New Jersey Clinical Trials Report — March 2026
33 New Studies, 94 Closing Soon
Hipa.ai Research - March 1, 2026 - Source: Research Data
Monthly Clinical Trials Infographic
New Jersey residents have a limited window to participate in several critical medical research studies before they stop accepting new patients. Within the next ninety days, 94 clinical trials across the state will permanently close their enrollment phases. This impending deadline affects both individuals managing specific diagnoses and those without underlying conditions, as 15 of these soon-to-close studies are actively seeking healthy volunteers to establish baseline data. Patients currently navigating oncology and mental health diagnoses face particularly urgent timelines to explore these experimental therapies before the opportunity passes. Final enrollment phases are rapidly approaching for studies focused on the following conditions:
- Breast cancer
- Pancreatic ductal adenocarcinoma
- Ovarian cancer
- Stage IV lung cancer
- Major depressive disorder
- Prostate cancer
- Cervical and endometrial cancers
- Coronary artery disease
Major Phase 3 Launches and High-Capacity Studies
Despite the upcoming closures, March brings a fresh wave of large-scale research projects to the state, with thirty-three new trials opening their doors. This matches the steady pace of research initiation seen at the start of the year, keeping New Jersey at the forefront of pharmaceutical and medical device development. Major industry sponsors are launching high-capacity studies that require thousands of participants, focusing heavily on infectious disease prevention, women's health, and advanced oncology treatments. These large-scale trials offer residents a direct role in advancing global healthcare standards.
- Pfizer is recruiting over one thousand seven hundred participants for a Phase 3 trial (NCT07500506) evaluating the safety and immunogenicity of a fifth dose of the VLA15 Lyme disease vaccine in healthy individuals aged seven and older.
- Novartis Pharmaceuticals has launched a massive observational study (NCT07450157) seeking more than six thousand four hundred prostate cancer patients to evaluate treatment patterns and overall survival following 177Lu-PSMA-617 therapy.
- Bayer is enrolling one thousand five hundred women for an observational study (NCT07490431) tracking the real-world effectiveness of Elinzanetant, a hormone-free medicine designed to block menopause symptoms like hot flashes and sleep disturbances.
- Novartis Pharmaceuticals is also conducting a real-world study (NCT07477756) with over one thousand two hundred participants to assess clinical outcomes in metastatic castration-resistant prostate cancer patients treated with lutetium-177 vipivotide tetraxetan.
- AI-Spectral is conducting a diagnostic study (NCT07446582) with one thousand adults over forty to determine if its DeepMSI artificial intelligence can detect age-related macular degeneration biomarkers as accurately as experienced clinicians.
- Axsome Therapeutics is running a Phase 3 trial (NCT07484217) requiring over five hundred participants to assess Solriamfetol as a treatment for major depressive disorder accompanied by excessive daytime sleepiness.
- Yale University is initiating a prehospital field study (NCT07475728) involving nearly five hundred participants to evaluate the feasibility and safety of EMS-initiated buprenorphine administration.
- Boehringer Ingelheim is seeking over four hundred adults for a Phase 3 trial (NCT07497087) testing whether Nerandomilast can improve symptoms and slow disease progression in patients with systemic sclerosis.
Geographic Distribution and Clinical Focus
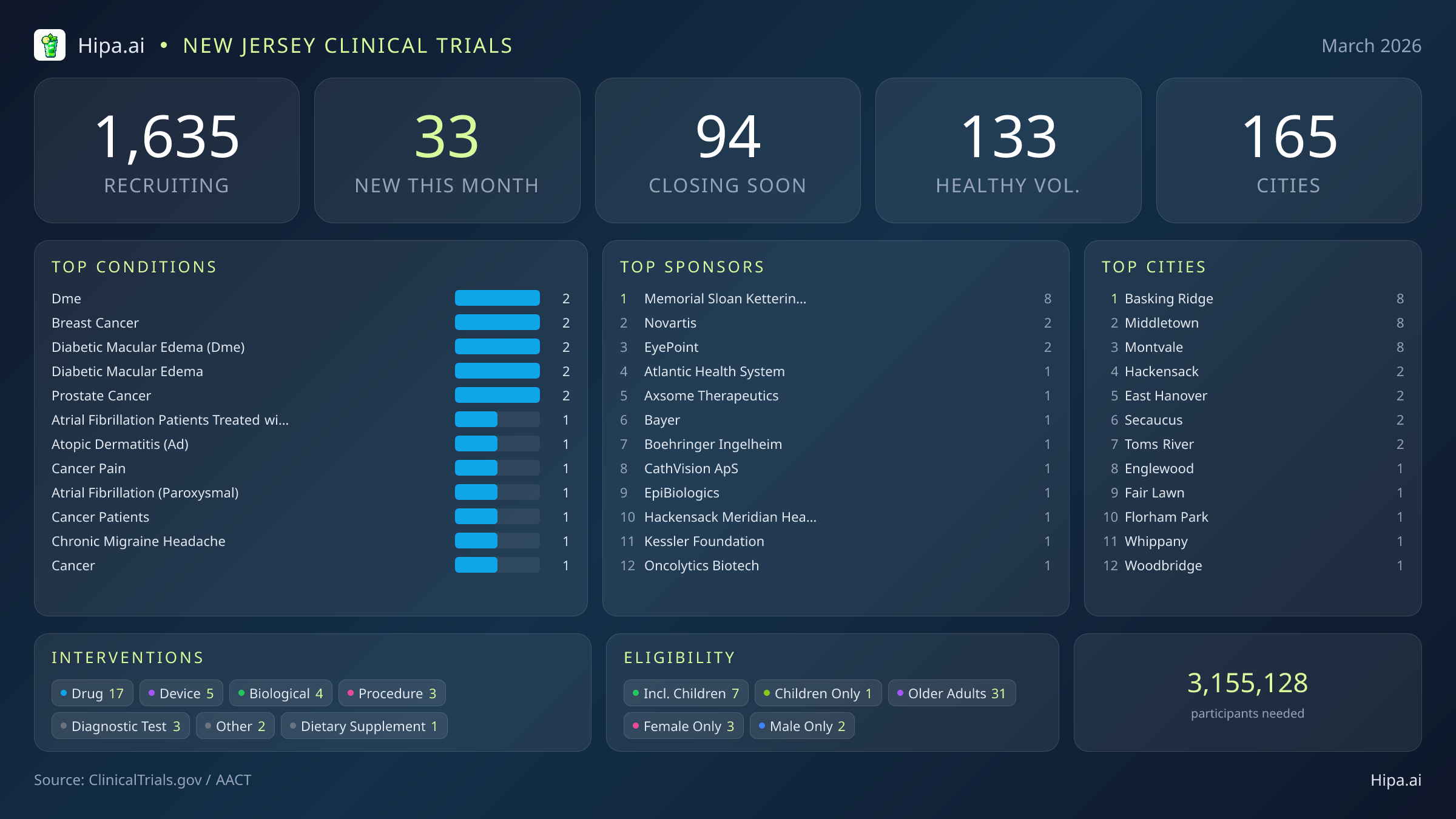
The clinical research infrastructure across New Jersey remains highly accessible, with 1,635 active trials currently recruiting across one hundred sixty-five cities and over one thousand individual research sites. Pharmaceutical interventions dominate this month's new research landscape, accounting for 17 of the newly launched studies. Medical device evaluations follow with five new trials, while biological testing, procedural research, and diagnostic tests make up the remainder of the new clinical portfolio. A single dietary supplement trial has also begun recruiting.
