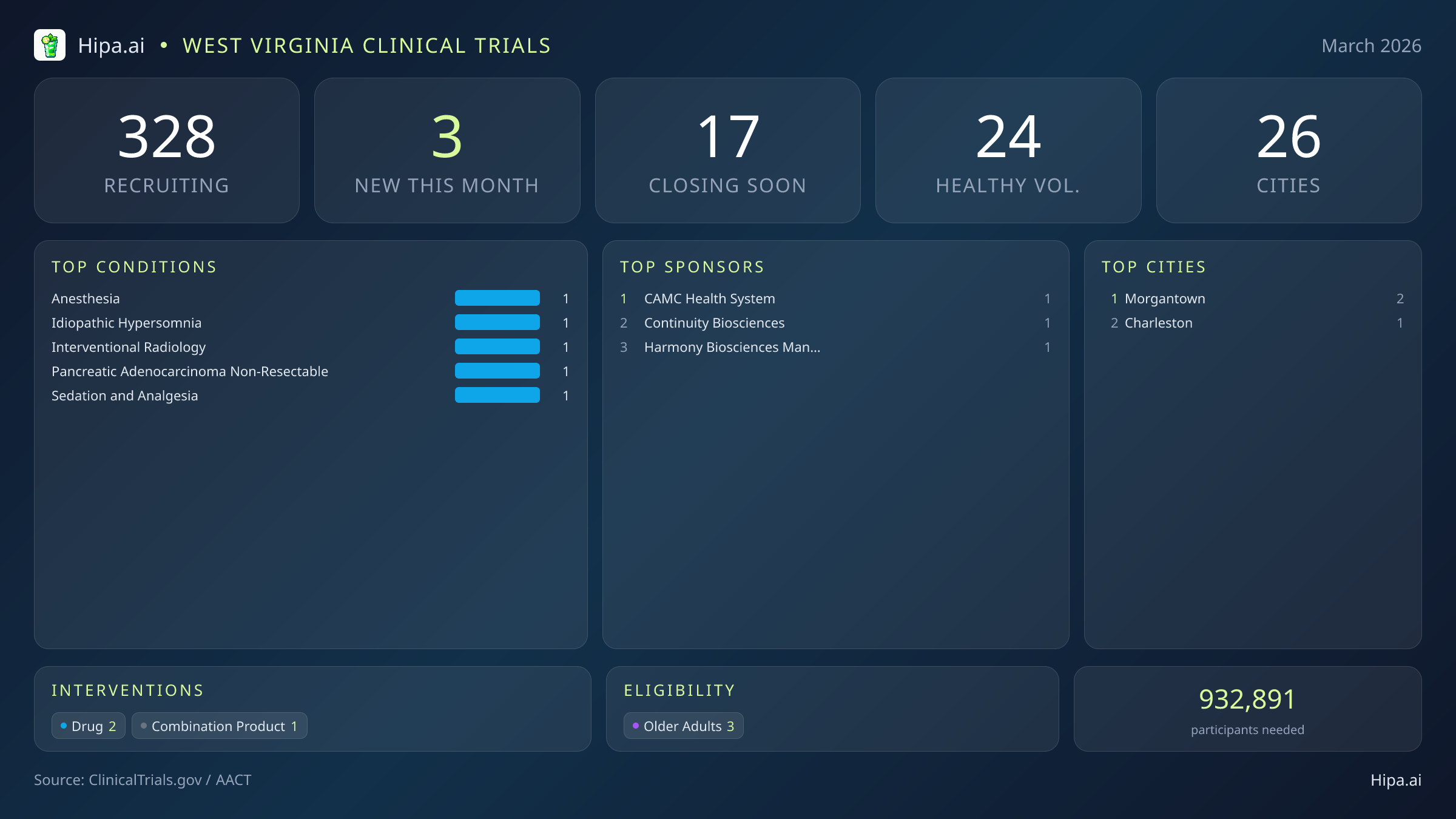
West Virginia Clinical Trials Report — March 2026
3 New Studies, 17 Closing Soon
Hipa.ai Research - March 1, 2026 - Source: Research Data
Monthly Clinical Trials Infographic
Critical Enrollment Windows Closing for West Virginia Patients
Time is rapidly running out for West Virginia residents seeking access to several advanced medical research programs. Over the next ninety days, exactly 17 clinical trials will permanently close their enrollment windows across the state. This impending deadline requires immediate action from patients managing specific, often severe, diagnoses who wish to explore experimental treatment avenues. Because none of these closing studies are accepting healthy volunteers, the remaining open slots are exclusively reserved for individuals requiring targeted therapeutic interventions. Patients must begin the screening process immediately to secure a place before these studies transition into their data analysis phases. The window to access these experimental therapies is narrowing quickly for studies focused on the following conditions:
- Stage IV lung cancer
- Anatomic stage II and stage III breast cancer
- Circadian rhythm disorders
- Bilateral nasal polyposis
- Cancer harboring BRAF alterations
- Chronic hepatitis C
- HER2-negative breast carcinoma
In addition to the conditions listed above, patients managing cardiovascular diseases and high-grade gliomas also face immediate deadlines to participate in specialized research protocols. Once these enrollment periods conclude, access to these specific experimental drugs and interventions will be restricted until potential future phases of research are authorized.
Phase 3 Sleep Research and Emerging Interventions
March brings a highly targeted wave of research to the state, with 3 new clinical trials opening their doors to participants. This represents a slight deceleration in new research launches compared to the six studies initiated earlier this year in January, reflecting a shift toward highly specialized, late-stage investigations rather than broad early-phase testing. Industry sponsors are driving the majority of this month's new clinical development, offering local patients access to experimental treatments before they reach the broader commercial market.
