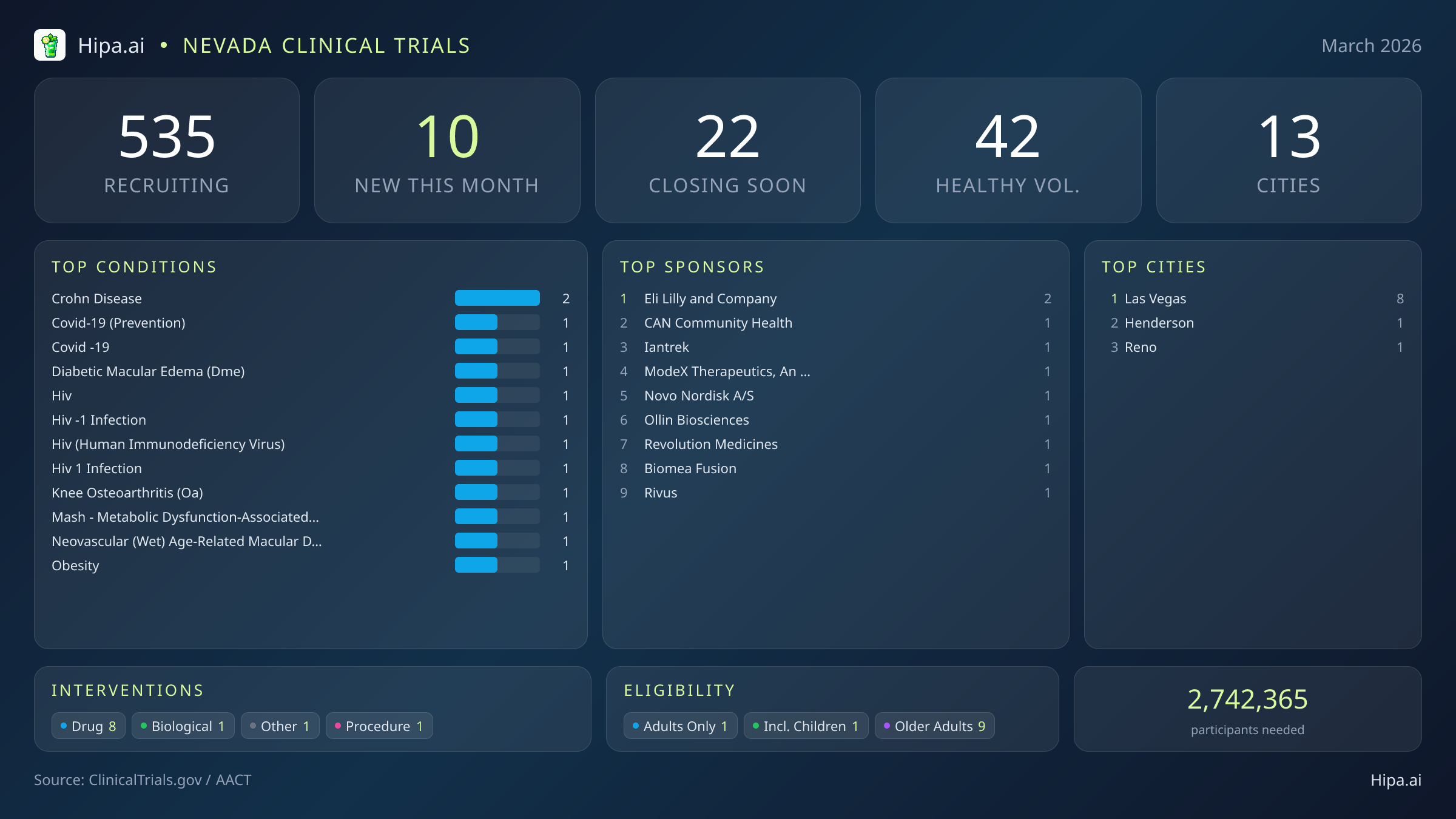
Nevada Clinical Trials Report — March 2026
10 New Studies, 22 Closing Soon
Hipa.ai Research - March 1, 2026 - Source: Research Data
Monthly Clinical Trials Infographic
Immediate Deadlines for Nevada Research Studies
Time is strictly limited for Nevada residents hoping to join several critical medical research initiatives. Over the next ninety days, 22 clinical trials will permanently close their enrollment windows statewide. This impending cutoff creates a narrow window for patients seeking access to experimental therapies before they are no longer available to the public. For individuals without underlying health conditions, 4 of these closing studies are still actively accepting healthy volunteers, offering a final chance to contribute to baseline scientific data. Final enrollment phases are rapidly approaching for studies focused on the following conditions:
- Attention deficit hyperactivity disorder (ADHD)
- Advanced solid tumors and advanced cancer
- Anatomic stage II and III breast cancer
- Asthma and chronic obstructive pulmonary disease
- Brain fog
- Endometrial adenocarcinoma
Major Pharmaceutical Pipelines and Late-Stage Trials
March brings a revitalized wave of high-profile research to the state, with 10 new trials opening this month. This represents a steady increase from the seven studies launched in February and nine in January, stabilizing a fluctuating winter research season that saw a peak of thirty trials in December. Industry leaders are driving this wave of clinical development, sponsoring nine of the newly launched studies. Eight of these new initiatives are focused on testing investigational drugs, providing local patients with access to experimental treatments before they reach the broader market. Several notable therapeutic trials from major pharmaceutical sponsors are currently seeking participants:
- Revolution Medicines is recruiting nine hundred participants for a massive Phase 3 trial (NCT07491445) evaluating daraxonrasib as a first-line treatment for metastatic pancreatic adenocarcinoma. The study compares this investigational inhibitor, alone or with chemotherapy, against standard care.
