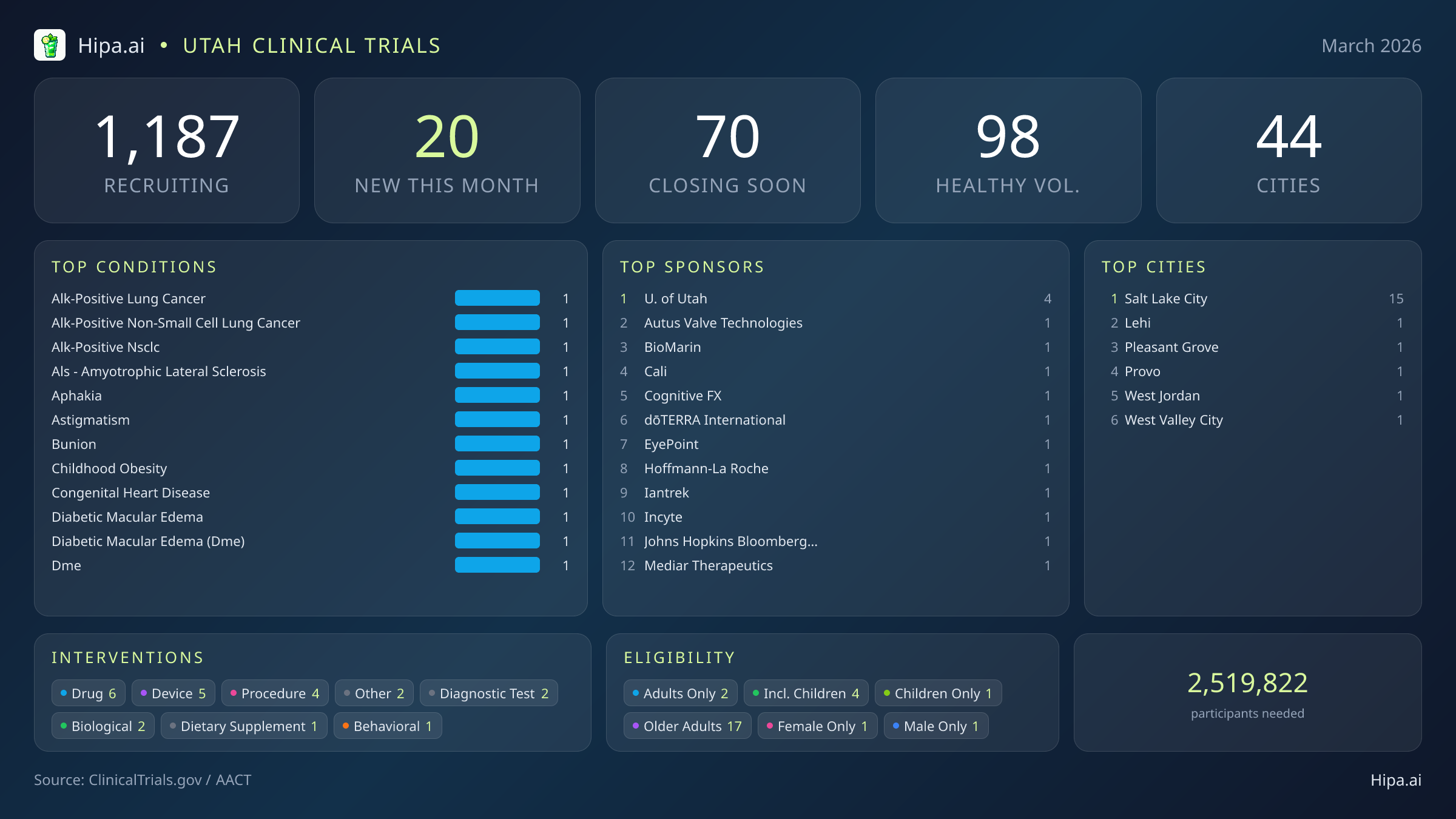
Utah Clinical Trials Report — March 2026
20 New Studies, 70 Closing Soon
Hipa.ai Research - March 1, 2026 - Source: Research Data
Monthly Clinical Trials Infographic
Impending Deadlines for Utah Research Initiatives
Time is running out for Utah residents looking to participate in several critical medical studies. Within the next 90 days, researchers will permanently close enrollment for 70 clinical trials across the state. This creates an immediate window for patients managing specific diagnoses to access experimental therapies before they advance to the next phase of regulatory review. Individuals without underlying health conditions also face a narrowing timeframe, as nine of these closing studies are actively seeking healthy volunteers to provide essential baseline data. Final enrollment phases are approaching rapidly for studies focused on the following conditions:
- Acute lymphoblastic leukemia
- Acute myeloid leukemia
- Advanced solid tumors
- Chronic spontaneous urticaria
- Major depressive disorder
- Multiple myeloma
- Non-small cell lung cancer
- Pulmonary hypertension
High-Profile Industry and Late-Stage Clinical Launches
March brings a fresh wave of scientific investigation to the state, with 20 new trials opening their doors—matching the enrollment pace seen earlier this year in January. Industry sponsors are driving the majority of this month's research, launching fourteen distinct studies that give local patients access to cutting-edge medical technology and experimental pharmaceuticals. Six of these newly launched studies focus on drug interventions, while five explore novel medical devices. Several notable therapeutic trials from major sponsors are currently seeking participants:
- Hoffmann-La Roche is recruiting two thousand participants for a massive diagnostic study (NCT07455136) to develop a blood-based biomarker test that aids in diagnosing traumatic brain injuries and monitoring secondary events in adult patients.
