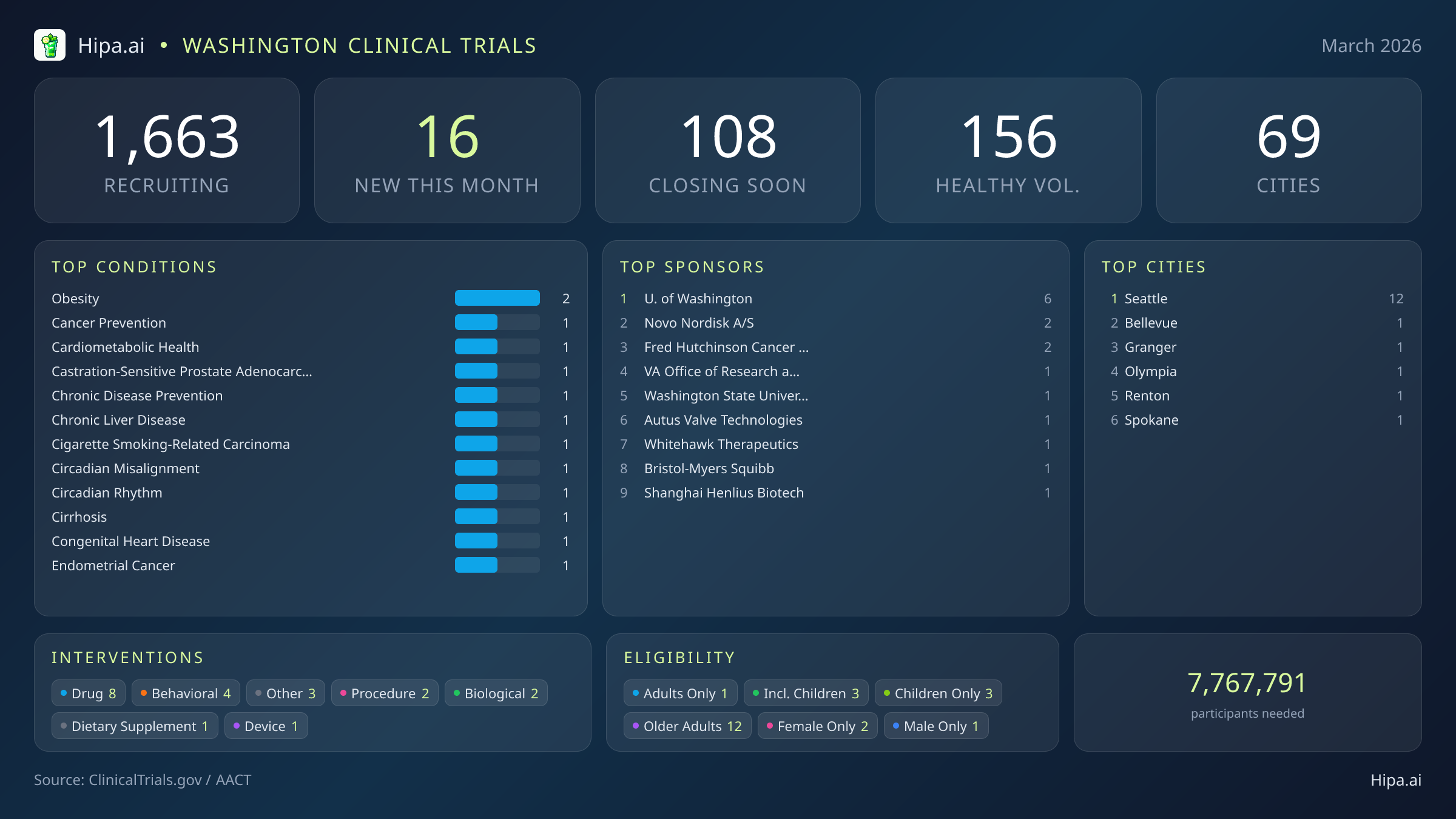
Washington Clinical Trials Report — March 2026
16 New Studies, 108 Closing Soon
Hipa.ai Research - March 1, 2026 - Source: Research Data
Monthly Clinical Trials Infographic
Approaching Deadlines for Washington Residents
Time is strictly limited for Washington residents seeking to enroll in several critical medical studies. Within the next ninety days, 108 clinical trials across the state will permanently close their enrollment windows. This impending deadline affects patients seeking experimental therapies for severe diagnoses, but it also creates an immediate window for individuals without underlying health conditions. Currently, 14 of these soon-to-close studies are actively seeking healthy volunteers to provide essential baseline data. Patients managing specific chronic and acute conditions face a rapidly shrinking timeframe to participate in research focused on the following areas:
- Multiple myeloma and hematopoietic neoplasms
- Post-traumatic stress disorder
- Stage IV lung cancer
- Major depressive disorder
- Alcohol use disorder
- Chronic spontaneous urticaria
- Heart failure
High-Profile Pharmaceutical and Behavioral Launches
March brings a highly specialized wave of research to the Pacific Northwest, heavily driven by major pharmaceutical companies and leading academic institutions. While the overall pace of new study launches has cooled from the winter peak—dropping from thirty-two new trials in January to 16 this month—the caliber of the research remains exceptional. Half of this month's new studies are focused on drug interventions, with industry giants like Novo Nordisk and Bristol-Myers Squibb initiating late-stage trials. Behavioral and observational research also features prominently, spearheaded by the University of Washington and the Fred Hutchinson Cancer Center. Several standout trials are currently seeking participants:
- Fred Hutchinson Cancer Center is recruiting over eighteen hundred individuals for a massive behavioral study (NCT07493252) comparing two smartphone applications, Actify! A and B, designed to improve smoking cessation outcomes using behavioral activation therapy.
