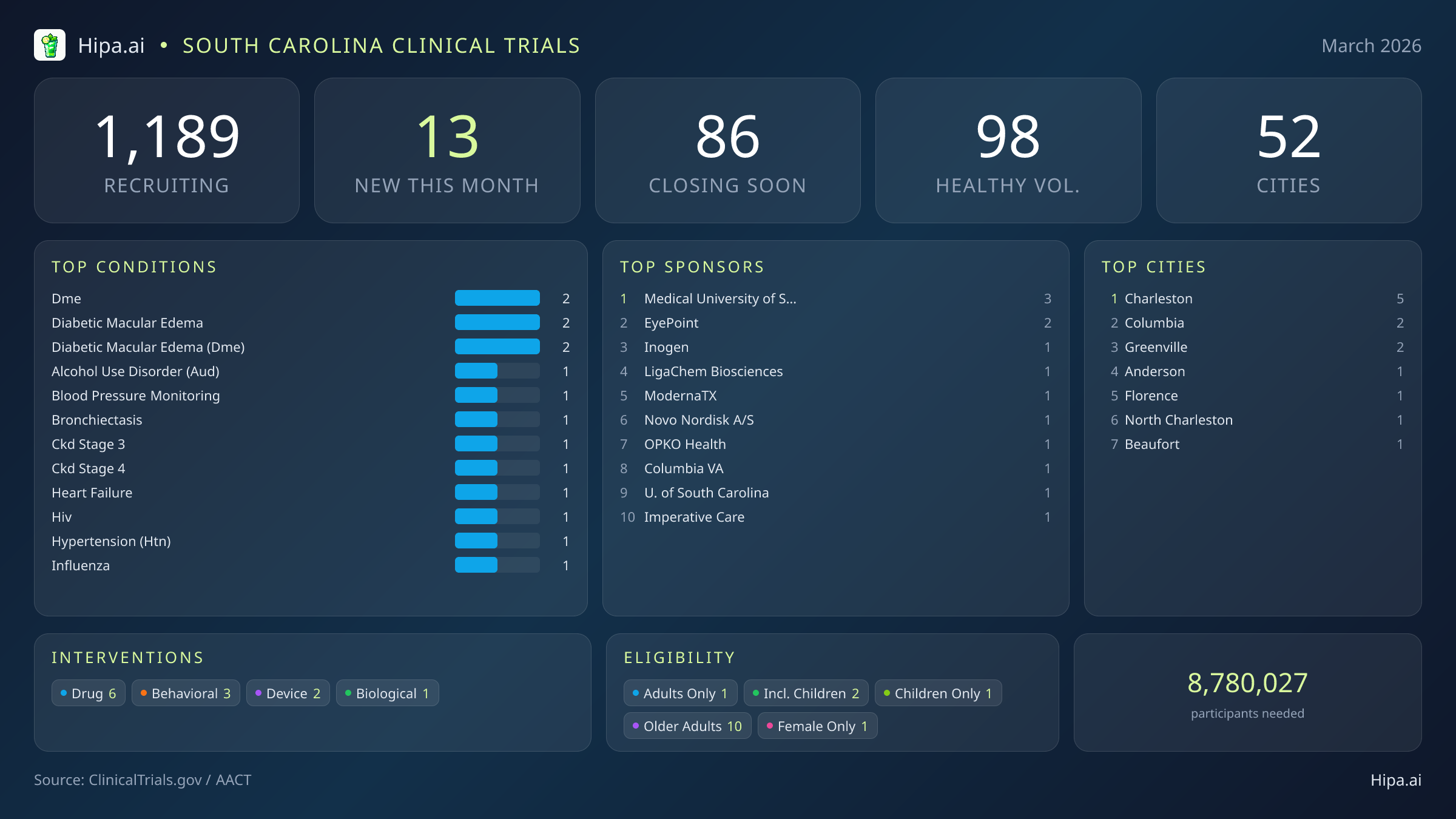
South Carolina Clinical Trials Report — March 2026
13 New Studies, 86 Closing Soon
Hipa.ai Research - March 1, 2026 - Source: Research Data
Monthly Clinical Trials Infographic
Approaching Deadlines for Oncology and Cardiovascular Studies
Time is strictly limited for South Carolina residents hoping to enroll in several critical medical studies. Over the next ninety days, 86 clinical trials across the state will permanently close their enrollment windows. This fast-approaching deadline affects a wide range of patients seeking access to experimental therapies, particularly in the fields of oncology and cardiology. People without underlying health conditions also have a brief window to participate, as 11 of these soon-to-close studies are actively looking for healthy volunteers to provide essential baseline data. Patients managing specific cancer diagnoses face a rapidly shrinking opportunity to access novel therapeutic interventions before these studies move into their data analysis phases. Final enrollment periods are imminent for research targeting the following severe conditions:
- Pancreatic cancer
- Multiple myeloma
- Bladder cancer
- Colorectal cancer
- Head and neck cancer
- Breast and cervical cancers
- Cardiovascular diseases
Late-Stage Pharmaceutical Research and Major Sponsors
Despite the upcoming closures, March introduces a fresh wave of high-profile research to the state. While the overall pace of new study launches has slowed slightly over the past quarter—dropping from thirty-five in December to 13 new trials this month—the caliber of the incoming research remains exceptionally high. Industry sponsors are driving the majority of this month's activity, launching eight new studies with a strong emphasis on late-stage drug development. Pharmaceutical giants are initiating massive Phase 3 trials, giving local residents early access to experimental treatments before they reach the general market. Several standout therapeutic trials are currently seeking participants:
- ModernaTX, Inc. is recruiting four thousand healthy adults for a massive Phase 3 trial (NCT07496450) to evaluate the safety, reactogenicity, and immune response of the mRNA-1018-H5 pandemic influenza vaccine.
