Pennsylvania Clinical Trials Report — March 2026
49 New Studies, 210 Closing Soon
Hipa.ai Research - March 1, 2026 - Source: Research Data
Monthly Clinical Trials Infographic
Time is running out for Pennsylvanians hoping to participate in several major medical research initiatives. Over the next ninety days, 210 clinical trials will permanently close their enrollment windows across the state. This impending deadline affects both patients seeking novel therapies and residents without underlying conditions, as 38 of these soon-to-close studies are actively looking for healthy volunteers to establish critical baseline data. The wave of closures will particularly impact research into behavioral health, women's health, and oncology, meaning individuals managing these specific diagnoses have a narrow window to explore these experimental care options. Final enrollment phases are rapidly approaching for studies focused on the following conditions:
- Opioid use disorder
- Sleep disorders
- Gestational diabetes
- Prostate cancer
- Endometrial cancer
- Coronary artery disease
- Gastric cancer
- Breast cancer
- Binge eating
Major Phase 3 and High-Capacity Research Launches
Despite these impending closures, March brings a fresh wave of high-capacity research projects to the state. Major pharmaceutical sponsors and leading academic institutions are launching large-scale studies requiring thousands of participants. Pfizer is currently recruiting for a massive Phase 3 clinical trial evaluating a Lyme disease vaccine, while Novo Nordisk is testing a new injectable treatment for weight management. Beyond traditional drug development, researchers are exploring innovative behavioral interventions, digital decision-making tools, and advanced diagnostic tests.
- The University of Pennsylvania is launching a massive randomized controlled trial (NCT07501247) seeking seven thousand two hundred participants to assess S.A.F.E. Firearm, an intervention adapted for nurse-led delivery to parents and caregivers of hospitalized youth.
- Hoffmann-La Roche requires two thousand volunteers to develop a novel blood-based biomarker test (NCT07455136) aimed at aiding the diagnosis and prognosis of traumatic brain injuries in adults, as well as monitoring secondary events.
- The University of Pittsburgh is enrolling nearly two thousand youth for a study (NCT07463118) evaluating whether a game-based intervention called Singularities can effectively reduce alcohol-related harms while improving adaptive coping skills and protective behavioral strategies.
- Pfizer is seeking over seventeen hundred healthy individuals aged seven and older for a Phase 3 trial (NCT07500506) to test the safety and immunogenicity of a fifth dose of its VLA15 Lyme disease vaccine.
- Dartmouth College is sponsoring a study (NCT07454733) involving eight hundred participants to determine if video recordings of multidisciplinary clinic visits improve the quality of life for people navigating amyotrophic lateral sclerosis and their caregivers.
- The University of Pittsburgh is recruiting seven hundred fifty participants to test a web-based decision support tool designed to provide evidence-based information about vasectomy and other birth control options (NCT07451067).
- Novo Nordisk A/S has opened the Phase 3 AMAZE 12 study (NCT07503210), seeking six hundred participants to investigate the safety and efficacy of the weekly subcutaneous injection NNC0487-0111 for maintaining weight loss in individuals with excess body weight.
- HeartBeam, Inc. is launching an ECG registry (NCT07501299) to collect clinical recordings from six hundred participants using their proprietary system in real-world clinical settings.
Geographic Accessibility and Trial Categories
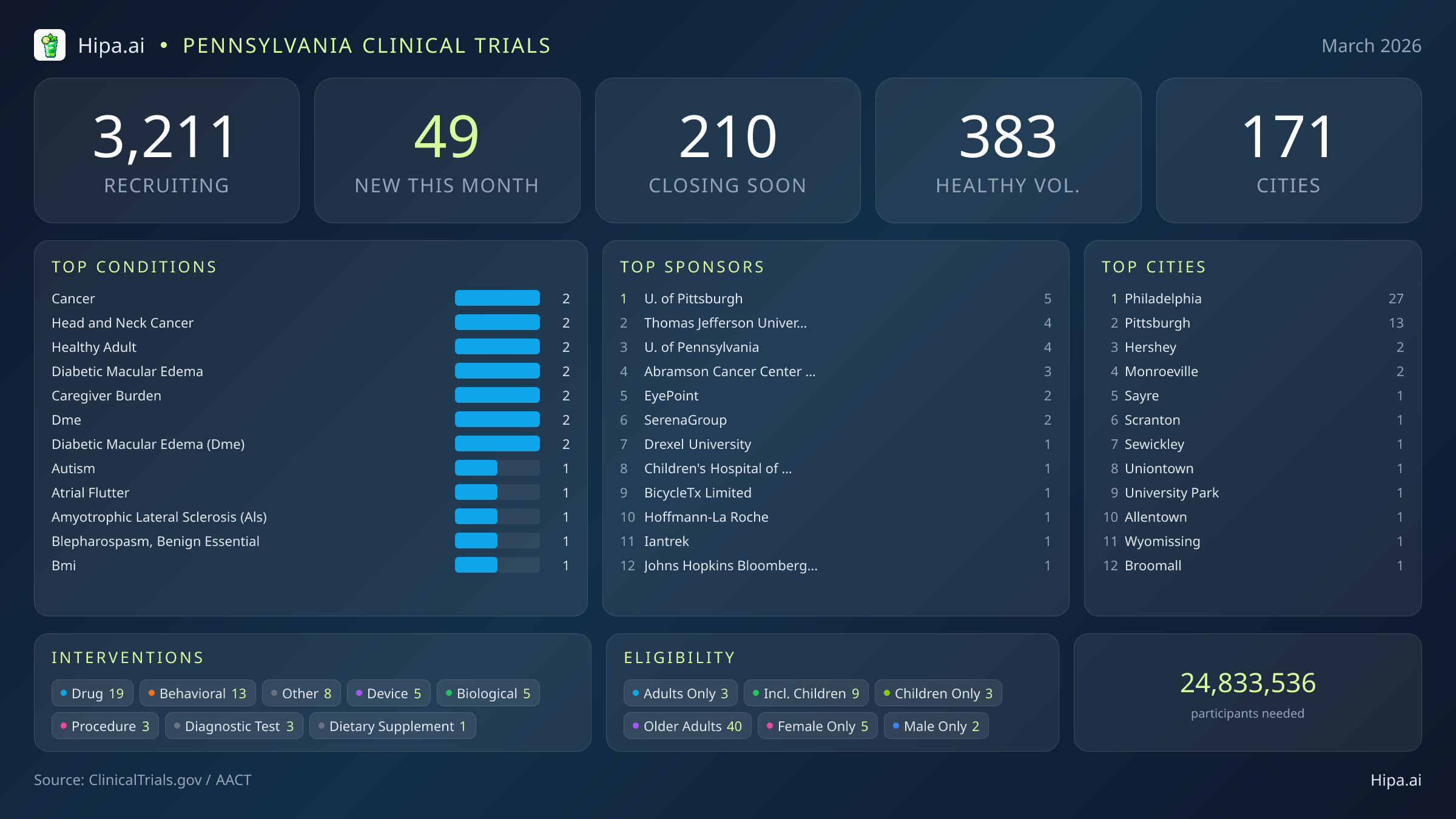
