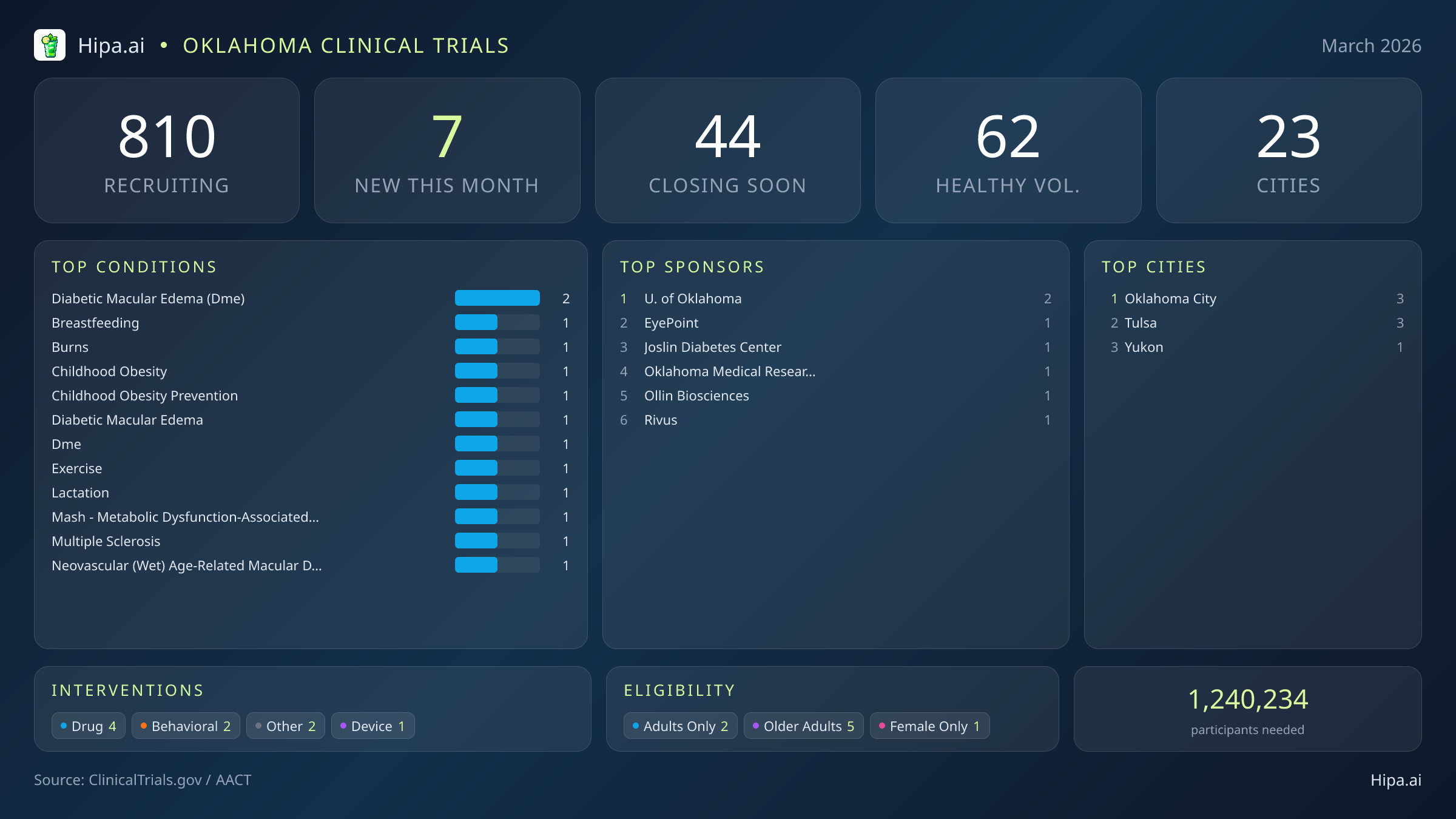
Oklahoma Clinical Trials Report — March 2026
7 New Studies, 44 Closing Soon
Hipa.ai Research - March 1, 2026 - Source: Research Data
Monthly Clinical Trials Infographic
Approaching Deadlines for Oklahoma Research Participants
Time is strictly limited for Oklahomans looking to join several critical medical studies. Within the next ninety days, 44 clinical trials across the state will permanently close their enrollment windows. This shrinking timeline creates an urgent need for participants, particularly those managing specific chronic or severe conditions who might benefit from experimental therapies before these studies stop accepting new patients. For individuals without underlying health issues, the window is even tighter. Only 3 of these soon-to-close studies are still actively seeking healthy volunteers, making immediate action necessary for those looking to contribute to baseline medical research.
Patients currently seeking alternative interventions face rapidly approaching final enrollment phases. The studies preparing to close span a wide spectrum of medical disciplines, from oncology and cardiology to mental health. Researchers are finalizing their participant cohorts for studies focused on the following conditions:
- Major depressive disorder
- Coronary artery disease
- Recurrent endometrial carcinoma
- Stage IV lung cancer
- Acute myeloid leukemia
- Anorexia nervosa
- Acute kidney injury
- Antiphospholipid syndrome
Late-Stage Pharmaceutical Innovations and Academic Studies
While the overall pace of new study launches in the state has cooled steadily over the past few months—dropping from nineteen new trials in December to nine in January, and settling at 7 this month—March brings highly credible, late-stage research to the state. The landscape of new studies is balanced between academic institutions and industry leaders, with four trials categorized as drug interventions, two focusing on behavioral approaches, and one testing a new medical device. Major pharmaceutical sponsors are offering local patients access to advanced experimental treatments before they reach the broader commercial market.
