North Carolina Clinical Trials Report — March 2026
41 New Studies, 176 Closing Soon
Hipa.ai Research - March 1, 2026 - Source: Research Data
Monthly Clinical Trials Infographic
Approaching Deadlines for North Carolina Research Initiatives
Time is running out for North Carolina residents to participate in several major medical studies. Within the next ninety days, 176 clinical trials will permanently close their enrollment windows across the state. This creates an immediate deadline for individuals seeking access to experimental therapies and observational studies. For those without underlying medical conditions, the window is also rapidly closing on 28 specific trials actively seeking healthy volunteers. Patients currently managing chronic or acute diagnoses face urgent timelines to join research programs focused on the following areas:
- Breast cancer and metastatic breast cancer
- Multiple myeloma
- Prostate cancer
- Pediatric obesity
- Clinical depression
- Mild cognitive impairment
- Sleep disorders
- Dry eye disease
- Focal segmental glomerulosclerosis
Major Phase 3 Launches and High-Capacity Diagnostics
Despite the wave of impending closures, March brings a fresh influx of high-profile research opportunities. Major pharmaceutical sponsors and academic institutions are launching large-scale diagnostic and therapeutic trials requiring thousands of participants. Novo Nordisk A/S is initiating two major Phase 3 trials targeting excess body weight, while companies like Hoffmann-La Roche and Harbinger Health are rolling out massive diagnostic studies for traumatic brain injury and colorectal cancer.
- Hoffmann-La Roche is seeking two thousand participants to develop a novel blood-based biomarker test (NCT07455136) aimed at diagnosing traumatic brain injuries and monitoring secondary events in adult patients.
- Harbinger Health has launched the CLEAR Study (NCT07479381), enrolling one thousand four hundred fifty individuals to evaluate a new blood-based screening test for colorectal cancer.
- Wake Forest University Health Sciences is recruiting one thousand two hundred fifty participants to study how different warning labels and icons on cannabis edibles packaging impact consumer knowledge and harm perception (NCT07496489).
- The University of North Carolina, Chapel Hill is conducting a randomized behavioral trial (NCT07458607) with one thousand one hundred eighty-5 adults to evaluate how a simulated menthol cigarette ban affects tobacco purchasing habits.
- Novo Nordisk A/S is running the Phase 3 AMAZE 12 study (NCT07503210), seeking six hundred participants to test the safety and efficacy of the weekly injectable medication NNC0487-0111 for maintaining weight loss.
- A parallel Phase 3 study by Novo Nordisk A/S (NCT07481630) will enroll four hundred individuals to evaluate the same medication's ability to reduce weight and alleviate pain in patients with knee osteoarthritis.
- Bristol-Myers Squibb is recruiting two hundred sixty participants for a Phase 2 trial (NCT07492680) evaluating BMS-986504, both as a monotherapy and in combination with other agents, for advanced solid tumors with MTAP deletion.
- Whitehawk Therapeutics, Inc. has opened a Phase 1 dose-escalation study (NCT07470853) testing a targeted antibody-drug conjugate in two hundred sixty-five adults with advanced or metastatic solid tumors.
Geographic Distribution and Intervention Landscape
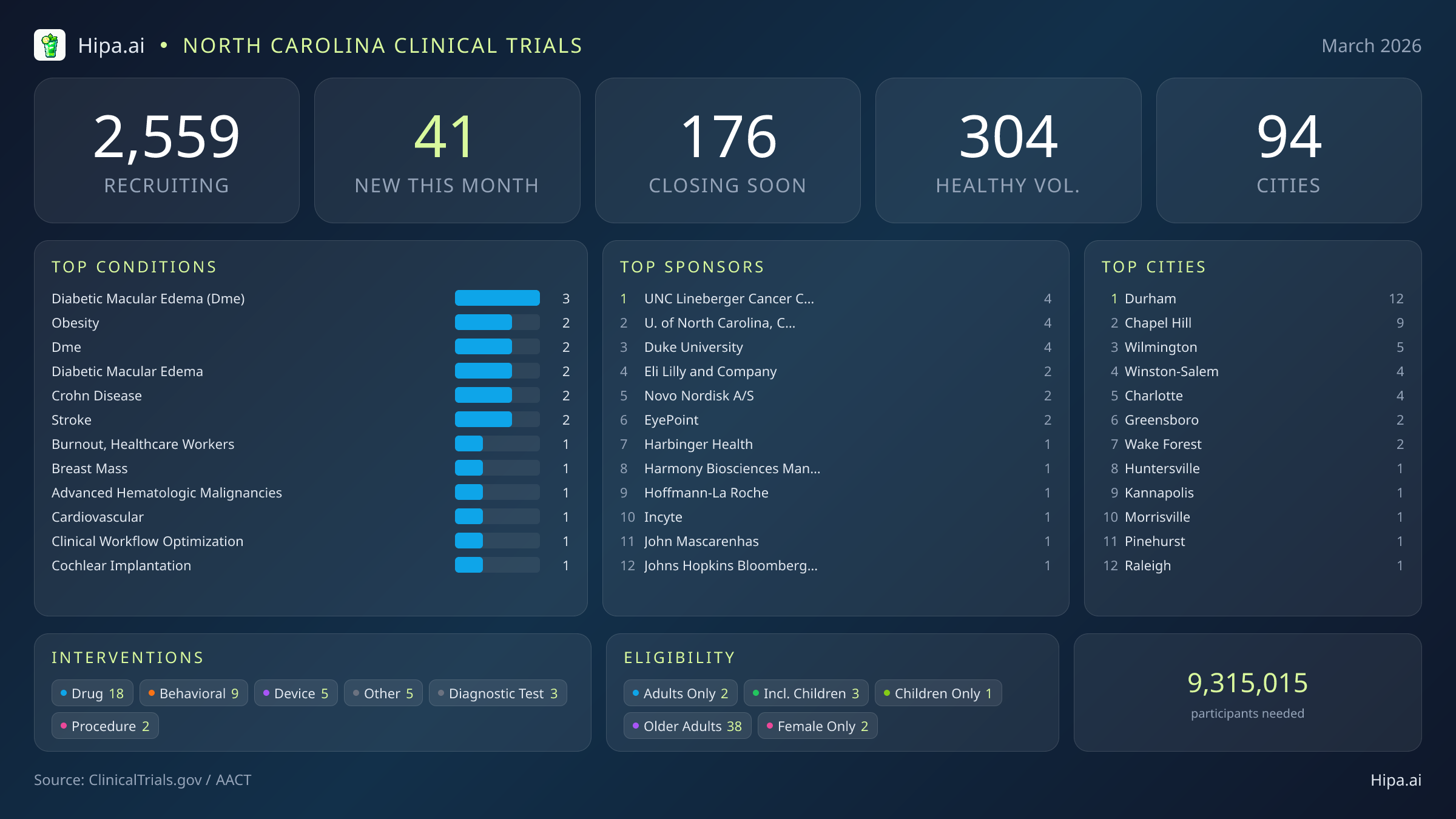
The clinical research infrastructure in North Carolina remains highly accessible, with 2,559 active trials currently recruiting across ninety-four cities and over one thousand five hundred individual research sites. The pace of new study launches has stabilized, with 41 new trials opening this month, representing a slight decrease from the forty-five studies initiated in January. The intervention landscape for these new studies is heavily weighted toward pharmaceutical development, featuring 18 new drug trials. The remaining new protocols are distributed across behavioral interventions, medical device evaluations, diagnostic tests, and clinical procedures.