The clinical research infrastructure in Pennsylvania remains robust and highly accessible. There are currently 3,211 active trials recruiting across one hundred seventy-one cities and over seventeen hundred individual research sites statewide. The pace of new study launches has remained relatively steady since the beginning of the year, with 49 new trials opening this month compared to fifty-one in January. The interventions being tested this month are heavily weighted toward pharmaceutical advancements, with nineteen new drug trials entering the recruitment phase. These are followed closely by thirteen behavioral studies, five medical device evaluations, five biological testing protocols, three procedural studies, and three diagnostic test evaluations.
Geographically, new research opportunities are heavily concentrated in the state's largest metropolitan areas. Philadelphia dominates the landscape with twenty-seven newly opened trials, driven largely by institutions like Thomas Jefferson University, the University of Pennsylvania, Drexel University, and the Children's Hospital of Philadelphia. Pittsburgh follows with thirteen new studies, heavily supported by the University of Pittsburgh's extensive research network. However, residents living outside these primary hubs still have access to cutting-edge medical research. New trials have opened their doors this month in several other municipalities across the state:
- Hershey
- Monroeville
- Sayre
- Scranton
- Sewickley
- Uniontown
- University Park
- Allentown
Targeted Conditions and Eligibility
March's new clinical trials target a diverse array of medical conditions, offering new avenues of care for patients managing complex diagnoses. Researchers are actively seeking individuals recently diagnosed with or currently managing diabetic macular edema, head and neck cancer, autism, atrial flutter, and caregiver burden. The sponsorship of these trials reflects a strong collaborative environment. Twenty-nine studies are driven by academic or other institutional organizations, while seventeen are funded directly by industry partners like EyePoint Pharmaceuticals and SerenaGroup, Inc. Federal and network sponsors make up the remainder of the new trial landscape.
Eligibility criteria for these new studies ensure that a wide range of demographic groups can participate in scientific advancement. Older adults have extensive options, with 40 of the new trials specifically designing their protocols to include senior populations. Pediatric research is also active, as nine new studies are open to children and adolescents. Highly targeted demographic research includes five trials exclusively for females and two exclusively for males.
For those without a specific medical diagnosis, ten of this month's new trials are actively welcoming healthy volunteers. This brings the total number of open, recruiting trials for healthy participants across Pennsylvania to 383, offering ample opportunity for residents to contribute to baseline medical research and preventative care initiatives like the Lyme disease vaccine study.
As the spring research season accelerates, Pennsylvania's clinical trial ecosystem is positioned to yield significant data from these large-scale behavioral and vaccine studies, paving the way for next-generation therapeutic breakthroughs and enhanced clinical care models in the latter half of the year.
Data Highlights
Conditions Closing Soon
- opioid use disorder (6)
- sleep (4)
- gestational diabetes (3)
- cancer (3)
- prostate cancer (3)
- endometrial cancer (2)
- coronary artery disease (2)
- gastric cancer (2)
Most Common New Trial Conditions
- cancer (2)
- head and neck cancer (2)
- healthy adult (2)
- diabetic macular edema (2)
- caregiver burden (2)
- dme (2)
- diabetic macular edema (dme) (2)
- autism (1)
Cities With the Most New Trials
- Philadelphia (27)
- Pittsburgh (13)
- Hershey (2)
- Monroeville (2)
- Sayre (1)
- Scranton (1)
- Sewickley (1)
- Uniontown (1)
Leading Sponsors
- University of Pittsburgh (5)
- Thomas Jefferson University (4)
- University of Pennsylvania (4)
- Abramson Cancer Center at Penn Medicine (3)
- EyePoint Pharmaceuticals, Inc. (2)
- SerenaGroup, Inc. (2)
- Drexel University (1)
- Children's Hospital of Philadelphia (1)
| Month | New Trials | Closing Soon |
|---|---|---|
| November 2025 | 40 | 10 |
| December 2025 | 85 | 64 |
| January 2026 | 41 | 15 |
| February 2026 | 51 | 27 |
| March 2026 | 49 | 59 |
| April 2026 | 5 | 0 |