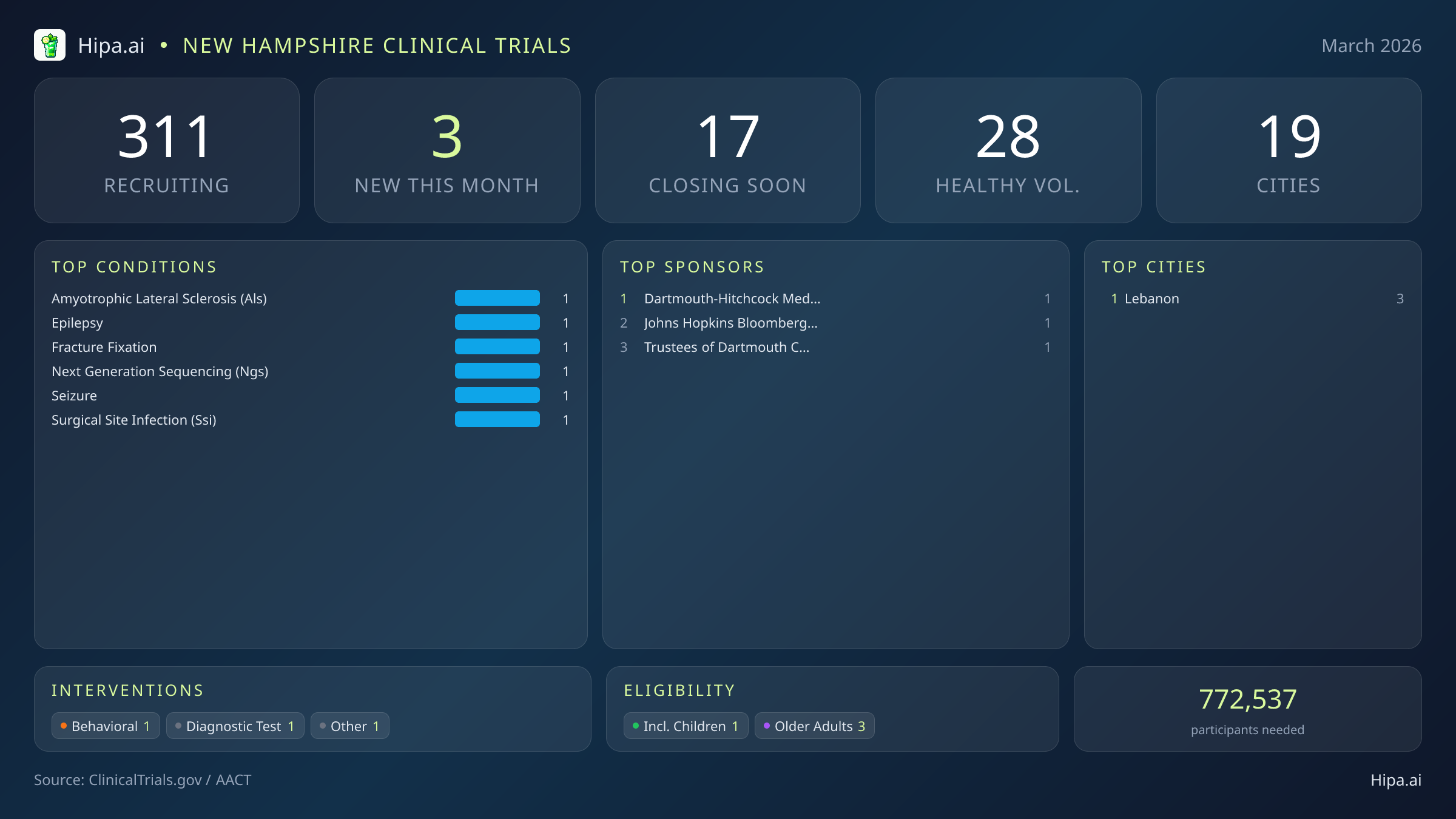
New Hampshire Clinical Trials Report — March 2026
3 New Studies, 17 Closing Soon
Hipa.ai Research - March 1, 2026 - Source: Research Data
Monthly Clinical Trials Infographic
Immediate Enrollment Deadlines for Oncology and Cardiac Studies
Time is running out for New Hampshire residents seeking access to several critical medical research initiatives. Over the next 90 days, 17 clinical trials will permanently close their enrollment windows across the state. Patients currently managing specific severe diagnoses face a rapidly narrowing window to access experimental therapies, behavioral interventions, and diagnostic tests. Final enrollment phases are approaching quickly for studies focused on advanced oncological and cardiovascular conditions, meaning those who wish to participate must act immediately before the research moves into the data analysis phase. Time-sensitive opportunities are closing for individuals managing the following conditions:
- Stage IV lung cancer
- Anatomic stage III breast cancer
- Hormone receptor-positive breast carcinoma
- Coronary artery disease and coronary stenosis
- Crohn disease
- Insomnia
This impending deadline also creates a distinct opportunity for individuals without underlying health conditions to participate in scientific discovery. Exactly 1 of these closing studies is actively seeking healthy volunteers, making immediate action necessary for those looking to contribute to baseline medical research before the spring season ends. For patients with advanced cancers, such as HER2-negative breast carcinoma and stage IV lung cancer, these closing trials represent some of the final local opportunities to access specific late-stage experimental protocols.
Academic Innovation Driving March Research Launches
March brings a highly targeted wave of academic research to the state, with 3 new trials opening this month. While this represents a decrease from the seven studies launched in January, the new initiatives are backed by major academic institutions and focus heavily on improving patient quality of life and post-surgical outcomes. Rather than traditional pharmaceutical interventions, this month's additions emphasize behavioral studies, diagnostic testing, and advanced care protocols designed to refine how existing treatments are delivered.