Geographically, the state's research footprint is anchored by the Research Triangle, though opportunities extend well beyond this central hub. Durham leads the state with twelve newly opened trials, closely followed by Chapel Hill with nine. Residents seeking to participate in medical research can also find new studies launching this month in several other municipalities:
- Wilmington
- Winston-Salem
- Charlotte
- Greensboro
- Wake Forest
- Huntersville
- Kannapolis
- Morrisville
Targeted Conditions and Broad Eligibility
March's newly launched trials address a diverse spectrum of medical conditions. Researchers are actively recruiting patients diagnosed with various stages of diabetic macular edema, Crohn's disease, stroke, and cardiovascular disease. Specialized studies are also focusing on advanced hematologic malignancies, breast masses, and occupational burnout among healthcare workers. The sponsorship driving these initiatives is evenly balanced between commercial and academic entities. Industry partners like Eli Lilly and Company, Novo Nordisk A/S, Harmony Biosciences Management, Inc., and EyePoint Pharmaceuticals, Inc. are sponsoring twenty of the new trials. Academic institutions—led by the UNC Lineberger Comprehensive Cancer Center, Duke University, and the University of North Carolina, Chapel Hill—are spearheading twenty-one.
Trial designers are prioritizing broad accessibility, ensuring that a wide demographic cross-section can participate in these scientific advancements. Older adults have extensive access to the latest research, with 38 of the new trials specifically structuring their protocols to include senior populations. Pediatric research continues to move forward, with three new studies open to children and adolescents. Highly specific demographic research includes two trials exclusively enrolling female participants.
Opportunities for individuals without underlying health conditions remain a vital component of the state's research ecosystem. Five of the newly launched trials are actively welcoming healthy volunteers. This recent addition brings the total number of open, recruiting trials available to healthy participants across North Carolina to 304, providing ample avenues for residents to contribute to baseline medical science and diagnostic development.
As the spring research calendar progresses, the state's clinical infrastructure is preparing to transition from the aggressive enrollment phases of early-year behavioral studies toward the long-term monitoring of massive diagnostic cohorts, setting the stage for a new wave of targeted oncological and metabolic interventions in the coming months.
Data Highlights
Conditions Closing Soon
- breast cancer (4)
- sleep (3)
- multiple myeloma (3)
- pediatric obesity (3)
- depression (3)
- mild cognitive impairment (2)
- metastatic breast cancer (2)
- prostate cancer (2)
Most Common New Trial Conditions
- diabetic macular edema (dme) (3)
- obesity (2)
- dme (2)
- diabetic macular edema (2)
- crohn disease (2)
- stroke (2)
- burnout, healthcare workers (1)
- breast mass (1)
Cities With the Most New Trials
- Durham (12)
- Chapel Hill (9)
- Wilmington (5)
- Winston-Salem (4)
- Charlotte (4)
- Greensboro (2)
- Wake Forest (2)
- Huntersville (1)
Leading Sponsors
- UNC Lineberger Comprehensive Cancer Center (4)
- University of North Carolina, Chapel Hill (4)
- Duke University (4)
- Eli Lilly and Company (2)
- Novo Nordisk A/S (2)
- EyePoint Pharmaceuticals, Inc. (2)
- Harbinger Health (1)
- Harmony Biosciences Management, Inc. (1)
| Month | New Trials | Closing Soon |
|---|---|---|
| November 2025 | 44 | 9 |
| December 2025 | 64 | 34 |
| January 2026 | 46 | 10 |
| February 2026 | 44 | 20 |
| March 2026 | 41 | 46 |
| April 2026 | 9 | 0 |