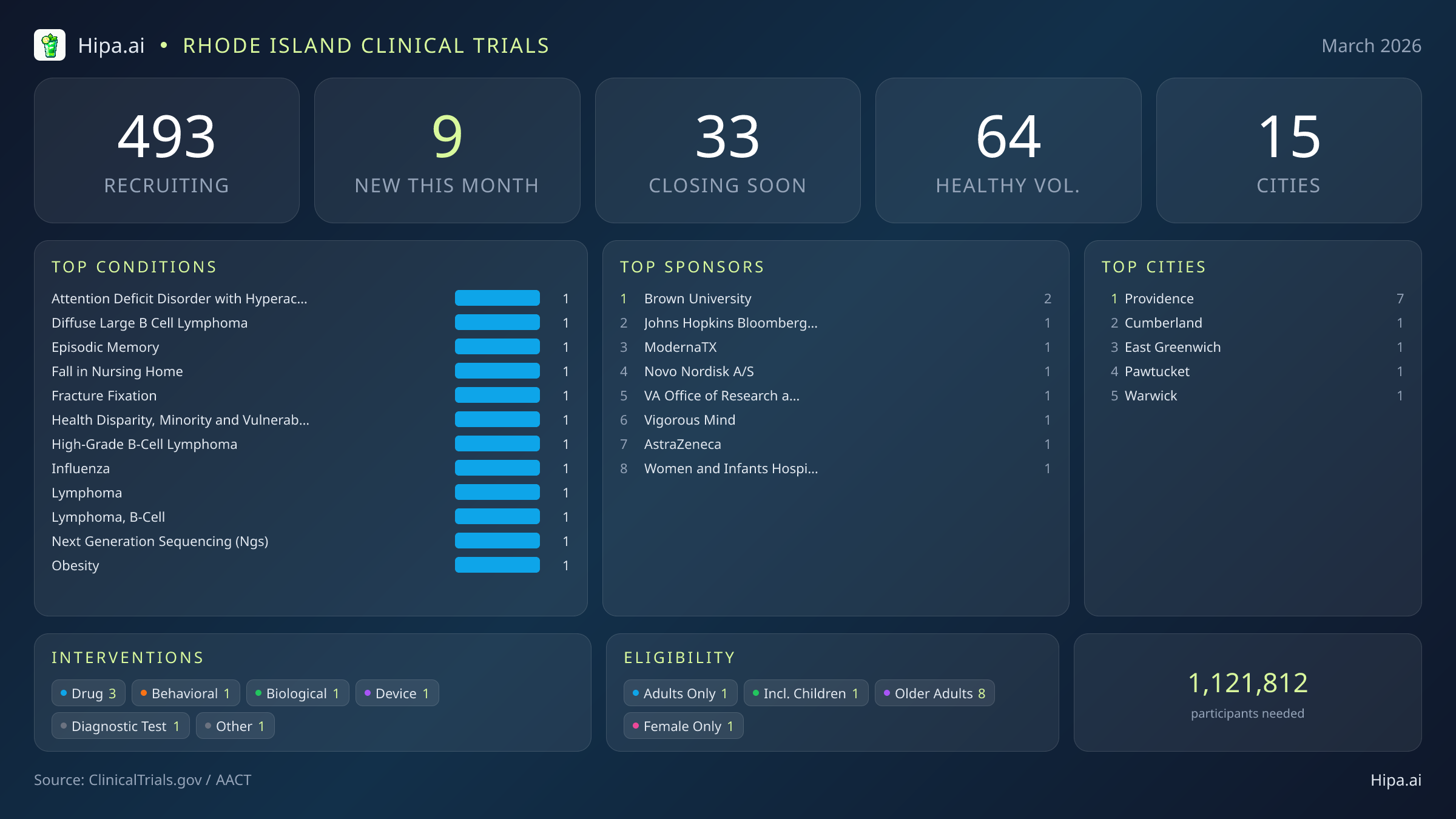
Rhode Island Clinical Trials Report — March 2026
9 New Studies, 33 Closing Soon
Hipa.ai Research - March 1, 2026 - Source: Research Data
Monthly Clinical Trials Infographic
Immediate Enrollment Deadlines for Rhode Island Residents
Time is running out for Rhode Island residents looking to participate in several critical medical research initiatives. Over the next ninety days, 33 clinical trials will permanently close their enrollment windows across the state. This impending deadline creates a brief but valuable opportunity for individuals without underlying health conditions to contribute to scientific discovery, as 8 of these closing studies are actively seeking healthy volunteers. For patients currently managing specific diagnoses, the window to access experimental therapies and behavioral interventions is rapidly disappearing. Final enrollment phases are approaching quickly for studies focused on the following conditions:
- Mild cognitive impairment
- Recurrent endometrial carcinoma
- Early onset Alzheimer's disease
- Anatomic stage III breast cancer
- Aortic stenosis
- Binge eating disorder
- Chronic headache disorder
High-Profile Pharmaceutical and Late-Stage Launches
March brings a revitalized wave of high-profile research to the state, with 9 new trials opening this month—a steady increase from the seven studies launched in January. Industry leaders are driving significant late-stage clinical development, offering local patients access to experimental treatments before they reach the broader market. Three of these newly launched studies are focused on drug interventions, alongside cutting-edge biological and medical device trials. Several notable therapeutic trials from major sponsors are currently seeking participants:
- ModernaTX, Inc. is recruiting four thousand participants for a massive Phase 3 trial (NCT07496450) evaluating the safety and humoral immunogenicity of two doses of the mRNA-1018-H5 pandemic influenza vaccine in healthy adults.
