Oregon Clinical Trials Report — March 2026
9 New Studies, 55 Closing Soon
Hipa.ai Research - March 1, 2026 - Source: Research Data
Monthly Clinical Trials Infographic
Approaching Enrollment Deadlines Across Oregon
Time is running out for Oregonians seeking access to several advanced medical research programs. Within the next ninety days, 55 clinical trials will permanently close their enrollment windows across the state. This impending deadline creates a critical window for patients managing complex diagnoses to access experimental therapies before they are no longer available to new participants. People without underlying health conditions also have a brief opportunity to contribute to medical science, as 8 of these closing studies are actively seeking healthy volunteers to establish baseline data. Final enrollment phases are rapidly approaching for research focused on a diverse array of severe and chronic conditions:
- Prostate cancer and multiple myeloma
- Stage IV lung cancer and anatomic stage III breast cancer
- Acute leukemia and acute undifferentiated leukemia
- Major depression and severe anxiety
- Glaucoma and related vision disorders
- Adolescent behavior and emotional regulation
Patients currently receiving standard care for these specific cancers or psychological conditions should consult with their healthcare providers immediately if they wish to explore these soon-to-close experimental avenues.
Late-Stage Drug Trials and Major Interventions
March brings a highly targeted wave of new research to the state, heavily driven by pharmaceutical and institutional innovation. While the pace of new study launches has slowed from seventeen in January to 9 new trials this month, the caliber of the research remains exceptional. Industry sponsors are backing the majority of these new initiatives, offering local patients access to late-stage experimental treatments before they reach the broader commercial market. Several major therapeutic and behavioral trials are currently seeking participants:
- Kaiser Permanente is recruiting eight hundred participants for a massive behavioral study (NCT07459621) evaluating a novel mobile health intervention. This randomized controlled trial is designed to help people who are ambivalent about quitting smoking by comparing two different cessation programs, both of which utilize a specialized app and publicly available resources.
- Axsome Therapeutics has launched a Phase 3, double-blind, placebo-controlled trial (NCT07484217) testing solriamfetol. The multicenter study is actively seeking over five hundred patients diagnosed with major depressive disorder who also experience debilitating excessive daytime sleepiness.
- EyePoint Pharmaceuticals is conducting a Phase 3 randomized, double-masked study (NCT07449923) to compare the efficacy of EYP-1901 against aflibercept for patients managing diabetic macular edema, a condition that can lead to severe vision loss.
- The OHSU Knight Cancer Institute is seeking ninety volunteers for a Phase 2 trial (NCT07498517) evaluating whether a single dose of gruticibart can prevent catheter-related thrombosis. This study aims to reduce the incidence of painful blood clots and swelling in patients who require central venous catheters for their medical treatments.
Targeted Conditions and Treatment Approaches
The clinical focus for March centers heavily on metabolic, cardiovascular, and vision-related conditions. Researchers are actively seeking patients diagnosed with diabetic macular edema, with multiple new studies exploring advanced interventions to preserve eyesight in diabetic populations. Cardiovascular research is also expanding, specifically targeting complex and inherited cholesterol disorders. New protocols are currently enrolling individuals diagnosed with dyslipidemias, heterozygous familial hypercholesterolemia, and homozygous familial hypercholesterolemia. Beyond physical health, new behavioral studies are opening to address child behavior and emotional adjustment challenges.
