Texas Clinical Trials Report — March 2026
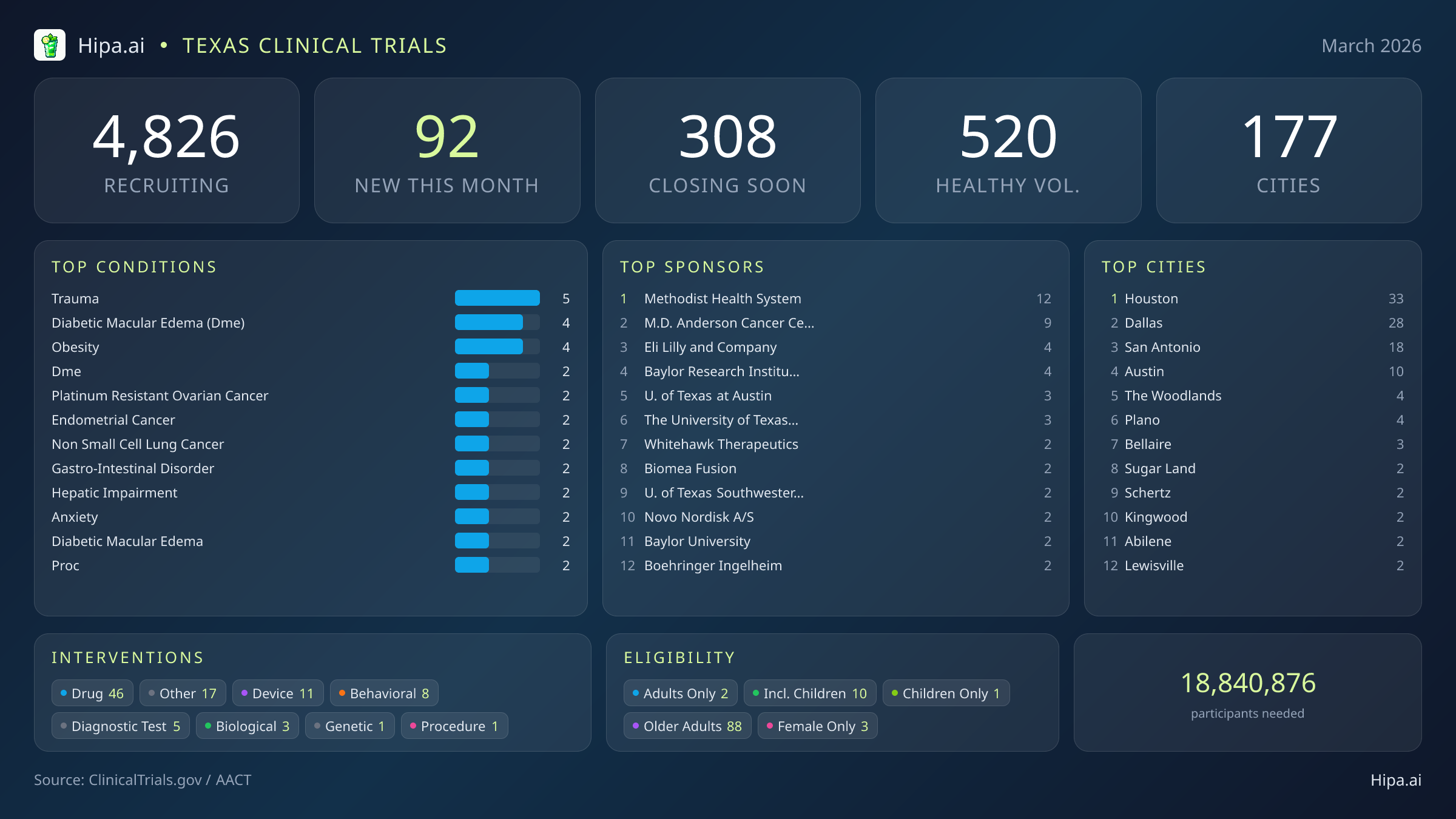
92 New Studies, 308 Closing Soon
Hipa.ai Research - March 1, 2026 - Source: Research Data
Monthly Clinical Trials Infographic
Texans exploring medical research face an immediate deadline as 308 clinical trials prepare to close their enrollment windows within the next ninety days. This wave of closures impacts both patients seeking novel therapies and individuals looking to contribute to general scientific advancement. For those without underlying medical conditions, 48 of these soon-to-close studies are actively seeking healthy volunteers, presenting a rapidly shrinking timeframe to participate in baseline health measurements and control group evaluations. Patients currently managing chronic or terminal diagnoses also face urgent timelines to join studies targeting specific diseases before they move into the data analysis phase. Final enrollment phases are approaching for protocols focused on the following conditions:
- Prostate cancer
- Alzheimer's disease
- Myelodysplastic syndrome
- Type 2 diabetes mellitus
- Acute myeloid leukemia
- Coronary artery disease
- Advanced solid tumors
- Metastatic malignant solid neoplasms
- Anxiety
High-Capacity Registries and Phase 3 Vaccine Research
Despite the impending closures, March brings a series of massive research initiatives to the state, requiring tens of thousands of participants across multiple medical disciplines. ModernaTX, Inc. is actively recruiting healthy adults for a Phase 3 clinical trial evaluating a pandemic influenza vaccine. This study seeks four thousand participants to measure the immune response and safety profile of the mRNA-1018-H5 vaccine, offering residents a direct role in advancing global infectious disease preparedness. Beyond infectious disease, major healthcare organizations and academic institutions are launching high-capacity observational studies and registries designed to reshape emergency response, mental health support, and cancer care.
- The National Minority Quality Forum is launching a massive initiative (NCT07461493) seeking one hundred thousand participants to establish a patient registry and biorepository. This project is aimed at investigating health disparities, access issues, and barriers to cancer screening and early detection technologies.