- The VA Office of Research and Development has launched the NOSE Study (NCT07469670), seeking over one thousand participants to evaluate non-traditional nasal swabs to combat supply chain shortages for respiratory testing.
- Novo Nordisk is enrolling six hundred participants in a Phase 3 trial (NCT07503210) to determine if weekly subcutaneous injections of NNC0487-0111 are safe and effective for weight maintenance in people with excess body weight.
- A parallel Phase 3 study by Novo Nordisk (NCT07481630) is recruiting four hundred individuals to test the same medication's ability to reduce weight and alleviate pain in patients with both excess body weight and knee osteoarthritis.
- The University of Washington is sponsoring a community-based trial (NCT07490652) aiming to increase access to healthier foods for rural Latino communities through interventions at small, independent grocery stores.
- Bristol-Myers Squibb is seeking two hundred sixty participants for a Phase 2 study (NCT07492680) evaluating BMS-986504, both as a monotherapy and in combination with other agents, for advanced solid tumors featuring a specific MTAP deletion.
- Shanghai Henlius Biotech has initiated a Phase 1 trial (NCT07477587) comparing the safety and efficacy of HLX15-SC against DARZALEX FASPRO for transplant-ineligible patients with newly diagnosed multiple myeloma.
Targeted Conditions and Demographic Reach
March's new clinical trials target a highly specific array of metabolic, oncological, and behavioral conditions. Researchers are actively seeking patients recently diagnosed with or currently managing obesity, castration-sensitive prostate adenocarcinoma, chronic liver disease, cirrhosis, and circadian rhythm misalignment. Preventive medicine is also a major focus this month, with new studies dedicated to cancer prevention, cardiometabolic health, and general chronic disease prevention. The University of Washington is expanding pediatric behavioral research with a new study (NCT07446413) testing an autism-friendly mobile app designed to improve toothbrushing habits in children and teens, comparing it against a standard digital mirror and countdown timer.
Eligibility criteria for this month's new studies ensure broad demographic participation. Older adults have extensive options, with twelve of the new trials specifically designing their protocols to include senior populations. Pediatric research is represented by three new studies open to children and adolescents. While most trials are open to all genders, two are exclusively seeking female participants, and one is restricted to male participants.
For those without a specific medical diagnosis, four of this month's new trials are actively welcoming healthy volunteers. This brings the total number of open, recruiting trials for healthy participants across Washington to 156. These volunteers are critical for establishing baseline safety and efficacy data for both pharmaceutical interventions and behavioral health applications.
Research Hubs and Statewide Accessibility
Washington's clinical research infrastructure remains robust and highly accessible, with 1,663 active trials currently recruiting across 69 cities and 797 individual research sites. The geographic distribution of this month's new studies heavily favors the Puget Sound region, driven by the concentration of major research institutions and medical centers in King County.
Seattle dominates the landscape, hosting twelve of the newly opened trials. However, residents living outside the immediate Seattle metropolitan area still have opportunities to participate in cutting-edge research. New trials have opened their doors this month in several key municipalities across the state:
- Seattle
- Bellevue
- Granger
- Olympia
- Renton
- Spokane
As the spring research season accelerates, Washington's clinical trial landscape is positioned to expand its focus on cardiometabolic therapies and digital health interventions, while the massive datasets collected from this month's behavioral and supply-chain studies will likely shape the next generation of public health infrastructure.
Data Highlights
Conditions Closing Soon
- multiple myeloma (4)
- post traumatic stress disorder (3)
- ptsd (2)
- hematopoietic and lymphatic system neoplasm (2)
- stage iv lung cancer ajcc v8 (2)
- alcohol use disorder (aud) (2)
- depressive disorder, major (2)
- chronic spontaneous urticaria (2)
Most Common New Trial Conditions
- obesity (2)
- cancer prevention (1)
- cardiometabolic health (1)
- castration-sensitive prostate adenocarcinoma (1)
- chronic disease prevention (1)
- chronic liver disease (1)
- cigarette smoking-related carcinoma (1)
- circadian misalignment (1)
Leading Sponsors
- University of Washington (6)
- Novo Nordisk A/S (2)
- Fred Hutchinson Cancer Center (2)
- VA Office of Research and Development (1)
- Washington State University (1)
- Autus Valve Technologies, Inc. (1)
- Whitehawk Therapeutics, Inc. (1)
- Bristol-Myers Squibb (1)
| Month | New Trials | Closing Soon |
|---|---|---|
| November 2025 | 25 | 6 |
| December 2025 | 36 | 29 |
| January 2026 | 15 | 4 |
| February 2026 | 31 | 11 |
| March 2026 | 16 | 37 |
| April 2026 | 5 | 0 |