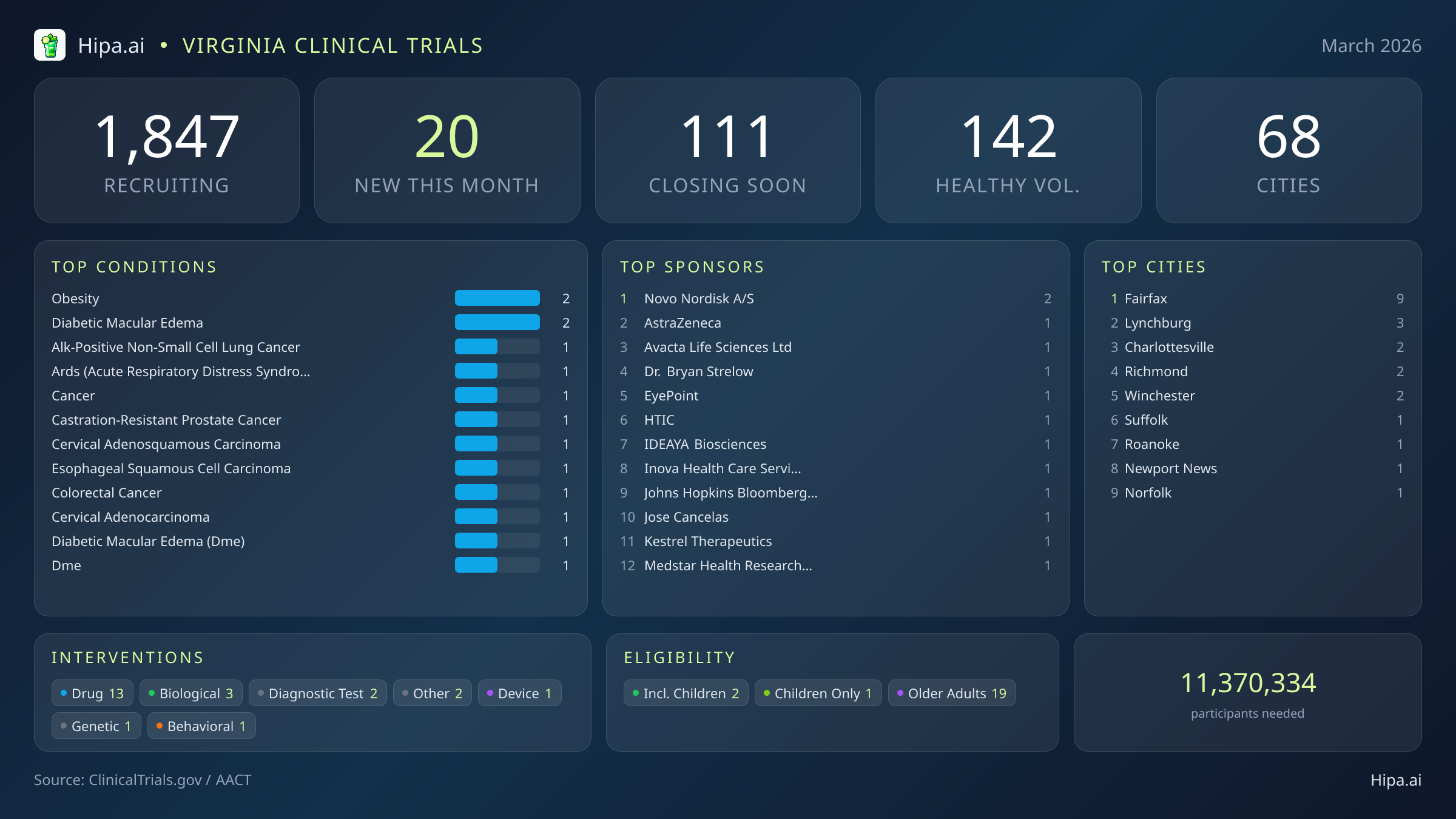
Virginia Clinical Trials Report — March 2026
20 New Studies, 111 Closing Soon
Hipa.ai Research - March 1, 2026 - Source: Research Data
Monthly Clinical Trials Infographic
Virginians have a rapidly shrinking timeframe to participate in several critical medical research studies. Within the next ninety days, 111 clinical trials across the state will permanently close their enrollment windows. This impending deadline affects both patients seeking novel therapies and individuals without underlying conditions. Specifically, 17 of these soon-to-close studies are actively looking for healthy volunteers to help establish baseline medical data. For patients currently managing specific diagnoses, the window to access experimental treatments is closing fast. Final enrollment phases are approaching for studies focused on the following conditions:
- Post-traumatic stress disorder
- Stage IV lung cancer
- Breast cancer
- Kidney transplant complications
- Multiple myeloma
- Prostate cancer
- Coronary artery disease
- Cholangiocarcinoma
- Focal segmental glomerulosclerosis
- Interstitial lung disease
Major Phase 3 Pharmaceutical and Vaccine Launches
Despite the wave of closures, March brings a fresh influx of high-capacity research projects to the Commonwealth. The pace of new study launches has slowed slightly compared to the beginning of the year, with 20 new trials opening this month compared to thirty-seven in January. However, the caliber of these new investigations is exceptionally high, driven heavily by major pharmaceutical sponsors like Moderna, Pfizer, and Novo Nordisk. Industry sponsors are funding thirteen of these new initiatives, while academic and other institutions are driving the remaining seven.