- Novo Nordisk A/S has launched a Phase 3 study (NCT07503210) investigating whether weekly subcutaneous injections of NNC0487-0111 can help individuals with excess body weight safely maintain their weight loss.
Academic Partnerships and Targeted Interventions
Beyond pharmaceutical testing, academic and medical institutions are launching highly targeted behavioral and procedural studies. Brown University is sponsoring two of the new trials, cementing its role as a central hub for medical advancement in the region. Among their new initiatives is a Phase 2 trial (NCT07502872) investigating a novel combination of tafasitamab, polatuzumab vedotin, and glofitamab as a first-line treatment for patients with diffuse large B-cell lymphoma and high-grade B-cell lymphoma.
Women and Infants Hospital of Rhode Island has launched a massive community-based initiative (NCT07446374) seeking three thousand participants. This project aims to improve perinatal health outcomes by integrating community health workers, doulas, and community-based organizations to build a service delivery model that directly addresses care coordination and social determinants of health.
National academic partnerships are also expanding into the state. Johns Hopkins Bloomberg School of Public Health has opened a Phase 3 trial (NCT07458230) focusing on the severe problem of surgical site infections following high-energy bone fractures. Because these complications can affect up to sixty percent of patients with severe injuries, this study utilizes next-generation sequencing to rapidly identify infections and improve wound-healing outcomes.
March's new clinical trials are also targeting a highly specific array of conditions. Researchers are actively seeking patients recently diagnosed with or currently managing attention deficit disorder with hyperactivity, episodic memory challenges, and fall prevention in nursing home settings.
Statewide Access and Research Hubs
Rhode Island's clinical research infrastructure remains highly accessible, with 493 active trials currently recruiting across fifteen cities and nearly two hundred individual research sites. Geographically, new research opportunities are heavily clustered in the state's capital. Providence leads the state with seven newly opened trials, providing a dense concentration of advanced care options. However, residents living outside the immediate metropolitan area still have opportunities to participate. New trials have opened their doors this month in several key municipalities across the state:
- Providence
- Cumberland
- East Greenwich
- Pawtucket
- Warwick
Broad Eligibility and Healthy Volunteer Opportunities
Eligibility criteria for this month's new studies are heavily skewed toward adult and senior populations, ensuring that aging demographics can participate in medical advancement. Older adults have extensive options, with eight of the new trials specifically designing their protocols to include senior participants. Pediatric research is represented by a single new study open to children, while one trial restricts participation exclusively to female patients. None of the new trials launched this month restrict participation exclusively to men.
For those without a specific medical diagnosis, two of this month's newly launched trials are actively welcoming healthy volunteers. This brings the total number of open, recruiting trials for healthy participants across Rhode Island to 64. These trials rely heavily on healthy individuals to establish crucial baseline data for future medical breakthroughs, making community participation essential for scientific progress.
As the spring research season advances, Rhode Island's clinical trial landscape is positioned to expand its focus on metabolic therapies and advanced oncological treatments, while the integration of community-based maternal care models will likely reshape regional approaches to perinatal health and social determinants of care.
Data Highlights
Conditions Closing Soon
- mild cognitive impairment (2)
- recurrent endometrial carcinoma (2)
- alzheimer disease, early onset (1)
- anatomic stage iii breast cancer ajcc v8 (1)
- aortic stenosis (1)
- arthritis (1)
- bone fractures (1)
- binge eating (1)
Most Common New Trial Conditions
- attention deficit disorder with hyperactivity (1)
- diffuse large b cell lymphoma (1)
- episodic memory (1)
- fall in nursing home (1)
- fracture fixation (1)
- health disparity, minority and vulnerable populations (1)
- high-grade b-cell lymphoma (1)
- influenza (1)
Cities With the Most New Trials
- Providence (7)
- Cumberland (1)
- East Greenwich (1)
- Pawtucket (1)
- Warwick (1)
Leading Sponsors
- Brown University (2)
- Johns Hopkins Bloomberg School of Public Health (1)
- ModernaTX, Inc. (1)
- Novo Nordisk A/S (1)
- VA Office of Research and Development (1)
- Vigorous Mind, Inc. (1)
- AstraZeneca (1)
- Women and Infants Hospital of Rhode Island (1)
| Month | New Trials | Closing Soon |
|---|---|---|
| November 2025 | 4 | 1 |
| December 2025 | 6 | 10 |
| January 2026 | 6 | 3 |
| February 2026 | 7 | 4 |
| March 2026 | 9 | 7 |
| April 2026 | 1 | 0 |