The types of interventions being tested this month are highly diverse, reflecting a comprehensive approach to patient care and disease management. Drug therapies dominate the current landscape, with six new pharmaceutical trials entering the active recruitment phase. However, the state's research ecosystem is also supported by three behavioral interventions, one biological study, and one procedure-based trial. This variety ensures that patients have options ranging from traditional medication testing to innovative psychological support systems.
Research Hubs and Statewide Accessibility
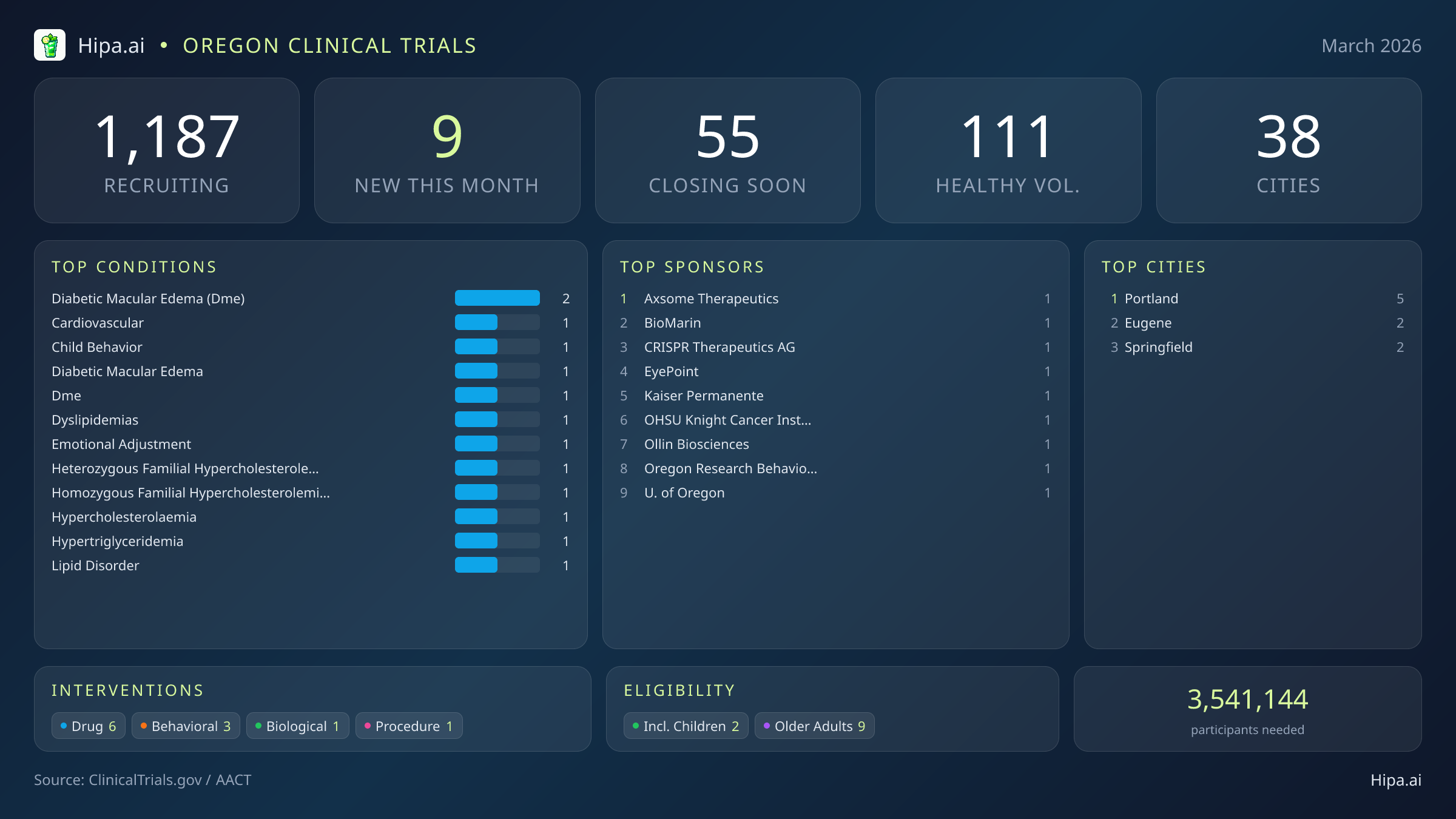
Oregon maintains a robust and highly accessible clinical research infrastructure, with 1,187 active trials currently recruiting participants. These studies are distributed across thirty-eight cities and operate out of more than five hundred individual research sites, ensuring that residents in both urban centers and regional communities have access to scientific advancements.
This month, new trial launches are highly concentrated in the Willamette Valley. Portland continues to serve as the state's primary medical research hub, hosting five of the newly opened studies at its major academic and private research facilities. Further south, the neighboring cities of Eugene and Springfield are expanding their local research footprints, each welcoming two new clinical trials. This regional clustering allows patients living in the state's most populous corridor to easily connect with cutting-edge medical facilities without requiring extensive out-of-state travel.
Broad Eligibility and Volunteer Opportunities
Eligibility criteria for March's new studies are designed to accommodate a wide range of demographic groups, ensuring that medical advancements benefit the entire population. Older adults have extensive options, as all nine of the newly launched trials have structured their protocols to include senior populations. Pediatric research is also moving forward, with two new studies specifically open to children and adolescents facing behavioral or medical challenges. Furthermore, none of the trials launched this month restrict participation based exclusively on sex, ensuring equal access for both male and female participants across all new protocols.
For Oregonians without a specific medical diagnosis, one of this month's new trials is actively welcoming healthy volunteers. This addition brings the total number of open, recruiting trials for healthy participants across the state to 111. These individuals play an essential role in the clinical research process. By participating, healthy volunteers provide the critical baseline data necessary for researchers to accurately evaluate the safety, tolerability, and efficacy of next-generation treatments before they are administered to vulnerable patient populations.
As the spring research season unfolds, Oregon's clinical trial landscape is positioned to drive significant advancements in cardiovascular care, vision preservation, and behavioral health, leveraging the state's strong academic partnerships and expanding regional medical centers to shape the future of patient treatment.
Data Highlights
Conditions Closing Soon
- prostate cancer (2)
- depression (2)
- multiple myeloma (2)
- stage iv lung cancer ajcc v8 (2)
- glaucoma (2)
- adolescent behavior (1)
- acute undifferentiated leukemia (1)
- acute leukemia (1)
Most Common New Trial Conditions
- diabetic macular edema (dme) (2)
- cardiovascular (1)
- child behavior (1)
- diabetic macular edema (1)
- dme (1)
- dyslipidemias (1)
- emotional adjustment (1)
- heterozygous familial hypercholesterolemia (hefh) (1)
Cities With the Most New Trials
- Portland (5)
- Eugene (2)
- Springfield (2)
Leading Sponsors
- Axsome Therapeutics, Inc. (1)
- BioMarin Pharmaceutical (1)
- CRISPR Therapeutics AG (1)
- EyePoint Pharmaceuticals, Inc. (1)
- Kaiser Permanente (1)
- OHSU Knight Cancer Institute (1)
- Ollin Biosciences, Inc. (1)
- Oregon Research Behavioral Intervention Strategies, Inc. (1)
| Month | New Trials | Closing Soon |
|---|---|---|
| November 2025 | 15 | 9 |
| December 2025 | 24 | 27 |
| January 2026 | 15 | 8 |
| February 2026 | 17 | 11 |
| March 2026 | 9 | 24 |
| April 2026 | 5 | 0 |