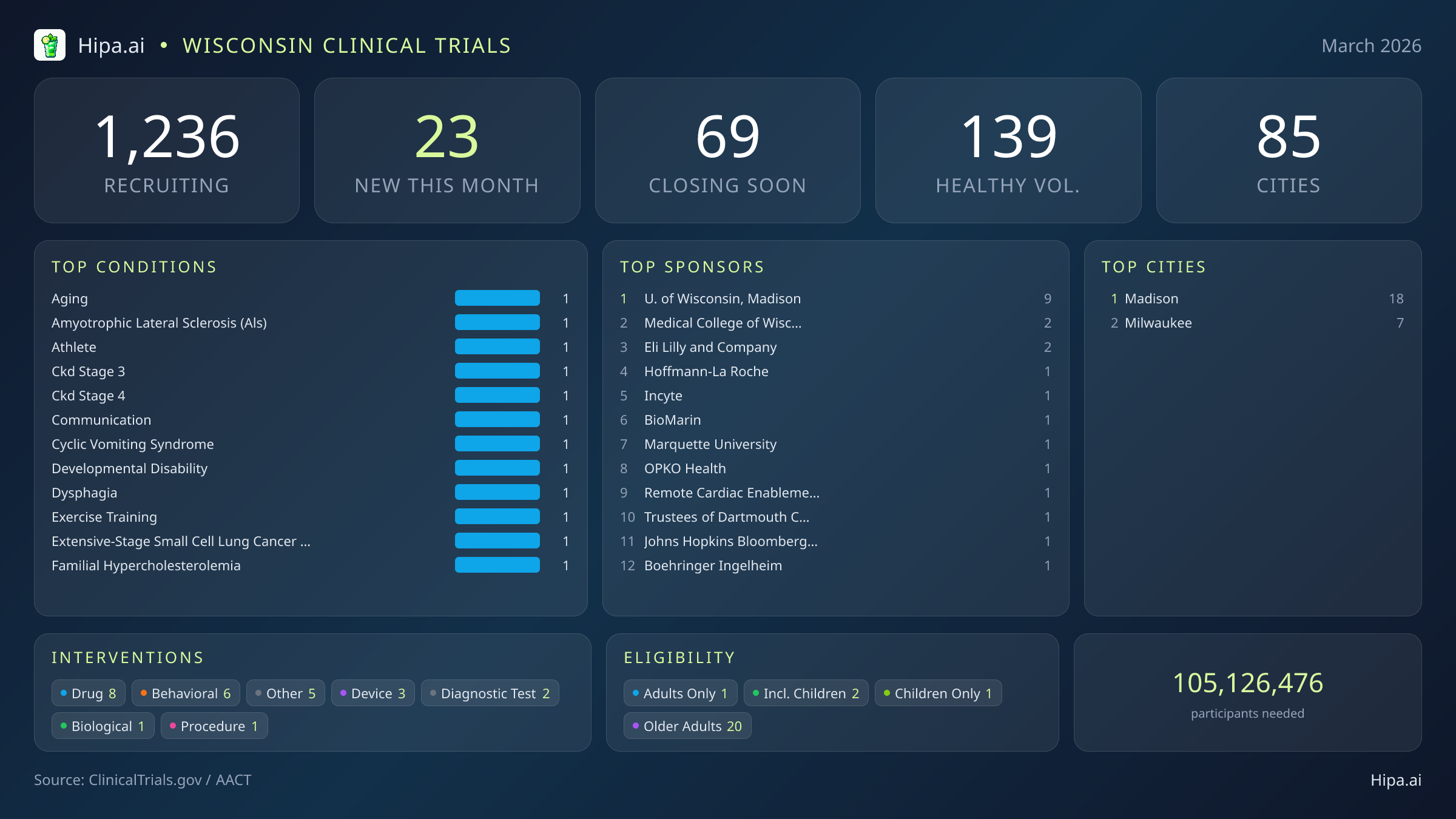
Wisconsin Clinical Trials Report — March 2026
23 New Studies, 69 Closing Soon
Hipa.ai Research - March 1, 2026 - Source: Research Data
Monthly Clinical Trials Infographic
Approaching Deadlines for Wisconsin Research Participants
Time is strictly limited for Wisconsin residents hoping to join several critical medical studies. Over the next ninety days, 69 clinical trials will permanently close their enrollment windows across the state. This impending deadline creates a narrow window of opportunity for patients seeking access to experimental therapies before researchers transition into the data analysis phase. People without underlying medical conditions also need to act quickly, as 13 of these soon-to-close studies are actively seeking healthy volunteers to establish vital baseline data. Patients currently managing specific diagnoses, as well as medical professionals experiencing occupational stress, face immediate deadlines to participate in research targeting the following conditions:
- Stroke
- Pancreatic cancer
- Metastatic cancer
- Recurrent endometrial carcinoma
- Stage IV lung cancer
- Paroxysmal atrial fibrillation
- Cholangiocarcinoma
- Healthcare worker burnout
High-Profile Diagnostics and Late-Stage Therapeutics
March brings a robust expansion to the state's research portfolio, with 23 new trials opening their doors—a steady increase from the twenty studies launched at the start of the year. Pharmaceutical and biotechnology leaders are driving significant innovation this month, particularly in the realms of oncology, neurology, and diagnostic testing. Eight of these newly launched studies focus on experimental drug interventions, while device and diagnostic test trials are also making a strong showing. Several major industry-sponsored studies are currently seeking participants:
- Hoffmann-La Roche is recruiting two thousand participants for a massive diagnostic study (NCT07455136) to develop a blood-based biomarker test that aids in diagnosing and predicting outcomes for adults with traumatic brain injuries.