Notable therapeutic trials currently seeking participants include a major effort by EyePoint Pharmaceuticals. They are recruiting two hundred forty participants for a Phase 3 trial (NCT07449936) comparing the efficacy of EYP-1901 against aflibercept for patients managing diabetic macular edema. Because this is a Phase 3 randomized, double-masked study, it represents one of the final stages of clinical testing required before potential regulatory approval. Additionally, Rivus Pharmaceuticals is enrolling one hundred eighty participants in a Phase 2 study (NCT07491458) to assess the safety, tolerability, and liver fat reduction potential of HU6 as a treatment for metabolic dysfunction-associated steatohepatitis (MASH). This trial will closely monitor pharmacokinetics and structural liver changes in affected adults.
Beyond these major pharmaceutical efforts, academic powerhouses and specialized research centers like the University of Oklahoma, the Oklahoma Medical Research Foundation, Ollin Biosciences, Inc., and the Joslin Diabetes Center are driving forward new initiatives. This month's newly launched trials target a diverse array of health focuses, including:
- Diabetic macular edema
- Metabolic dysfunction-associated steatohepatitis (MASH)
- Severe burns and tissue recovery
- Lactation and breastfeeding mechanics
- Exercise protocols and physical behavioral therapies
Interestingly, while conditions like childhood obesity and childhood obesity prevention are targeted in this month's new research portfolio, the specific eligibility criteria for these newly opened phases currently exclude pediatric participants. Instead, these specific study phases are focusing on adult caregivers, family dynamics, or related adult demographics to address pediatric health outcomes indirectly.
Research Hubs and Statewide Accessibility
Despite the recent dip in new monthly trial launches, Oklahoma maintains a robust and highly accessible clinical research infrastructure. Currently, 810 active trials are recruiting across the state, operating out of 415 individual research sites spread across twenty-three different cities. This extensive network ensures that residents have multiple access points to cutting-edge medical research, whether they live in major metropolitan areas or smaller regional communities.
Geographically, the newest research opportunities are tightly clustered in the state's largest economic and medical hubs. The trials launched this month are distributed across the following municipalities:
- Oklahoma City
- Tulsa
- Yukon
Oklahoma City and Tulsa lead the state's research expansion, each hosting three of the newly opened trials. These cities naturally attract the bulk of Phase 2 and Phase 3 pharmaceutical studies due to their dense populations and large hospital networks. However, the addition of a new trial in Yukon highlights a continuing effort by sponsors to push research access slightly beyond the immediate urban cores. By opening sites in suburban and adjacent communities, researchers can provide residents with closer opportunities to participate in medical advancement without facing the logistical hurdles of commuting into downtown medical districts.
Demographics and Healthy Volunteer Enrollment
Eligibility criteria for this month's new studies lean heavily toward adult populations, ensuring that mature demographics have ample opportunity to engage with new therapies. Older adults have excellent access to new research, with 5 of the newly launched trials specifically designing their protocols to include senior demographics. The gender breakdown remains largely inclusive, ensuring equitable access to experimental care. None of the new trials restrict participation exclusively to men, and only 1 trial limits enrollment entirely to female participants—a restriction that aligns directly with the new studies focused on lactation and breastfeeding mechanics.
For Oklahomans without a specific medical diagnosis, the opportunity to contribute to scientific discovery remains steady. One of this month's new trials is actively welcoming healthy volunteers. This addition brings the total number of open, recruiting trials for healthy participants across Oklahoma to 62. These healthy volunteer cohorts are absolutely essential for the clinical trial ecosystem. By participating, healthy individuals provide the critical baseline data that allows researchers to accurately measure the safety, side effects, and true efficacy of new interventions against standard human baselines.
As the spring research cycle accelerates, Oklahoma's clinical trial ecosystem is positioned to expand its footprint in metabolic and ophthalmic therapies, potentially drawing larger investments from national pharmaceutical sponsors looking to leverage the state's highly concentrated urban research corridors and engaged patient populations.
Data Highlights
Conditions Closing Soon
- major depressive disorder (2)
- coronary artery disease (2)
- recurrent endometrial carcinoma (2)
- stage iv lung cancer ajcc v8 (2)
- advanced lung non-small cell carcinoma (1)
- ambiguous lineage leukemia or lymphoma (1)
- acute kidney injury (1)
- anorexia nervosa (1)
Most Common New Trial Conditions
- diabetic macular edema (dme) (2)
- breastfeeding (1)
- burns (1)
- childhood obesity (1)
- childhood obesity prevention (1)
- diabetic macular edema (1)
- dme (1)
- exercise (1)
Cities With the Most New Trials
- Oklahoma City (3)
- Tulsa (3)
- Yukon (1)
Leading Sponsors
- University of Oklahoma (2)
- EyePoint Pharmaceuticals, Inc. (1)
- Joslin Diabetes Center (1)
- Oklahoma Medical Research Foundation (1)
- Ollin Biosciences, Inc. (1)
- Rivus Pharmaceuticals, Inc. (1)
| Month | New Trials | Closing Soon |
|---|---|---|
| October 2025 | 19 | 2 |
| November 2025 | 11 | 4 |
| December 2025 | 19 | 6 |
| January 2026 | 10 | 4 |
| February 2026 | 8 | 6 |
| March 2026 | 7 | 12 |