- Novo Nordisk has launched a Phase 3 study (NCT07481630) testing weekly subcutaneous injections of NNC0487-0111 to determine if the medication safely helps individuals with excess body weight and knee osteoarthritis lose weight and reduce joint pain.
- Eli Lilly and Company is conducting two distinct Phase 2 trials under a master protocol for inflammatory bowel diseases. One study (NCT07483073) evaluates multiple drugs for adults with ulcerative colitis or Crohn's disease, while the second (NCT07483099) specifically tests an FXR agonist combined with mirikizumab for moderately to severely active Crohn's disease.
- Rivus Pharmaceuticals is seeking volunteers for a Phase 2 trial (NCT07491458) assessing the safety and efficacy of HU6 as a potential treatment for metabolic dysfunction-associated steatohepatitis (MASH), focusing on changes in liver fat.
- Biomea Fusion has opened a Phase 2 trial (NCT07502495) testing icovamenib in participants with Type 2 diabetes who are struggling to achieve glycemic targets despite using standard antihyperglycemic medications.
Emerging Treatments for Chronic and Infectious Diseases
Beyond the large-scale metabolic and oncology trials, this month's new clinical research targets a highly specific array of chronic conditions and infectious diseases. Researchers are actively seeking patients recently diagnosed with or currently managing Crohn's disease, diabetic macular edema, and knee osteoarthritis. Infectious disease research also sees a notable boost this month, with multiple new protocols opening for HIV infection, HIV-1, and COVID-19 prevention.
While drug trials dominate the landscape, procedural, biological, and observational research continues to play a vital role in medical advancement. Iantrek is currently enrolling participants for a long-term observational study (NCT07480343) to collect five-year postoperative outcomes from patients who previously completed the CREST study. This research focuses on individuals who managed their intraocular pressure without needing secondary surgery, ensuring the long-term safety and efficacy of glaucoma interventions.
Research Hubs and Statewide Accessibility
Nevada's clinical research infrastructure remains highly active and accessible, with 535 trials currently recruiting across thirteen cities and nearly four hundred individual research sites. Geographically, new research opportunities are heavily clustered in the southern portion of the state, creating a centralized hub for advanced medical care. Las Vegas leads the region with eight newly opened trials, providing a dense concentration of options for residents in the metropolitan area. However, the research footprint extends beyond the primary city center, ensuring that patients outside the immediate Vegas strip have access to cutting-edge science. New trials have opened their doors this month in the following municipalities:
- Las Vegas
- Henderson
- Reno
Broad Eligibility and Community Participation
Eligibility criteria for this month's new studies are heavily skewed toward adult and senior populations, ensuring that aging demographics can participate in medical advancement. Older adults have extensive options, with nine of the new trials specifically designing their protocols to include senior participants. Pediatric research is represented by a single new study open to children. None of the new trials launched this month restrict participation exclusively to men or exclusively to women, ensuring broad gender access across all newly available therapies.
For those without a specific medical diagnosis, one of this month's newly launched trials is actively welcoming healthy volunteers. This brings the total number of open, recruiting trials for healthy participants across Nevada to 42. These trials rely heavily on healthy individuals to establish crucial baseline data for future medical breakthroughs, making community participation essential for scientific progress.
As the spring research season advances, Nevada's clinical trial landscape is positioned to expand its focus on metabolic therapies and advanced gastrointestinal treatments, while the integration of novel inhibitors in oncology will continue to shape the future of cancer care.
Data Highlights
Conditions Closing Soon
- adhd (1)
- advanced cancer (1)
- advanced solid tumors (1)
- anatomic stage ii breast cancer ajcc v8 (1)
- anatomic stage iii breast cancer ajcc v8 (1)
- asthma (1)
- brain fog (1)
- breast cancer (1)
Most Common New Trial Conditions
- crohn disease (2)
- covid-19 (prevention) (1)
- covid -19 (1)
- diabetic macular edema (dme) (1)
- hiv (1)
- hiv -1 infection (1)
- hiv (human immunodeficiency virus) (1)
- hiv 1 infection (1)
Leading Sponsors
- Eli Lilly and Company (2)
- CAN Community Health (1)
- Iantrek, Inc. (1)
- ModeX Therapeutics, An OPKO Health Company (1)
- Novo Nordisk A/S (1)
- Ollin Biosciences, Inc. (1)
- Revolution Medicines, Inc. (1)
- Biomea Fusion Inc. (1)
| Month | New Trials | Closing Soon |
|---|---|---|
| October 2025 | 24 | 2 |
| November 2025 | 3 | 1 |
| December 2025 | 30 | 6 |
| January 2026 | 9 | 5 |
| February 2026 | 7 | 1 |
| March 2026 | 10 | 7 |