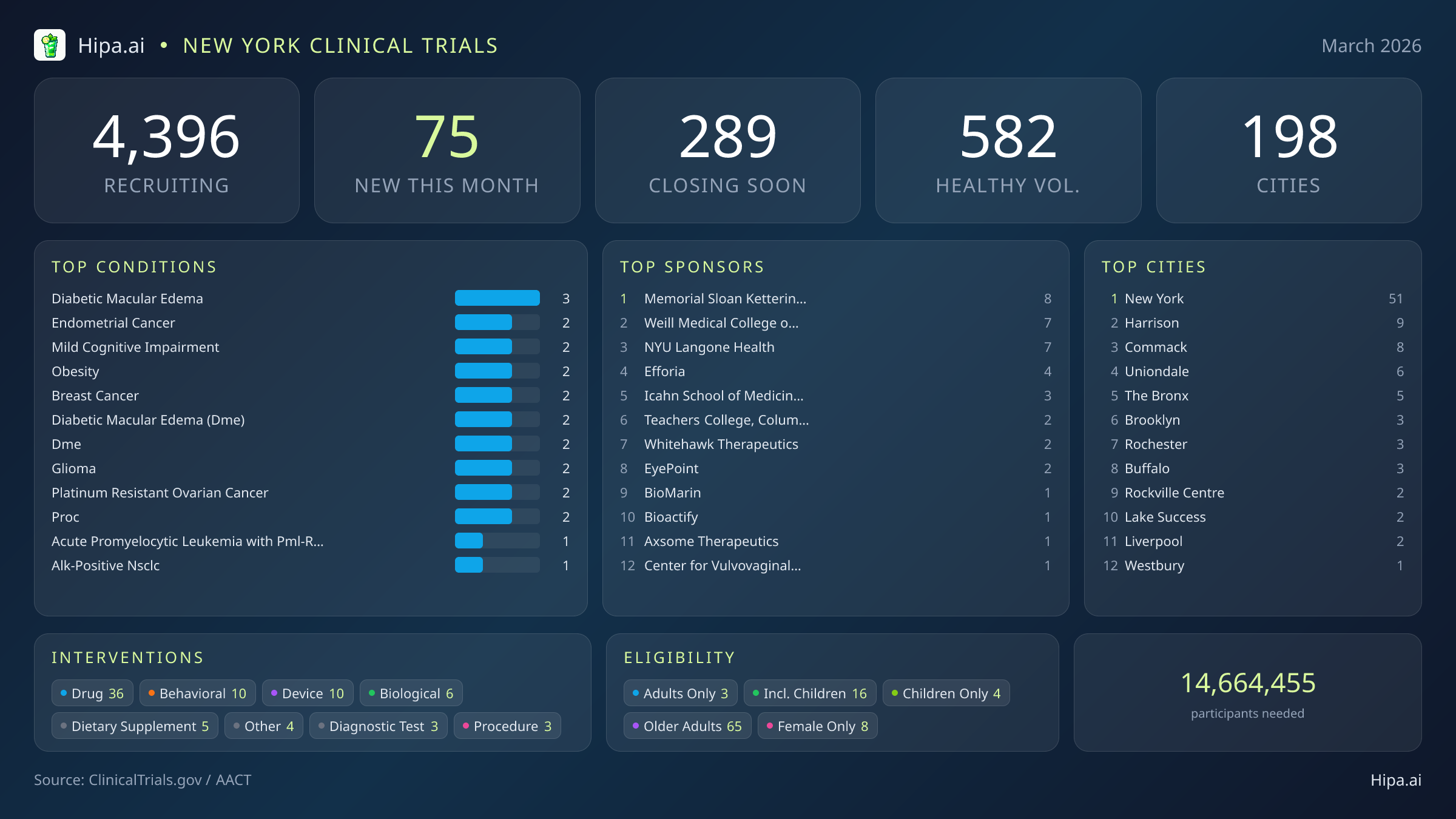
New York Clinical Trials Report — March 2026
75 New Studies, 289 Closing Soon
Hipa.ai Research - March 1, 2026 - Source: Research Data
Monthly Clinical Trials Infographic
New Yorkers have a limited window to enroll in hundreds of medical research studies before they permanently close their doors. Within the next ninety days, 289 clinical trials will conclude their enrollment phases across the state. For residents without underlying medical conditions, this creates an immediate deadline to participate in scientific advancement, as 55 of these soon-to-close studies are actively seeking healthy volunteers. Patients managing specific diagnoses also face urgent timelines. Oncology and neurological research are particularly affected by this wave of closures. Final enrollment phases are rapidly approaching for studies focused on the following conditions:
- Breast cancer
- Prostate cancer
- Post-traumatic stress disorder
- Major depressive disorder
- COVID-19
- Stroke
- BRCA1 and BRCA2 mutations
- Multiple myeloma
- Ovarian cancer
Major Phase 3 and High-Capacity Research Launches
Despite the impending closures, March brings a robust wave of large-scale research projects to the state. While the pace of new study launches has cooled slightly since the beginning of the year—dropping from ninety-two new trials in January to 75 this month—the scale of these new initiatives is massive. NYU Langone Health is currently recruiting thousands of adults for a comprehensive study evaluating the long-term impacts of COVID-19. Pfizer is also launching a major Phase 3 clinical trial to test the safety and efficacy of a fifth dose of a Lyme disease vaccine. Beyond infectious diseases, several major healthcare organizations and academic institutions are launching high-capacity studies that require thousands of participants across New York.