- EyePoint Pharmaceuticals, Inc. has launched a Phase 3 randomized, double-masked trial (NCT07449936) comparing the efficacy of EYP-1901 against aflibercept for patients managing diabetic macular edema.
- Incyte Corporation is enrolling participants in a Phase 1 trial (NCT07441694) to determine the safety, tolerability, and maximum tolerated dose of INCA036978 for individuals diagnosed with myeloproliferative neoplasms.
- Whitehawk Therapeutics, Inc. is conducting a first-in-human Phase 1 study (NCT07470853) evaluating HWK-016, a targeted antibody-drug conjugate designed for adult participants with advanced or metastatic solid tumors. The study employs a dose escalation and dose expansion design without a control group.
- Iantrek, Inc. is seeking volunteers for a long-term observational study (NCT07480343) to collect five-year postoperative clinical data from patients who previously completed the CREST Study for intraocular pressure-lowering interventions.
Academic Innovation and Targeted Interventions
Beyond pharmaceutical development, academic institutions are launching highly targeted diagnostic and procedural studies this month. The University of Utah is sponsoring four of the new trials, reinforcing its position as a primary engine for medical advancement in the Mountain West. Among their new initiatives is a diagnostic study (NCT07460440) testing an artificial intelligence-enabled RNA-based classifier. This trial will analyze retrospective platelet transcriptomic data from over five hundred patients to determine if unique analytical approaches can accurately screen for non-small cell lung cancer, the most common type of lung cancer.
National academic partnerships are also expanding their footprint in the state. Johns Hopkins Bloomberg School of Public Health has opened a Phase 3 trial (NCT07458230) targeting the severe problem of surgical site infections following high-energy bone fractures. Because these complications can affect up to sixty percent of patients with severe injuries, researchers are utilizing next-generation sequencing to rapidly identify infections and improve wound-healing outcomes.
Behavioral health and preventative medicine are also receiving significant attention. A new hybrid effectiveness-implementation trial (NCT07479498) sponsored by Paul Estabrooks is recruiting six hundred participants to assess population health management strategies. The study utilizes text messaging and enrollment in the Building Healthy Families program to support lower-income families managing childhood obesity. March's new clinical trials are also targeting a highly specific array of chronic and acute conditions, actively seeking patients recently diagnosed with or currently managing alk-positive lung cancer, amyotrophic lateral sclerosis, astigmatism, and congenital heart disease.
Geographic Accessibility Across the Mountain West
Utah's clinical research infrastructure provides extensive access to experimental care, with 1,187 active trials currently recruiting across 44 cities and over five hundred individual research sites. Geographically, new research opportunities are heavily concentrated in the state's primary metropolitan and academic center. Salt Lake City leads the state with 15 newly opened trials, offering a dense cluster of advanced care options for residents in the immediate area.
However, individuals living outside the capital still have opportunities to participate in emerging research. New trials have opened their doors this month in several key municipalities across the Wasatch Front:
- Lehi
- Pleasant Grove
- Provo
- West Jordan
- West Valley City
Broad Eligibility and Healthy Volunteer Opportunities
Eligibility criteria for this month's new studies heavily favor adult and senior populations, ensuring that aging demographics can actively participate in medical advancement. Older adults have extensive options, with seventeen of the new trials specifically designing their protocols to include senior participants. Pediatric research is represented by four new studies open to children, while gender-specific research includes one trial restricted exclusively to female patients and one trial restricted exclusively to men.
For those without a specific medical diagnosis, five of this month's newly launched trials are actively welcoming healthy volunteers. This brings the total number of open, recruiting trials for healthy participants across Utah to 98. These trials rely on healthy individuals to establish crucial baseline data for future medical breakthroughs, making community participation essential for scientific progress.
As the spring research season advances, Utah's clinical trial landscape is positioned to expand its focus on artificial intelligence diagnostics and targeted oncological therapies, while the integration of population health management strategies will likely shape the future of preventative pediatric care.
Data Highlights
Conditions Closing Soon
- non-small cell lung cancer (2)
- acute lymphoblastic leukemia (2)
- chronic spontaneous urticaria (2)
- multiple myeloma (2)
- stage ivb prostate cancer ajcc v8 (2)
- pulmonary hypertension (2)
- advanced solid tumor (2)
- acute myeloid leukemia (2)
Most Common New Trial Conditions
- alk-positive lung cancer (1)
- alk-positive non-small cell lung cancer (1)
- alk-positive nsclc (1)
- als - amyotrophic lateral sclerosis (1)
- aphakia (1)
- astigmatism (1)
- bunion (1)
- childhood obesity (1)
Cities With the Most New Trials
- Salt Lake City (15)
- Lehi (1)
- Pleasant Grove (1)
- Provo (1)
- West Jordan (1)
- West Valley City (1)
Leading Sponsors
- University of Utah (4)
- Autus Valve Technologies, Inc. (1)
- BioMarin Pharmaceutical (1)
- Cali Pharmaceuticals LLC (1)
- Cognitive FX (1)
- dōTERRA International (1)
- EyePoint Pharmaceuticals, Inc. (1)
- Hoffmann-La Roche (1)
| Month | New Trials | Closing Soon |
|---|---|---|
| November 2025 | 20 | 8 |
| December 2025 | 34 | 23 |
| January 2026 | 22 | 4 |
| February 2026 | 18 | 4 |
| March 2026 | 20 | 23 |
| April 2026 | 5 | 0 |