- Novo Nordisk A/S has launched a Phase 3 study (NCT07481630) seeking four hundred participants to determine if weekly subcutaneous injections of NNC0487-0111 can help individuals with excess body weight and knee osteoarthritis lose weight and reduce chronic joint pain.
- EyePoint Pharmaceuticals, Inc. is running two concurrent Phase 3 trials, CAPRI (NCT07449923) and COMO (NCT07449936), each recruiting two hundred forty patients to compare the efficacy of EYP-1901 against aflibercept for treating diabetic macular edema.
- OPKO Health, Inc. is enrolling forty pediatric patients with Stage 3 or 4 chronic kidney disease and vitamin D insufficiency in a Phase 2 trial (NCT07494045) to evaluate the safety and pharmacokinetics of CTAP101 extended-release capsules.
Advancements in Medical Devices and Behavioral Interventions
Beyond traditional pharmaceutical testing, South Carolina's academic and medical institutions are launching targeted behavioral and device-based studies this month. The Medical University of South Carolina is sponsoring a highly specialized Phase 2 trial (NCT07503782) evaluating the effects of oleoylethanolamide supplementation on inflammation, the oral microbiome, neurocognitive function, and alcohol use in young adults ages eighteen to twenty-five with alcohol use disorder. In the realm of maternal health, the University of South Carolina has initiated a behavioral study (NCT07457970) comparing remote blood pressure monitoring alone versus an enhanced lifestyle program led by a community health worker for women who experienced hypertensive disorders during pregnancy.
Medical device innovation is also heavily represented in this month's new research portfolio. Imperative Care, Inc. is seeking seven hundred fifty participants for a large-scale study (NCT07491952) designed to evaluate the clinical performance, effectiveness, and safety of the ADAPT 2.0 first-line aspiration neurothrombectomy system for stroke patients. These diverse research avenues ensure that patients managing a wide spectrum of conditions—ranging from bronchiectasis and heart failure to HIV—have immediate opportunities to explore novel care strategies and advanced medical technologies.
Research Hubs and Statewide Accessibility
South Carolina maintains a robust and highly accessible clinical research infrastructure, with 1,189 active trials currently recruiting across fifty-two cities and six hundred seventy-five individual research sites. Geographically, new research opportunities launched in March are heavily concentrated in the coastal Lowcountry and the central Midlands regions. Charleston leads the state's research expansion with five newly opened trials, bolstered significantly by the academic and clinical presence of the Medical University of South Carolina. However, residents living outside the immediate Charleston metropolitan area still have multiple access points to participate in cutting-edge medical research. New trials have opened their doors this month in several key municipalities spanning from the Upstate to the coast:
- Charleston
- Columbia
- Greenville
- Anderson
- Florence
- North Charleston
- Beaufort
Inclusive Eligibility and Healthy Volunteer Opportunities
Eligibility criteria for March's new studies are intentionally designed to accommodate a wide demographic cross-section of South Carolina residents, ensuring that medical advancements benefit diverse populations. Older adults have extensive options for participation, with ten of the new trials specifically designing their protocols to include senior populations who often face higher burdens of chronic disease. Pediatric research is also actively recruiting, as two new studies are open to children and adolescents requiring specialized interventions. The state's research landscape remains highly accessible across gender lines, with only one new trial restricting participation exclusively to females and zero trials restricting participation to males.
For individuals without a specific medical diagnosis, 98 total trials are currently recruiting healthy volunteers statewide. While only one of this month's newly launched studies—the massive ModernaTX pandemic influenza vaccine trial—is actively welcoming healthy participants, these individuals remain a crucial component of the clinical research ecosystem. Healthy volunteers provide the essential baseline data required to accurately measure the safety, tolerability, and efficacy of new treatments before they can be approved for widespread public use.
As the spring research season unfolds, South Carolina's clinical trial landscape is positioned to drive significant advancements in metabolic therapies and infectious disease prevention, while the comprehensive datasets collected from this month's behavioral studies will likely shape the next generation of maternal and cardiovascular care protocols.
Data Highlights
Conditions Closing Soon
- pancreatic cancer (3)
- multiple myeloma (2)
- bladder cancer (2)
- colorectal cancer (2)
- head and neck cancer (2)
- lung cancer (2)
- cancer (2)
- breast cancer (2)
Most Common New Trial Conditions
- dme (2)
- diabetic macular edema (2)
- diabetic macular edema (dme) (2)
- alcohol use disorder (aud) (1)
- blood pressure monitoring (1)
- bronchiectasis (1)
- ckd stage 3 (1)
- ckd stage 4 (1)
Cities With the Most New Trials
- Charleston (5)
- Columbia (2)
- Greenville (2)
- Anderson (1)
- Florence (1)
- North Charleston (1)
- Beaufort (1)
Leading Sponsors
- Medical University of South Carolina (3)
- EyePoint Pharmaceuticals, Inc. (2)
- Inogen Inc. (1)
- LigaChem Biosciences, Inc. (1)
- ModernaTX, Inc. (1)
- Novo Nordisk A/S (1)
- OPKO Health, Inc. (1)
- Columbia VA Health Care System (1)
| Month | New Trials | Closing Soon |
|---|---|---|
| October 2025 | 26 | 3 |
| November 2025 | 24 | 3 |
| December 2025 | 35 | 14 |
| January 2026 | 29 | 11 |
| February 2026 | 18 | 8 |
| March 2026 | 13 | 21 |