- Methodist Health System is recruiting five thousand individuals for an observational study (NCT07490145) to quantify over-triage and under-triage rates relative to trauma activation criteria reform, which could streamline emergency room protocols.
- ModernaTX, Inc. is seeking healthy adults for its Phase 3 pandemic influenza vaccine trial (NCT07496450) to evaluate humoral immunogenicity and reactogenicity after two doses of the mRNA-1018-H5 vaccine.
- The University of Texas at Austin is enrolling one thousand patients with high cholesterol (NCT07464574) to coordinate follow-up appointments and review medications in an effort to optimize non-statin agents for cardiovascular disease management.
- Methodist Health System is also establishing a registry of nine hundred ninety-nine participants (NCT07492758) to track extremity injuries. This dataset will support quality improvement observations specific to life and limb salvage outcomes.
- Stony Brook University is recruiting eight hundred first responders (NCT07478250) for resilience training designed to reduce the risk of developing post-traumatic stress disorder and other mental health symptoms resulting from chronic exposure to stressful events.
- The University of Texas Health Science Center at Houston is testing a novel employment-based housing program (NCT07470372) for seven hundred fifty adults at risk of homelessness. The project examines how housing and vocational services can be integrated directly into hospital emergency departments.
- Imperative Care, Inc. is evaluating a continuous dual aspiration technique for stroke treatment (NCT07491952) across seven hundred fifty participants to measure the clinical performance of first-line aspiration neurothrombectomy.
Emerging Therapeutic Focus Areas
The pace of new study launches has moderated slightly since the beginning of the year, dropping from one hundred twenty-nine new trials in January to 92 this month. Pharmaceutical interventions dominate this new landscape, with 46 drug trials entering the recruitment phase. Researchers are also evaluating eleven new medical devices, eight behavioral interventions, five diagnostic tests, three biological treatments, one genetic study, and one procedural evaluation.
