Tennessee Clinical Trials Report — March 2026
27 New Studies, 97 Closing Soon
Hipa.ai Research - March 1, 2026 - Source: Research Data
Monthly Clinical Trials Infographic
Approaching Deadlines for Tennessee Residents
Time is running out for Tennesseans seeking access to several critical medical research programs. Over the next ninety days, 97 clinical trials across the state will permanently close their enrollment windows. This impending deadline creates a narrow window for individuals looking to access experimental therapies before they potentially move to the next phase of regulatory review or become unavailable. People without underlying medical conditions also have an immediate opportunity to contribute to scientific advancement, as 14 of these soon-to-close studies are actively seeking healthy volunteers. Patients currently managing specific diagnoses face a rapidly shrinking timeframe to join studies focused on the following conditions:
- Breast cancer and metastatic breast cancer
- Major depressive disorder
- Glaucoma
- Acute kidney injury
- Prostate cancer
- Alzheimer's disease
- Acute myeloid leukemia
- Colorectal cancer
- Dry eye disease
Major Pharmaceutical and Late-Stage Clinical Launches
March brings a fresh wave of high-profile research to the state, heavily driven by pharmaceutical innovation and advanced diagnostic testing. Of the new trials launched this month, 16 are focused on drug interventions, while others explore medical devices and diagnostic tools. Industry leaders like Boehringer Ingelheim and Hoffmann-La Roche are initiating massive, late-stage studies, offering local patients access to experimental treatments and cutting-edge monitoring technologies. Several notable therapeutic and diagnostic trials are currently seeking participants:
- Hoffmann-La Roche is recruiting two thousand participants for a massive diagnostic study (NCT07455136) to develop a blood-based biomarker test. This test aims to aid in the diagnosis and prognosis of traumatic brain injury in adults, while also monitoring the development of secondary events in diagnosed patients.
- Imperative Care, Inc. is seeking seven hundred fifty volunteers for a device trial (NCT07491952) evaluating the safety and clinical performance of the ADAPT 2.0 first-line aspiration neurothrombectomy system, utilizing a continuous dual aspiration technique for stroke patients.
- Boehringer Ingelheim has launched a Phase 3 study (NCT07472517) enrolling six hundred seventy adults with advanced small cell lung cancer. The trial will determine if an experimental medicine called obrixtamig, when combined with standard atezolizumab, carboplatin, and etoposide treatments, improves survival rates compared to standard chemotherapy alone.
- Axsome Therapeutics, Inc. is conducting a Phase 3, double-blind, placebo-controlled randomized withdrawal trial (NCT07484217) testing solriamfetol in patients suffering from major depressive disorder accompanied by excessive daytime sleepiness.
- Boehringer Ingelheim is also sponsoring a Phase 3 trial (NCT07497087) evaluating nerandomilast for adults with systemic sclerosis. The study aims to improve symptoms and slow disease progression in patients with limited or diffuse cutaneous manifestations.
- Johns Hopkins Bloomberg School of Public Health is running a Phase 3 study (NCT07458230) investigating the efficacy of next-generation sequencing for treating surgical site infections following high-energy bone fracture fixation, a complication that can affect up to sixty percent of patients with severe injuries.
- Iantrek, Inc. is collecting long-term data through a follow-up study (NCT07480343) tracking the five-year postoperative outcomes of patients who previously completed the CREST glaucoma study without requiring secondary intraocular pressure-lowering surgery.
Beyond these massive initiatives, researchers are also launching targeted studies for specific chronic and acute conditions. New trials opening this month are actively seeking patients diagnosed with diabetic macular edema, head and neck squamous cell carcinoma, ALK-positive non-small cell lung cancer, gait disorders, and acute uncomplicated diverticulitis.
Broad Eligibility and Healthy Volunteer Opportunities
Eligibility criteria for this month's new studies are exceptionally inclusive, ensuring that diverse demographic groups can participate in medical advancement. Older adults have extensive options, with twenty-five of the newly launched trials specifically designing their protocols to include senior populations. None of the new trials restrict participation exclusively by sex, ensuring equal access for both male and female participants.
Pediatric research is also strongly represented this month, driven largely by local academic and medical institutions. St. Jude Children's Research Hospital is sponsoring three new studies, including a highly specialized observational trial (NCT07502885) examining gait deficits in pediatric cancer patients who have undergone orthopedic surgery. This specific study is actively recruiting healthy participants without cancer to establish baseline walking metrics, helping researchers better understand the severity of mobility issues in recovering children and how those issues change over time.
For those without a specific medical diagnosis, three of this month's 27 new trials are actively welcoming healthy volunteers. This brings the total number of open, recruiting trials for healthy participants across Tennessee to 141. These healthy volunteer cohorts remain essential for establishing baseline data across both pharmaceutical drug trials and behavioral interventions.