Several massive Phase 3 trials are actively recruiting thousands of participants, offering Virginians a direct role in advancing global public health and chronic disease management:
- ModernaTX, Inc. is seeking four thousand healthy adults for a Phase 3 pandemic influenza vaccine trial (NCT07496450) to evaluate humoral immunogenicity, safety, and reactogenicity after two doses of the mRNA-1018-H5 vaccine.
- Pfizer is enrolling over seventeen hundred participants aged seven and older for a Phase 3 study (NCT07500506) testing the safety and protective capabilities of a fifth dose of the VLA15 Lyme disease vaccine.
- Novo Nordisk A/S has launched two major Phase 3 trials evaluating NNC0487-0111, an injectable medication for excess body weight. One study (NCT07503210) seeks six hundred participants to measure weight loss maintenance, while a parallel study (NCT07481630) is recruiting four hundred individuals to assess weight loss and pain reduction in patients with both excess weight and knee osteoarthritis.
- Virginia Commonwealth University is conducting a hospital-based intervention study (NCT07475247) for three hundred sixty youth injured through firearm violence, addressing a critical public health epidemic.
- Adverum Biotechnologies, Inc. is evaluating a single intravitreal injection of Ixo-vec in a Phase 3 trial (NCT07482176) for two hundred eighty-four patients with neovascular age-related macular degeneration.
- Johns Hopkins Bloomberg School of Public Health is recruiting two hundred fifty participants for a Phase 3 trial (NCT07458230) integrating next-generation sequencing to treat surgical site infections after severe bone fractures.
- EyePoint Pharmaceuticals, Inc. is comparing the efficacy of EYP-1901 against Aflibercept in a Phase 3 study (NCT07449936) involving two hundred forty patients with diabetic macular edema.
Targeted Diagnoses and Broad Eligibility
The therapeutic focus of this month's new research spans a wide array of chronic conditions, acute injuries, and preventative care. Oncology remains a primary target, with researchers actively seeking patients diagnosed with ALK-positive non-small cell lung cancer, castration-resistant prostate cancer, cervical adenosquamous carcinoma, esophageal squamous cell carcinoma, and colorectal cancer. Beyond cancer research, new protocols are targeting acute respiratory distress syndrome and diabetic macular edema.
The intervention types are heavily weighted toward pharmaceutical development, with 13 new drug trials entering the recruitment phase. The remaining studies are testing biological interventions, diagnostic tests, medical devices, genetic therapies, and behavioral modifications.
Eligibility criteria for these new studies ensure that diverse demographic groups can participate. Older adults have extensive access to this month's research, as nineteen of the new trials have designed their protocols to include senior populations. Pediatric research is also represented, with two new studies open to children and adolescents. None of the new trials restrict participation exclusively by gender. For those without a specific medical diagnosis, five of this month's new trials are actively welcoming healthy volunteers. This expands the total number of open, recruiting trials for healthy participants across Virginia to 142.
Geographic Distribution Across the Commonwealth
The clinical research infrastructure across Virginia remains robust and highly accessible. Currently, 1,847 active trials are recruiting across sixty-eight cities, utilizing over one thousand individual research sites. This extensive network ensures that both urban and rural residents have pathways to experimental therapies and preventative vaccines.
This month, new research opportunities are heavily clustered in Northern Virginia, though central and coastal regions also feature fresh trial launches. Fairfax leads the state with nine newly opened trials, serving as a major hub for the latest Phase 3 pharmaceutical investigations, largely supported by regional health systems like Inova Health Care Services. Meanwhile, academic centers continue to anchor research in central Virginia, with new studies launching in Charlottesville and Richmond. Residents living outside the immediate metropolitan areas still have excellent access to cutting-edge medical research. New trials have opened their doors this month in the following Virginia municipalities:
- Fairfax
- Lynchburg
- Charlottesville
- Richmond
- Winchester
- Suffolk
- Roanoke
- Newport News
- Norfolk
As the spring season progresses, Virginia's clinical research ecosystem is positioned to shift its focus toward the rapid enrollment of large-scale vaccine initiatives and the subsequent data analysis required to advance next-generation infectious disease protocols and weight management therapies.
Data Highlights
Conditions Closing Soon
- post traumatic stress disorder (4)
- stage iv lung cancer ajcc v8 (2)
- breast cancer (2)
- kidney transplant; complications (2)
- multiple myeloma (2)
- prostate cancer (2)
- coronary artery disease (2)
- cholangiocarcinoma (2)
Most Common New Trial Conditions
- obesity (2)
- diabetic macular edema (2)
- alk-positive non-small cell lung cancer (1)
- ards (acute respiratory distress syndrome) (1)
- cancer (1)
- castration-resistant prostate cancer (1)
- cervical adenosquamous carcinoma (1)
- esophageal squamous cell carcinoma (1)
Cities With the Most New Trials
- Fairfax (9)
- Lynchburg (3)
- Charlottesville (2)
- Richmond (2)
- Winchester (2)
- Suffolk (1)
- Roanoke (1)
- Newport News (1)
Leading Sponsors
- Novo Nordisk A/S (2)
- AstraZeneca (1)
- Avacta Life Sciences Ltd (1)
- Dr. Bryan Strelow (1)
- EyePoint Pharmaceuticals, Inc. (1)
- HTIC, Inc (1)
- IDEAYA Biosciences (1)
- Inova Health Care Services (1)
| Month | New Trials | Closing Soon |
|---|---|---|
| November 2025 | 25 | 6 |
| December 2025 | 59 | 35 |
| January 2026 | 31 | 8 |
| February 2026 | 35 | 14 |
| March 2026 | 20 | 34 |
| April 2026 | 3 | 0 |