- Boehringer Ingelheim has launched a Phase 3 trial (NCT07472517) enrolling six hundred seventy adults with advanced small cell lung cancer to determine if adding obrixtamig to standard chemotherapy improves overall survival rates.
- Eli Lilly and Company is seeking four hundred fifty healthy participants for a Phase 1 study (NCT07446101) assessing the safety and tolerability of a new solution formulation of mirikizumab administered via injection.
- Remote Cardiac Enablement is conducting a device trial (NCT07464704) with three hundred fifty participants to validate the Infrasensor's ability to detect high-grade obstructive non-ST-segment elevation acute coronary syndrome.
Academic Leadership and Behavioral Health Initiatives
Beyond pharmaceutical development, Wisconsin's academic institutions are spearheading large-scale behavioral and observational research. The University of Wisconsin, Madison, is sponsoring nine of this month's new trials, heavily emphasizing patient communication and community health. One standout initiative (NCT07475117) aims to enroll over one thousand participants to evaluate whether health coaching embedded within Special Olympics programming improves healthcare access for adults with intellectual or developmental disabilities. Another major university-led study (NCT07475104) is recruiting six hundred sixty patients to test an evidence-based communication framework designed to help surgical patients better understand treatment downsides and expectations.
The Medical College of Wisconsin is also launching critical rehabilitation research, including a trial (NCT07463027) focused on veterans and first responders. This study will evaluate an interdisciplinary outpatient program designed to reduce long-lasting symptoms of traumatic brain injury and co-occurring disorders. Additionally, Dartmouth College is sponsoring a multi-state effort (NCT07454733) that includes Wisconsin sites, investigating whether video recordings of multidisciplinary clinic visits can improve the quality of life for patients with amyotrophic lateral sclerosis.
This month's newly opened trials cater to a highly specific array of medical needs. Researchers are actively recruiting individuals diagnosed with or experiencing the following conditions:
- Amyotrophic lateral sclerosis
- Chronic kidney disease (Stages 3 and 4)
- Cyclic vomiting syndrome
- Developmental disabilities
- Dysphagia
- General aging and athletic exercise training
Research Accessibility Across the State
Wisconsin maintains a highly accessible clinical research infrastructure, boasting 1,236 active trials currently recruiting across 85 different cities. These studies are distributed among more than six hundred individual research sites, ensuring that cutting-edge medical advancements are available to both urban and rural populations.
While the state's overall geographic footprint is broad, new research opportunities launched this month are heavily clustered in Wisconsin's primary metropolitan hubs. Madison leads the state by a wide margin, hosting eighteen of the newly opened trials. Milwaukee follows as the second major center for new research, providing seven fresh opportunities for residents in the southeastern region. This concentration in major university and medical centers means that patients seeking the newest experimental therapies or behavioral interventions will likely need to travel to these primary academic hubs to participate.
Inclusive Enrollment and Opportunities for Healthy Volunteers
The eligibility criteria for March's new studies demonstrate a strong commitment to inclusive research. Older adults have extensive opportunities to participate, with twenty of the newly launched trials specifically designing their protocols to accommodate senior populations. Pediatric research is also moving forward, as two new studies are open to children and adolescents. Furthermore, none of the trials launched this month restrict participation exclusively by sex, ensuring equal access for both male and female volunteers.
Individuals without specific medical diagnoses play a crucial role in advancing this month's research agenda. Of the newly opened studies, 8 are actively welcoming healthy volunteers. This influx brings the total number of open, recruiting trials for healthy participants across Wisconsin to 139. Whether participating in Eli Lilly's early-stage safety testing or contributing baseline blood samples for Hoffmann-La Roche's traumatic brain injury diagnostics, healthy individuals are essential for establishing the safety and efficacy of tomorrow's medical breakthroughs.
As the spring research season accelerates, Wisconsin's clinical trial landscape is positioned to drive significant advancements in neurological diagnostics, surgical communication frameworks, and targeted oncology treatments, ultimately shaping more personalized and effective patient care models for the future.
Data Highlights
Conditions Closing Soon
- stroke (3)
- pancreatic cancer (2)
- metastatic cancer (2)
- recurrent endometrial carcinoma (2)
- stage iv lung cancer ajcc v8 (2)
- paroxysmal atrial fibrillation (2)
- cholangiocarcinoma (2)
- healthy (2)
Most Common New Trial Conditions
- aging (1)
- amyotrophic lateral sclerosis (als) (1)
- athlete (1)
- ckd stage 3 (1)
- ckd stage 4 (1)
- communication (1)
- cyclic vomiting syndrome (1)
- developmental disability (1)
Leading Sponsors
- University of Wisconsin, Madison (9)
- Medical College of Wisconsin (2)
- Eli Lilly and Company (2)
- Hoffmann-La Roche (1)
- Incyte Corporation (1)
- BioMarin Pharmaceutical (1)
- Marquette University (1)
- OPKO Health, Inc. (1)
| Month | New Trials | Closing Soon |
|---|---|---|
| November 2025 | 25 | 4 |
| December 2025 | 26 | 15 |
| January 2026 | 14 | 4 |
| February 2026 | 20 | 8 |
| March 2026 | 23 | 18 |
| April 2026 | 4 | 0 |