Geographically, new research opportunities are heavily clustered in several key municipalities rather than being isolated to a single metropolitan hub. Basking Ridge, Middletown, and Montvale lead the state this month, each hosting eight newly opened trials. This distribution ensures that suburban residents have direct access to advanced medical research without needing to commute into major urban centers. Residents living outside these primary clusters still have access to cutting-edge medical research, as new trials have also opened their doors in the following cities:
- Hackensack
- East Hanover
- Secaucus
- Toms River
- Englewood
- Fair Lawn
- Florham Park
March's new clinical trials target a diverse array of chronic and acute conditions. Researchers are actively seeking patients recently diagnosed with or currently managing diabetic macular edema, breast cancer, prostate cancer, atopic dermatitis, and cancer-related pain. Additional studies are focusing on patients with paroxysmal atrial fibrillation, specifically those treated with the Farapulse PFA system.
Participant Eligibility and Industry Sponsorship
Eligibility criteria for these new studies ensure that diverse demographic groups can participate in scientific advancement. Older adults have extensive options, with 31 of the new trials specifically designing their protocols to include senior populations. Pediatric research is also active, as seven new studies are open to children and adolescents. Highly targeted demographic research includes three trials exclusively for females and two exclusively for males.
For those without a specific medical diagnosis, six of this month's new trials are actively welcoming healthy volunteers. This brings the total number of open, recruiting trials for healthy participants across New Jersey to 133. Healthy volunteers play a crucial role in early-phase safety testing and provide necessary comparative data for diagnostic studies.
Industry sponsors are driving the majority of this month's new research, funding twenty of the newly launched trials. Academic and institutional organizations account for the remaining thirteen. Memorial Sloan Kettering Cancer Center is particularly active in the region, sponsoring eight new protocols focused on advancing cancer care. Other notable sponsors launching trials this month include EyePoint Pharmaceuticals, Atlantic Health System, and CathVision ApS.
Moving into the spring, New Jersey's clinical research ecosystem is positioned to rapidly expand its focus on artificial intelligence diagnostics and advanced radioligand therapies for oncology patients.
Data Highlights
Conditions Closing Soon
- breast cancer (3)
- pancreatic ductal adenocarcinoma (3)
- ovarian cancer (3)
- stage iv lung cancer ajcc v8 (2)
- major depressive disorder (2)
- prostate cancer (2)
- healthy (2)
- cervical cancer (2)
Most Common New Trial Conditions
- dme (2)
- breast cancer (2)
- diabetic macular edema (dme) (2)
- diabetic macular edema (2)
- prostate cancer (2)
- atrial fibrillation patients treated with the farapulse™ pfa system (1)
- atopic dermatitis (ad) (1)
- cancer pain (1)
Cities With the Most New Trials
- Basking Ridge (8)
- Middletown (8)
- Montvale (8)
- Hackensack (2)
- East Hanover (2)
- Secaucus (2)
- Toms River (2)
- Englewood (1)
Leading Sponsors
- Memorial Sloan Kettering Cancer Center (8)
- Novartis Pharmaceuticals (2)
- EyePoint Pharmaceuticals, Inc. (2)
- Atlantic Health System (1)
- Axsome Therapeutics, Inc. (1)
- Bayer (1)
- Boehringer Ingelheim (1)
- CathVision ApS (1)
| Month | New Trials | Closing Soon |
|---|---|---|
| November 2025 | 19 | 6 |
| December 2025 | 41 | 35 |
| January 2026 | 33 | 12 |
| February 2026 | 29 | 9 |
| March 2026 | 33 | 31 |
| April 2026 | 7 | 0 |