The most prominent addition to the state's research portfolio is a major late-stage drug trial focused on severe sleep disorders. Harmony Biosciences Management, Inc. is actively recruiting 248 participants for a Phase 3 trial (NCT07500090) investigating HBS-301. This multicenter, randomized, double-blind, parallel-group, placebo-controlled clinical study is designed to assess the efficacy and safety of the experimental drug in treating idiopathic hypersomnia. Researchers are specifically evaluating whether the intervention can alleviate debilitating symptoms such as excessive daytime sleepiness, sleep inertia, and chronic fatigue in adult participants. Because this is a Phase 3 trial, it represents one of the final steps in the clinical evaluation process before potential regulatory approval.
Beyond this late-stage sleep study, regional healthcare leaders and emerging biotechnology firms are also launching new initiatives. CAMC Health System and Continuity Biosciences, LLC have initiated protocols focusing on a diverse array of medical needs. Two of this month's new studies involve experimental drug therapies, while the third focuses on a combination product that merges pharmaceutical and device-based approaches. These newly launched trials are actively seeking patients recently diagnosed with or currently managing the following conditions:
- Idiopathic hypersomnia
- Non-resectable pancreatic adenocarcinoma
- Conditions requiring sedation and analgesia
- Procedures involving interventional radiology
- General anesthesia requirements
Research Hubs and Statewide Accessibility
The broader clinical research infrastructure in West Virginia remains robust and highly accessible to residents across multiple regions, overcoming the geographic challenges often associated with mountainous terrain. Currently, 328 active clinical trials are recruiting participants statewide. These research opportunities are distributed across 26 different cities, utilizing a vast network of 160 distinct clinical research sites. This extensive footprint ensures that clinical research is not solely confined to major academic centers but is integrated into community clinics and regional hospitals.
Geographically, this month's new research opportunities are heavily clustered in the state's established medical and academic corridors. Morgantown leads the state's new research initiatives in March, hosting two of the newly opened trials. This concentration provides residents in the northern part of the state with direct access to advanced care options and experimental therapies. Meanwhile, Charleston has added one new clinical trial to its active roster, ensuring that patients in the capital region continue to see fresh opportunities for medical participation. While the newest studies are concentrated in these two major metropolitan areas, the vast network of hundreds of ongoing trials ensures that residents living outside these immediate urban centers still have viable pathways to participate in medical research.
Adult Eligibility and Healthy Volunteer Opportunities
Eligibility criteria for the studies launched this month are entirely focused on adult populations, ensuring that mature demographics can actively participate in medical advancement. All three of the new trials have designed their protocols to include older adults, reflecting a strong commitment to understanding how experimental therapies affect aging populations and ensuring that seniors are represented in vital safety data. None of the new studies restrict participation exclusively to male or female patients, ensuring broad gender access for the targeted conditions. Additionally, pediatric research is not represented in this month's new launches, as zero trials are currently open to children.
For individuals without a specific medical diagnosis, the immediate options in this month's new cohort are limited, as none of the newly launched trials are accepting healthy volunteers. However, residents looking to contribute to scientific discovery still have significant opportunities available. A total of 24 active trials are currently recruiting healthy volunteers across West Virginia. These ongoing studies rely heavily on individuals without underlying conditions to establish crucial baseline data, evaluate safety profiles, and drive future medical breakthroughs. Healthy participants often serve as the critical control group against which new therapies are measured, making their involvement essential to the scientific process.
As the spring research season progresses, West Virginia's clinical trial landscape is positioned to expand its focus on targeted oncological therapies and advanced neurological interventions, while the integration of specialized combination products will likely shape the next wave of regional medical development.
Data Highlights
Conditions Closing Soon
- stage iv lung cancer ajcc v8 (2)
- anatomic stage ii breast cancer ajcc v8 (1)
- circadian rhythm disorders (1)
- bilateral nasal polyposis (1)
- cancer harboring braf alterations (1)
- cardiovascular diseases (1)
- anatomic stage iii breast cancer ajcc v8 (1)
- hepatitis c, chronic (1)
Most Common New Trial Conditions
- anesthesia (1)
- idiopathic hypersomnia (1)
- interventional radiology (1)
- pancreatic adenocarcinoma non-resectable (1)
- sedation and analgesia (1)
Cities With the Most New Trials
- Morgantown (2)
- Charleston (1)
Leading Sponsors
- CAMC Health System (1)
- Continuity Biosciences, LLC (1)
- Harmony Biosciences Management, Inc. (1)
| Month | New Trials | Closing Soon |
|---|---|---|
| October 2025 | 9 | 0 |
| November 2025 | 3 | 1 |
| December 2025 | 5 | 5 |
| January 2026 | 5 | 2 |
| February 2026 | 6 | 0 |
| March 2026 | 3 | 6 |
New Studies This Month (3)
| NCT ID | Title | Phase | Enrollment | Sponsor | Condition | City |
|---|---|---|---|---|---|---|
| NCT07443254 | Prospective IR-led Sedation Feasibility | Phase 1/PHASE2 | 40 | CAMC Health System | Anesthesia | Charleston |
| NCT07481383 | A First-in-Human Study to Evaluate Implantable Iontophoresis Chemotherapy Delivery Device With Gemcitabine Once Weekly or Twice Weekly in Participants With Pancreatic Cancer | Phase 1 | 12 | Continuity Biosciences, LLC | Pancreatic Adenocarcinoma Non-resectable | Morgantown |
| NCT07500090 | A Phase 3 Efficacy and Safety Study of HBS-301 in Participants With Idiopathic Hypersomnia (IH) | Phase 3 | 248 | Harmony Biosciences Management, Inc. | Idiopathic Hypersomnia | Morgantown |