Leading academic centers, including Dartmouth-Hitchcock Medical Center and the Trustees of Dartmouth College, are driving local medical advancement. National partnerships are also expanding into the region, bringing high-profile Phase 3 research to local facilities. Several notable trials and targeted condition studies are currently seeking participants:
- The Trustees of Dartmouth College are recruiting 800 participants for a behavioral study (NCT07454733) investigating whether video recordings of multidisciplinary clinics can improve the quality of life for people diagnosed with amyotrophic lateral sclerosis (ALS) and their caregivers. Because ALS is a complex neurodegenerative disease requiring input from multiple specialists during lengthy clinic visits, this research aims to determine if providing patients and caregivers with video recordings of their sessions helps them better retain critical care information and manage the disease at home.
- Johns Hopkins Bloomberg School of Public Health has launched a Phase 3 diagnostic trial (NCT07458230) enrolling 250 participants. The study utilizes next-generation sequencing to rapidly identify and treat surgical site infections following high-energy bone fracture fixation. Because severe fractures carry a high risk of complex infections that can delay healing, this trial seeks to prove that advanced genomic sequencing can identify pathogens faster and more accurately than traditional cultures, leading to better targeted antibiotic therapy.
- Dartmouth-Hitchcock Medical Center is sponsoring newly launched research focusing on patients managing epilepsy and seizure disorders, aiming to refine diagnostic accuracy and improve long-term neurological disease management for conditions that severely impact daily functioning.
Lebanon Emerges as a Central Research Hub
New Hampshire's clinical research infrastructure remains highly accessible, with 311 active trials currently recruiting across 19 cities and 109 individual research sites. Geographically, this month's new research opportunities are exclusively clustered in Lebanon. As the home to major academic and medical institutions, Lebanon provides a dense concentration of advanced care options and specialized diagnostic equipment necessary for these complex studies. The city's robust medical infrastructure allows researchers to conduct large-scale behavioral and diagnostic trials that require multidisciplinary teams and advanced technology.