Statewide Access and Research Hubs
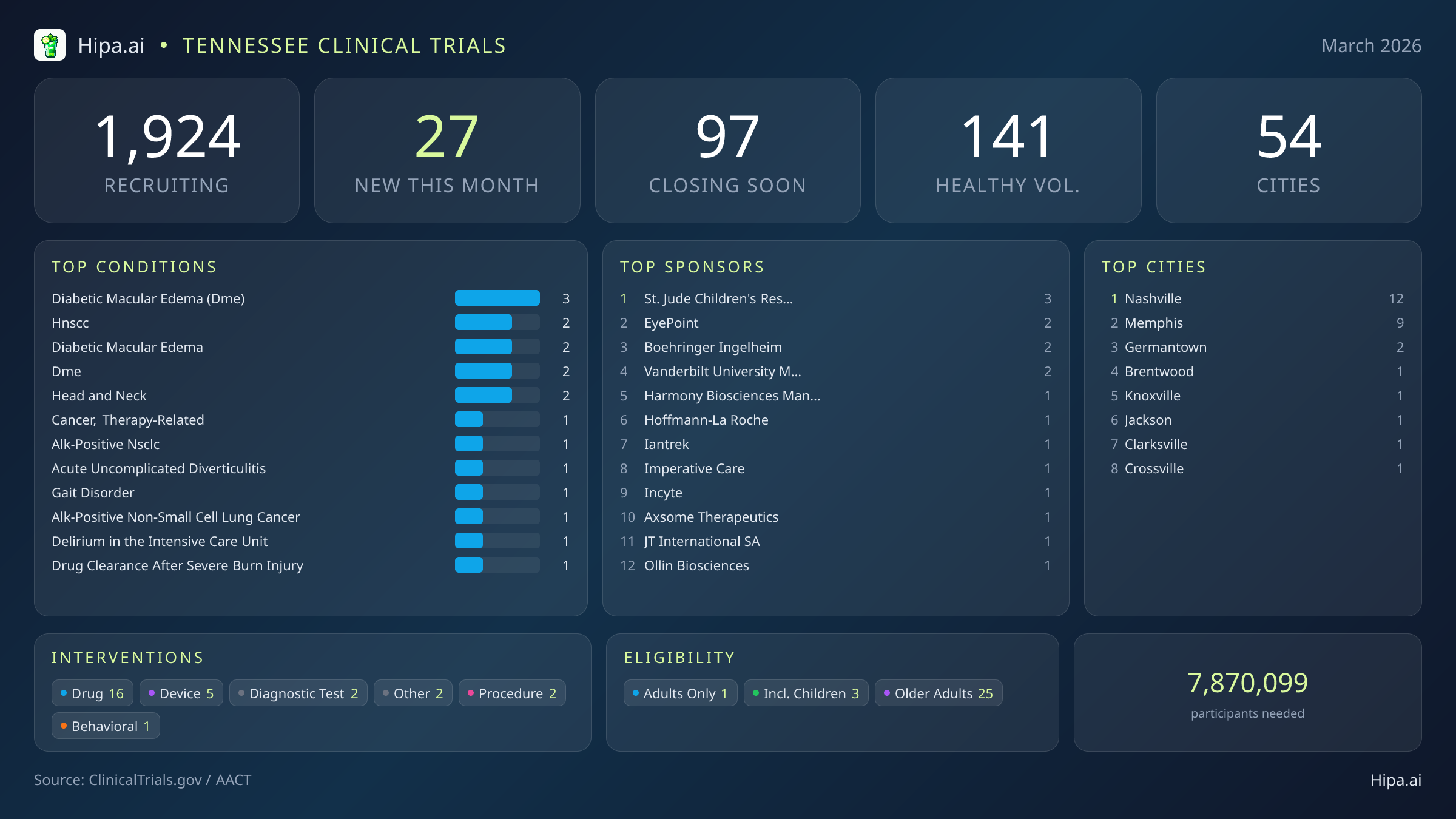
Tennessee's clinical research infrastructure remains robust and highly accessible, with 1,924 active trials currently recruiting across fifty-four cities and over one thousand individual research sites. While the pace of new study launches has cooled slightly since the beginning of the year—dropping from thirty-six new trials in January to twenty-seven this month—the geographic distribution ensures that residents across the state have access to cutting-edge medical research.
Geographically, new research opportunities are heavily clustered in the state's largest metropolitan areas. Nashville leads the state with twelve newly opened trials, bolstered by institutions like Vanderbilt University Medical Center. Memphis follows closely with nine new studies. However, residents living outside the immediate capital and major urban centers still have opportunities to participate. New trials have opened their doors this month in several key municipalities:
- Nashville
- Memphis
- Germantown
- Brentwood
- Knoxville
- Jackson
- Clarksville
- Crossville
As the spring research season accelerates, Tennessee's clinical trial landscape is positioned to expand its focus on advanced diagnostic tools and targeted oncology therapies, while the massive datasets collected from this month's biomarker studies will likely shape the next generation of traumatic brain injury protocols.
Data Highlights
Conditions Closing Soon
- breast cancer (3)
- metastatic breast cancer (3)
- major depressive disorder (2)
- glaucoma (2)
- acute kidney injury (2)
- prostate cancer (2)
- alzheimer disease (2)
- acute myeloid leukemia (2)
Most Common New Trial Conditions
- diabetic macular edema (dme) (3)
- hnscc (2)
- diabetic macular edema (2)
- dme (2)
- head and neck (2)
- cancer, therapy-related (1)
- alk-positive nsclc (1)
- acute uncomplicated diverticulitis (1)
Cities With the Most New Trials
- Nashville (12)
- Memphis (9)
- Germantown (2)
- Brentwood (1)
- Knoxville (1)
- Jackson (1)
- Clarksville (1)
- Crossville (1)
Leading Sponsors
- St. Jude Children's Research Hospital (3)
- EyePoint Pharmaceuticals, Inc. (2)
- Boehringer Ingelheim (2)
- Vanderbilt University Medical Center (2)
- Harmony Biosciences Management, Inc. (1)
- Hoffmann-La Roche (1)
- Iantrek, Inc. (1)
- Imperative Care, Inc. (1)
| Month | New Trials | Closing Soon |
|---|---|---|
| November 2025 | 31 | 5 |
| December 2025 | 46 | 31 |
| January 2026 | 34 | 12 |
| February 2026 | 35 | 13 |
| March 2026 | 27 | 29 |
| April 2026 | 5 | 0 |