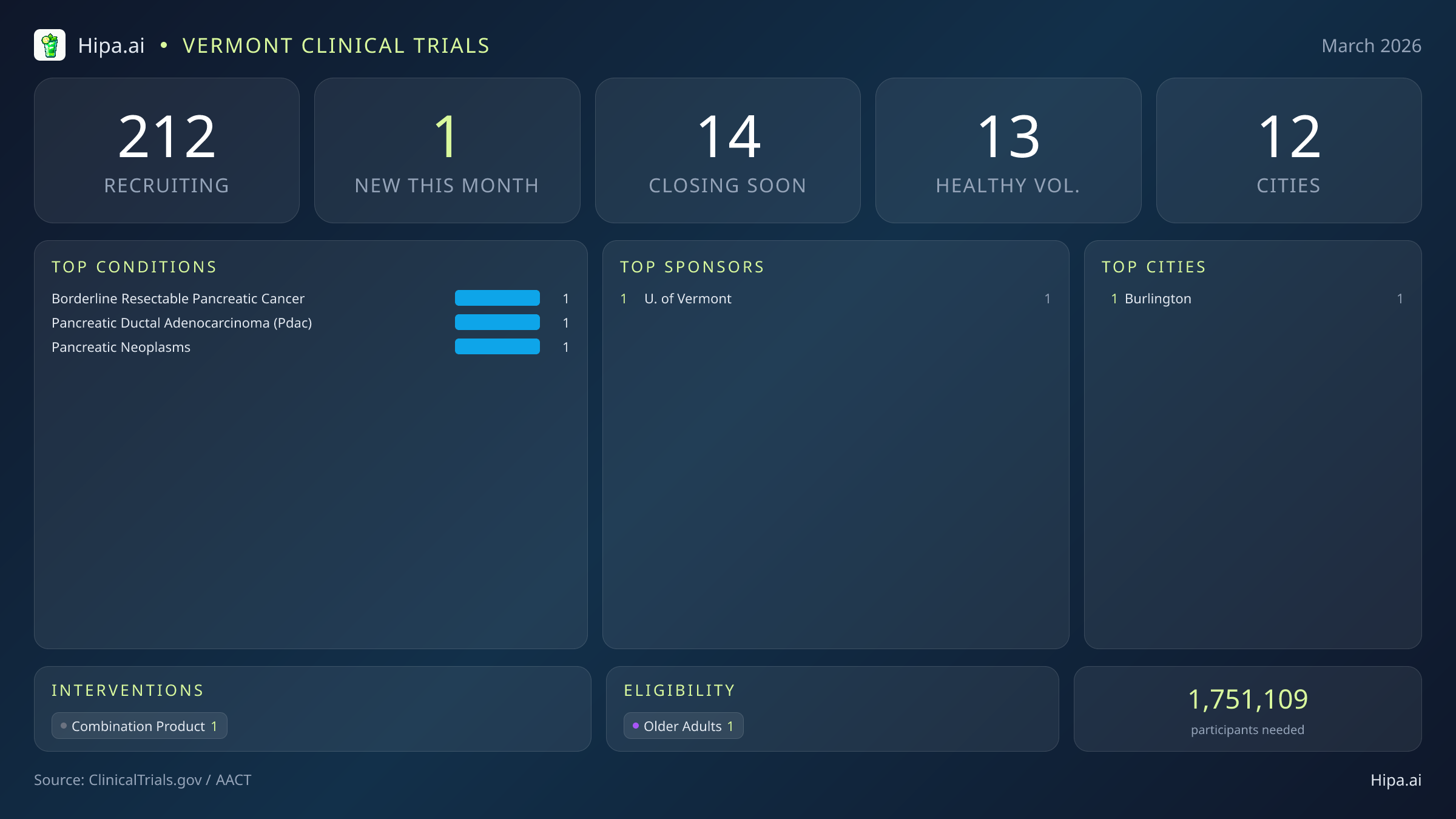
Vermont Clinical Trials Report — March 2026
1 New Studies, 14 Closing Soon
Hipa.ai Research - March 1, 2026 - Source: Research Data
Monthly Clinical Trials Infographic
Impending Deadlines for Vermont Research Participants
Time is strictly limited for Vermonters looking to participate in several critical medical studies currently operating within the state. Over the next ninety days, 14 clinical trials will permanently close their enrollment windows. This impending deadline creates a narrow window of opportunity for patients seeking access to experimental therapies before these studies transition out of the public recruitment phase and into closed data analysis. Among these closing studies, researchers are urgently seeking participants to fill the final available spots. Notably, individuals without underlying health conditions still have an immediate chance to contribute to scientific advancement, as 2 of these closing trials are actively accepting healthy volunteers.