While Lebanon serves as the focal point for March's newly launched trials, residents living outside this immediate area still have extensive opportunities to participate in ongoing research. The statewide distribution of active trials ensures that patients in various regions can access experimental treatments and behavioral interventions without requiring extensive travel to major metropolitan centers. Community hospitals and specialized clinics throughout the state continue to host a wide array of ongoing studies, ensuring that rural and suburban populations remain connected to cutting-edge medical advancements.
Broad Demographic Inclusion and Volunteer Access
Eligibility criteria for this month's new studies are designed to accommodate a wide range of age demographics, ensuring that both young and aging populations can participate in medical advancement. Older adults have extensive options, with all three of the newly launched trials specifically designing their protocols to include senior participants. This is particularly relevant for the ALS and fracture fixation studies, as these conditions frequently impact older demographics who require specialized care protocols. Pediatric research is also represented, with one new study open to children, ensuring that younger patients managing neurological conditions like epilepsy have access to emerging diagnostic tools. None of the new trials launched this month restrict participation exclusively to male or female patients, ensuring broad access across all genders.
While none of the newly launched trials for March are currently accepting healthy volunteers, opportunities for those without specific medical diagnoses remain robust across the state. There are currently 28 total open, recruiting trials for healthy participants statewide. These ongoing trials rely heavily on healthy individuals to establish crucial baseline data for future medical breakthroughs, making community participation essential for scientific progress. Healthy volunteers in New Hampshire can still find numerous opportunities to participate in vaccine trials, observational studies, and preventive health research at various sites across the state.
As the region transitions into the spring research season, New Hampshire's clinical trial landscape is positioned to expand its focus on neurodegenerative care optimization and advanced genomic diagnostics, setting a foundation for more personalized approaches to chronic disease management and post-surgical recovery.
Data Highlights
Conditions Closing Soon
- stage iv lung cancer ajcc v8 (2)
- breast cancer (2)
- anatomic stage iii breast cancer ajcc v8 (1)
- hormone receptor-positive breast carcinoma (1)
- coronary artery disease (1)
- coronary stenosis (1)
- her2-negative breast carcinoma (1)
- crohn disease (1)
Most Common New Trial Conditions
- amyotrophic lateral sclerosis (als) (1)
- epilepsy (1)
- fracture fixation (1)
- next generation sequencing (ngs) (1)
- seizure (1)
- surgical site infection (ssi) (1)
Cities With the Most New Trials
- Lebanon (3)
Leading Sponsors
- Dartmouth-Hitchcock Medical Center (1)
- Johns Hopkins Bloomberg School of Public Health (1)
- Trustees of Dartmouth College (1)
| Month | New Trials | Closing Soon |
|---|---|---|
| November 2025 | 3 | 0 |
| December 2025 | 2 | 4 |
| January 2026 | 2 | 1 |
| February 2026 | 7 | 2 |
| March 2026 | 3 | 3 |
| April 2026 | 1 | 0 |
New Studies This Month (3)
| NCT ID | Title | Phase | Enrollment | Sponsor | Condition | City |
|---|---|---|---|---|---|---|
| NCT07451093 | Epileptiform Potential of Fully Immersive Virtual Reality | NA | 40 | Dartmouth-Hitchcock Medical Center | Epilepsy | Lebanon |
| NCT07454733 | Do Video Recordings of Multidisciplinary Clinics Improve Quality of Life for People With ALS and Their Caregivers? | NA | 800 | Trustees of Dartmouth College | Amyotrophic Lateral Sclerosis (ALS) | Lebanon |
| NCT07458230 | Efficacy of Integrating Next Generation Sequencing for Treatment of Surgical Site Infection After Fracture Fixation: | Phase 3 | 250 | Johns Hopkins Bloomberg School of Public Health | Next Generation Sequencing (NGS) | Lebanon |