March's new clinical trials target a diverse array of acute injuries and chronic diseases. Trauma and obesity are major focus areas this month, alongside multiple new protocols for diabetic macular edema. Oncology research continues to expand its footprint in the state, with specialized studies opening for patients managing platinum-resistant ovarian cancer, endometrial cancer, and non-small cell lung cancer. Additional studies are investigating treatments for gastrointestinal disorders, hepatic impairment, and anxiety.
Institutional and academic sponsors are driving the majority of this month's new research, funding forty-nine of the newly opened studies, while industry partners sponsor forty-three. Methodist Health System leads the state with twelve new trials, followed closely by M.D. Anderson Cancer Center with nine. Pharmaceutical companies like Eli Lilly and Company, Biomea Fusion Inc., and Whitehawk Therapeutics, Inc. are also initiating new therapeutic investigations alongside academic powerhouses like the Baylor Research Institute and the University of Texas system.
Statewide Access and Volunteer Eligibility
The clinical research infrastructure across Texas remains vast and highly accessible. There are currently 4,826 active trials recruiting across one hundred seventy-seven cities and over four thousand three hundred individual research sites. Geographically, new research opportunities are heavily clustered in the state's largest metropolitan areas. Houston leads the state with thirty-three newly opened trials, while Dallas follows closely with twenty-eight. San Antonio and Austin also offer significant new opportunities, hosting eighteen and ten new trials, respectively. Residents living outside these primary urban centers still have access to cutting-edge medical research, as new trials have opened their doors this month in several other municipalities:
- The Woodlands
- Plano
- Bellaire
- Sugar Land
- Schertz
- Kingwood
Eligibility criteria for these new studies are exceptionally broad, ensuring that diverse demographic groups can participate in medical advancement. Older adults have extensive options, with eighty-eight of the new trials specifically designing their protocols to include senior populations. Pediatric research is also available, as ten new studies are open to children and adolescents. Highly targeted demographic research includes three trials exclusively for females, while no new trials this month are restricted exclusively to males.
For those without a specific medical diagnosis, 17 of this month's new trials are actively welcoming healthy volunteers. This brings the total number of open, recruiting trials for healthy participants across Texas to 520, offering ample opportunity for residents to engage with the scientific community.
As researchers begin processing the massive influx of data from this month's high-capacity trauma registries and Phase 3 vaccine trials, the Texas clinical research ecosystem is positioning itself to deliver critical insights into emergency medicine protocols and global infectious disease preparedness by the third quarter of the year.
Data Highlights
Conditions Closing Soon
- prostate cancer (5)
- alzheimer disease (4)
- myelodysplastic syndrome (3)
- diabetes mellitus, type 2 (3)
- metastatic cancer (3)
- metastatic malignant solid neoplasm (3)
- advanced solid tumor (3)
- coronary artery disease (3)
Most Common New Trial Conditions
- trauma (5)
- diabetic macular edema (dme) (4)
- obesity (4)
- dme (2)
- platinum resistant ovarian cancer (2)
- endometrial cancer (2)
- non small cell lung cancer (2)
- gastro-intestinal disorder (2)
Cities With the Most New Trials
- Houston (33)
- Dallas (28)
- San Antonio (18)
- Austin (10)
- The Woodlands (4)
- Plano (4)
- Bellaire (3)
- Sugar Land (2)
Leading Sponsors
- Methodist Health System (12)
- M.D. Anderson Cancer Center (9)
- Eli Lilly and Company (4)
- Baylor Research Institute (4)
- University of Texas at Austin (3)
- The University of Texas Health Science Center, Houston (3)
- Whitehawk Therapeutics, Inc. (2)
- Biomea Fusion Inc. (2)
| Month | New Trials | Closing Soon |
|---|---|---|
| November 2025 | 90 | 21 |
| December 2025 | 136 | 100 |
| January 2026 | 111 | 31 |
| February 2026 | 123 | 32 |
| March 2026 | 92 | 85 |
| April 2026 | 17 | 0 |