For patients currently managing specific, severe diagnoses, the cutoff dates are approaching rapidly. The closure of these trials means that access to these particular experimental interventions, behavioral therapies, and medical devices will soon be restricted. Final enrollment phases are approaching quickly for studies focused on the following conditions:
- Stage IV lung cancer
- Coronary artery disease and coronary stenosis
- Cystic fibrosis, including pediatric cases
- Generalized anxiety disorder
- Acute myeloid leukemia
- Interstitial lung disease
- Developmental delay
Academic Innovation in Pancreatic Oncology
March brings a highly targeted, albeit quiet, focus to Vermont's research landscape. Following a more active winter season that saw five studies launch in February, three in January, and five in December, research initiations have slowed significantly. Just 1 new clinical trial opened its doors this month, matching the similarly slow output seen last November. Sponsored by the University of Vermont, this newly launched study highlights the state's strong reliance on academic institutions to drive local medical innovation, particularly in specialized fields where commercial pharmaceutical companies may not be currently initiating local sites.
Rather than a traditional oral medication or standard intravenous drug, this month's sole addition is evaluating a combination product. This therapeutic approach typically integrates a drug, device, or biologic into a single treatment protocol, such as a localized delivery system or a biologically coated surgical implant. The University of Vermont research team is specifically recruiting patients facing severe gastrointestinal diagnoses. Individuals currently managing the following conditions are encouraged to explore this new enrollment opportunity:
- Borderline resectable pancreatic cancer
- Pancreatic ductal adenocarcinoma
- General pancreatic neoplasms
While this month does not feature any massive late-stage pharmaceutical trials or Phase 3 studies enrolling thousands of patients, the focus on complex pancreatic cancers provides a vital resource. Pancreatic ductal adenocarcinoma remains one of the most challenging conditions to treat using standard therapies, making access to experimental combination products a critical option for local patients requiring advanced, specialized oncological care.
Statewide Research Footprint and Accessibility
Despite the quiet month for new study launches, Vermont maintains a robust and highly accessible clinical research infrastructure for a state of its population size. There are currently 212 active clinical trials recruiting participants throughout the state. These research initiatives are distributed across 12 different cities, ensuring that residents living outside the immediate metropolitan centers still have viable pathways to experimental care and do not necessarily have to cross state lines to find advanced treatment options.
The state's clinical trial activity operates through 57 distinct research sites, ranging from large hospital systems to specialized private clinics and academic laboratories. Unsurprisingly, Burlington continues to serve as the primary hub for medical advancement in the region. As the home of the state's major academic and medical centers, Burlington is the exclusive host city for this month's newly launched pancreatic cancer study. However, the broader network of active trials ensures that patients managing chronic conditions or seeking specialized care can often find research opportunities closer to home, reducing the travel burden typically associated with clinical trial participation.
Demographics and Healthy Volunteer Participation
Eligibility for March's newly opened pancreatic cancer study is tailored specifically toward adult populations. The protocol is designed to include older adults, ensuring that senior demographics—who are disproportionately affected by pancreatic neoplasms—have direct access to this combination product therapy. The study does not restrict participation based on gender, remaining open to both male and female patients, though it does exclude pediatric participants entirely.
For Vermont residents who do not have a cancer diagnosis or other underlying medical conditions, participation in medical research remains highly accessible. While the single new trial launched this month requires a specific oncological diagnosis and does not accept healthy individuals, the broader statewide network relies heavily on community participation. Across Vermont, 13 active clinical trials are currently recruiting healthy volunteers.
These individuals play an essential role in the clinical research ecosystem. Healthy volunteers provide the baseline health metrics that investigators need to evaluate the safety, tolerability, and efficacy of new treatments accurately. By participating, healthy individuals help researchers understand how a new intervention behaves in a system free of disease, which is a mandatory step before treatments can be widely administered to sick patients. Without the participation of healthy individuals, the progression of experimental therapies from the laboratory to the pharmacy shelf would be impossible.
As the spring season progresses, the concentration of academic-led oncology research in Burlington is expected to pave the way for more specialized combination therapies, potentially expanding treatment avenues for complex gastrointestinal cancers across the broader New England medical corridor.
Data Highlights
Conditions Closing Soon
- stage iv lung cancer ajcc v8 (2)
- coronary artery disease (2)
- cystic fibrosis (2)
- coronary artery calcification (1)
- coronary stenosis (1)
- cystic fibrosis in children (1)
- developmental delay (1)
- generalized anxiety disorder (1)
Most Common New Trial Conditions
- borderline resectable pancreatic cancer (1)
- pancreatic ductal adenocarcinoma (pdac) (1)
- pancreatic neoplasms (1)
Cities With the Most New Trials
- Burlington (1)
Leading Sponsors
- University of Vermont (1)
| Month | New Trials | Closing Soon |
|---|---|---|
| November 2025 | 1 | 0 |
| December 2025 | 5 | 4 |
| January 2026 | 3 | 0 |
| February 2026 | 5 | 1 |
| March 2026 | 1 | 3 |
New Studies This Month (1)
| NCT ID | Title | Phase | Enrollment | Sponsor | Condition | City |
|---|---|---|---|---|---|---|
| NCT07477418 | PDAC Regression and Intraoperative Surgical Margin With Neoadjuvant TAMP (PRISM-TAMP) | Phase 1/PHASE2 | 10 | University of Vermont | Pancreatic Ductal Adenocarcinoma (PDAC) | Burlington |