy individuals to establish crucial baseline data for future medical breakthroughs, particularly in large-scale biological and vaccine initiatives like the one sponsored by Moderna.
Eligibility criteria for this month's new studies span a variety of demographics. Three of the new trials have designed their protocols to include older adults, ensuring that aging populations are represented in medical advancement. Pediatric research is represented by a single new study open to children. Additionally, researchers are conducting highly targeted studies based on sex, with two trials restricting participation exclusively to men and one trial open only to female patients. Beyond healthy volunteers, these new studies are actively seeking individuals recently diagnosed with or currently managing specific conditions, including:
- Autism spectrum disorder and autism susceptibility
- Extensive-stage small cell lung cancer
- Nonobstructive azoospermia and oligospermia
- Pregnancy and general infertility
- Influenza
Geographic Distribution and Statewide Access
Nebraska's clinical research infrastructure remains highly accessible. Statewide, there are currently 706 active trials recruiting across twenty-three cities and 394 individual research sites. Geographically, this month's new research opportunities are heavily concentrated in the state's two largest metropolitan areas, creating dense hubs of medical innovation.
Omaha leads the state with five newly opened trials, providing a concentration of advanced care options and academic research facilities. Lincoln follows closely, hosting the remaining four new studies launched this month. This clustering ensures that residents in the eastern half of the state have immediate access to cutting-edge medical interventions. However, the broader statewide network continues to support hundreds of ongoing trials. This extensive infrastructure ensures that patients living outside these primary eastern hubs still have avenues to participate in clinical research, bridging the gap between urban medical centers and rural communities.
As the spring research season advances, Nebraska's clinical trial landscape is positioned to drive significant advancements in pandemic preparedness and targeted oncology, while local academic institutions will likely continue expanding their focus on specialized reproductive health interventions.
Data Highlights
Conditions Closing Soon
- heart failure (2)
- coronary artery disease (2)
- alcohol drinking (1)
- anaplastic oligodendroglioma (1)
- aortic stenosis (1)
- asthma (1)
- cancer harboring braf alterations (1)
- atrial fibrillation (1)
Most Common New Trial Conditions
- healthy (2)
- autism spectrum disorder (1)
- autism, susceptibility to, 6 (1)
- azoospermia, nonobstructive (1)
- extensive-stage small cell lung cancer (es-sclc) (1)
- healthy volunteer (1)
- infertility (1)
- influenza (1)
Leading Sponsors
- University of Nebraska (2)
- Boehringer Ingelheim (1)
- Father Flanagan's Boys' Home (1)
- Formation Bio, Inc. (1)
- Alumis Inc (1)
- ModernaTX, Inc. (1)
- Otsuka Pharmaceutical Development & Commercialization, Inc. (1)
- Merck Sharp & Dohme LLC (1)
| Month | New Trials | Closing Soon |
|---|---|---|
| November 2025 | 12 | 1 |
| December 2025 | 21 | 9 |
| January 2026 | 6 | 1 |
| February 2026 | 9 | 2 |
| March 2026 | 9 | 9 |
| April 2026 | 5 | 0 |