- NYU Langone Health is seeking participants for a massive observational study (NCT07498504) to understand the long-term physical, cognitive, and functional impacts of COVID-19 in adults, aiming to enroll over five thousand individuals.
- Weill Medical College of Cornell University is enrolling nearly two thousand participants for the CARES4You study (NCT07501689), testing a middle school science curriculum designed to address cancer health disparities and teach students about cancer risk and prevention.
- Pfizer has launched a Phase 3 trial (NCT07500506) seeking over seventeen hundred healthy volunteers aged seven and older to evaluate the safety and immunogenicity of a fifth dose of the VLA15 Lyme disease vaccine.
- Weill Medical College of Cornell University is also conducting a surgical study (NCT07485738) with over twelve hundred participants to evaluate how Transit-Time Flow Measurement during coronary artery bypass graft surgery affects patient recovery.
- Sun Pharmaceutical Industries, Inc. is recruiting over one thousand participants for a Phase 1 trial (NCT07469228) comparing the efficacy and safety of two tretinoin gel microsphere treatments for acne vulgaris.
- Meyer Children's Hospital IRCCS is seeking one thousand participants for a study (NCT07454343) focused on Erdheim-Chester Disease, a rare form of non-Langerhans cell histiocytosis.
- Stony Brook University is enrolling eight hundred participants in the First Responder Safety Training program (NCT07478250) to evaluate resilience training for reducing post-traumatic stress disorder symptoms.
- Imperative Care, Inc. is testing a continuous dual aspiration technique for stroke treatment (NCT07491952), requiring seven hundred fifty participants.
Geographic Spread and Targeted Diagnoses
The clinical research infrastructure across the state remains highly accessible, with 4,396 active trials currently recruiting across one hundred ninety-eight New York cities and nearly three thousand individual research sites. Geographically, new research opportunities are heavily clustered in the New York City metropolitan area. Manhattan and the surrounding boroughs lead the state, with fifty-one newly opened trials in New York City alone, followed by five in The Bronx and three in Brooklyn. Residents living outside the immediate five boroughs or in upstate regions still have access to cutting-edge medical research. New trials have opened their doors this month in several other key municipalities:
- Harrison
- Commack
- Uniondale
- Rochester
- Buffalo
- Rockville Centre
- Lake Success
March's new clinical trials are targeting a wide array of chronic and acute conditions. Researchers are actively seeking patients recently diagnosed with or currently managing diabetic macular edema, endometrial cancer, mild cognitive impairment, obesity, breast cancer, glioma, and platinum-resistant ovarian cancer. The variety of interventions being tested remains diverse. Pharmaceutical interventions dominate the new landscape with 36 drug trials, followed by behavioral studies, medical device evaluations, biological testing, and dietary supplement research.
Eligibility and Institutional Sponsorship
Eligibility criteria for these new studies are notably broad, ensuring that diverse demographic groups can participate in medical advancement. Older adults have extensive options, with 65 of the new trials specifically designing their protocols to include senior populations. Pediatric research is also expanding, as sixteen new studies are open to children and adolescents. Highly targeted demographic research includes eight trials exclusively for females. For those without a specific medical diagnosis, twenty-one of this month's new trials are actively welcoming healthy volunteers. This brings the total number of open, recruiting trials for healthy participants across New York to 582.
Academic medical centers continue to serve as the primary engines for this clinical research. Memorial Sloan Kettering Cancer Center leads the state in new study launches this month, followed closely by Weill Medical College of Cornell University and NYU Langone Health. Other prominent institutions driving new protocols include the Icahn School of Medicine at Mount Sinai and Teachers College at Columbia University. Industry sponsors are also highly active, with companies like Efforia, Inc., Whitehawk Therapeutics, Inc., and EyePoint Pharmaceuticals, Inc. funding dozens of new therapeutic investigations. Overall, the sponsorship of these trials leans slightly toward academic and institutional organizations, which are driving forty-one of the new studies, while industry partners are funding thirty-four.
Moving into the spring, the state's clinical research ecosystem is positioned to shift focus toward analyzing the massive datasets generated by this season's high-capacity infectious disease and behavioral trials, while preparing for a new wave of targeted oncological and neurological interventions.
Data Highlights
Conditions Closing Soon
- breast cancer (10)
- prostate cancer (7)
- post traumatic stress disorder (5)
- major depressive disorder (5)
- covid-19 (4)
- stroke (3)
- brca2 mutation (3)
- brca1 mutation (3)
Most Common New Trial Conditions
- diabetic macular edema (3)
- endometrial cancer (2)
- mild cognitive impairment (2)
- obesity (2)
- breast cancer (2)
- diabetic macular edema (dme) (2)
- dme (2)
- glioma (2)
Cities With the Most New Trials
Leading Sponsors
- Memorial Sloan Kettering Cancer Center (8)
- Weill Medical College of Cornell University (7)
- NYU Langone Health (7)
- Efforia, Inc (4)
- Icahn School of Medicine at Mount Sinai (3)
- Teachers College, Columbia University (2)
- Whitehawk Therapeutics, Inc. (2)
- EyePoint Pharmaceuticals, Inc. (2)
| Month | New Trials | Closing Soon |
|---|---|---|
| November 2025 | 65 | 23 |
| December 2025 | 144 | 91 |
| January 2026 | 66 | 33 |
| February 2026 | 90 | 39 |
| March 2026 | 75 | 105 |
| April 2026 | 18 | 0 |